Lecture 6/7-Human Viruses & Disease
1/51
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
52 Terms
Asymptomatic
Infection that does not lead to disease
How does infection occur?
Virus enters a host and multiplies
When does Disease Occur?
host’s body cells are damaged as a result of infection and signs/symptoms of an illness appear
Immune Symptom
Activated in response to infection
signs/symptoms of disease result from immune system activity
Viral Virulence
Capacity of a virus to cause disease in a host
Virulent Virus Strain
causes significant disease
Avirulent/Attenuated Virus
causes no or reduced disease
Animal Models for Virus Study
Animal viruses that resemble human infection
Human viruses in animals
Plaque Assay
a technique to measure virus titer (determining the quantity of infectious virus) and to biologically clone a virus
Virus Propagation Allows
Vaccine development by providing viral antigens
Greater knowledge about virus lifecycle
Greater knowledge about virus pathogenicity
Anti-viral drug development
Measuring Virulence
Virus Titer
Mean time to death
Mean time to appearance of signs
Measurement of fever/weight loss
Measurement of Pathological Indicators
LD50
ID50
Virus Titer
measures the concentration of infectious or total virus particles in a sample
LD50
Lethal dose for 50% death
ID50
infectious dose for 50% symptoms
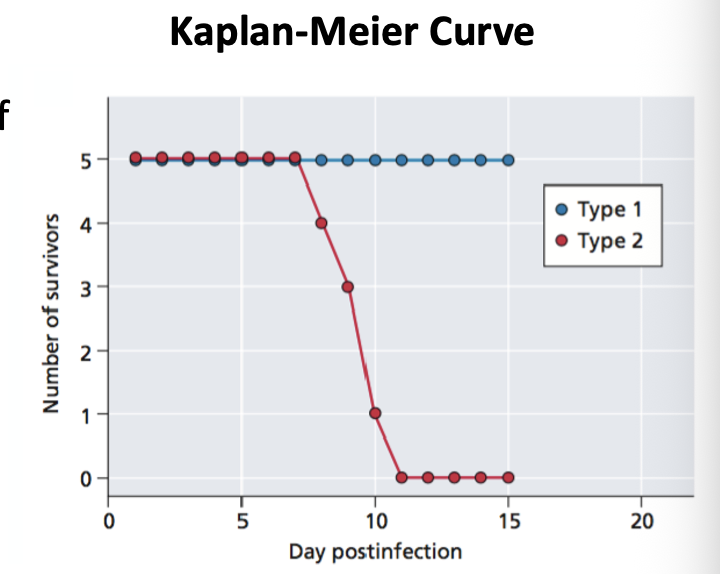
Kaplan-Meier Curve
a step-plot graph that visualizes the probability of an event (e.g., survival, disease relapse) over time
Factors Influencing Viral Virulence
Dose
Route of infection
Species
Age
Gender
Susceptibility of host
Social factors
Economic factors
Genetic Determinants of Viral Virulence
Major goal of virology
Discovering Genetic Determinants of Viral Virulence
Usually identified by mutation
A virus that causes reduced/no disease in a specified system
Viral Virulence Genes May
Affect ability of the virus to replicate
Enable the virus to spread within a host or between hosts
Defeat host defence mechanisms
Have intrinsic cell killing effects
Cross-species Virus Transmission and Virulence (Increase)
Cause of some of the most devastating epidemics and is associated with an increase in virulence
ex: jump of SIV and non-human primates to HIV in humans
Cross-species Virus Transmission and Virulence (No Change)
No apparent change in virulence following a host jump
ex: Influenza A virus H3N8 that jumped from horses to dogs
Cross-species Virus Transmission and Virulence (Decrease)
Often overlooked
ex: decreased virulence when infectious haematopoietic necrosis virus spread from salmon to trout
Pathogenesis
the process by which virus infection leads to disease
Disease occurs only if the virus replicates sufficiently
Damage essential cells directly
cause the release of toxic substances from infected tissues
damage cellular genes
damage organ function indirectly as a result of the immune response to the presence of virus antigens a
Cytolytic Viruses
Causes the most productive infections because they kill the host cell
Cytopathic Effects (CPE)
Changes in cell morphology caused by infecting virus
Virus vs Host
Speed of virus replication and spread vs location/timing/magnitude of the summe response = outcome of viral infection
Acute Infection
high but transient viral replication
Acute Replication
Rapid onset of viral replication
Short but possibly severe course of disease
Production of large numbers of virus particles
Immune Clearance
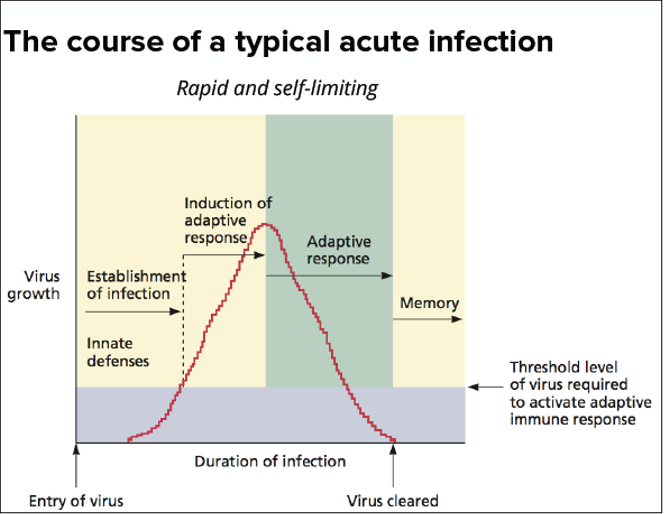
Examples of Acute Infection
Influenza A Virus (IAV)
SARS-CoV2
Polio Virus
Measles Virus (MeV)
Ebola Virus (EBOV)
Variola Virus
Chronic/Persistent Infection
continuous productive infection and/or latency and reactivation
Patterns of Viral Infection and Disease
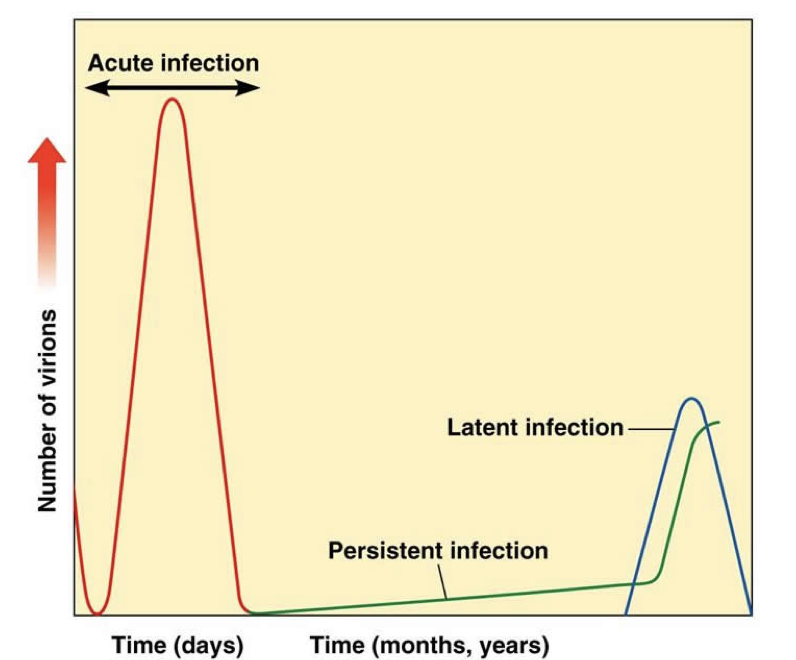
Continuous Reproductive Replication
Continuous regeneration of infectious virus despite ongoing antiviral immunity
Continuous stimulation of immune system due to continuous expression of viral proteins
Generally non-cytopathic viruses: low levels of cell death/tissue damage
Examples of Continuous Reproductive Replication
HIV
HCV
HPV
EBOV
MeV
ZIKV
Latent Infection and Reactivation
Viruses go into transcriptionally and antigenically silent state
Intermittently reactivate to generate new infectious virus
No known immune mechanism to clear latent virus-infected cells
Examples of Latent Infection and Reactivation
HIV
HCMV
Major Viral Strategies for Persistent Infection
Selection of cell subsets
Modulation of viral gene expression
Subversion of cellular apoptotic pathways
Avoidance of the immune system
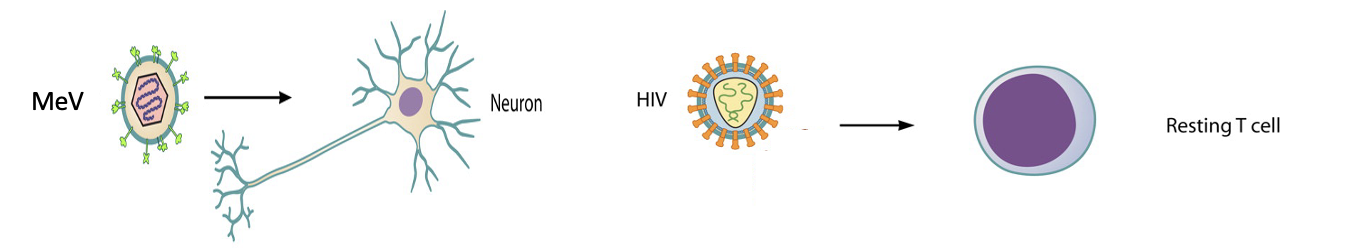
Selection of Cell Subsets
Some cells can serve as sanctuaries for a persistent infection
Ex: neurons (MeV) and memory T-cells (HIV) —> live a long time
Examples of Selection of Cell Subsets
MeV: Established chronic infection in neurons, ling-lived cells in the central nervous system (CNS)
HIV: Established a latent infection in the resting memory CD4+T cells, long-lived cells of the immune system
Modulation of Viral Gene Expression
Ensures survival of the host cell if the virus is cytopathic and to avoid immune recognition
Example of Viral Gene Expression
Chromatin Modification
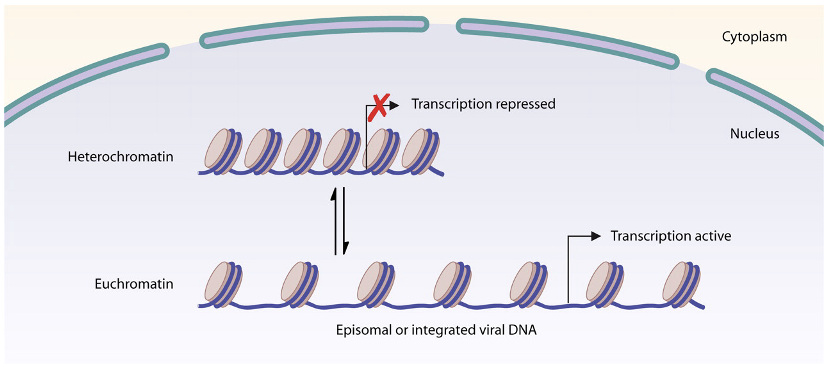
Subversion of cellular apoptotic pathways
During acute infection, a virus needs to prevent apoptosis of infected cells to keep them around longer
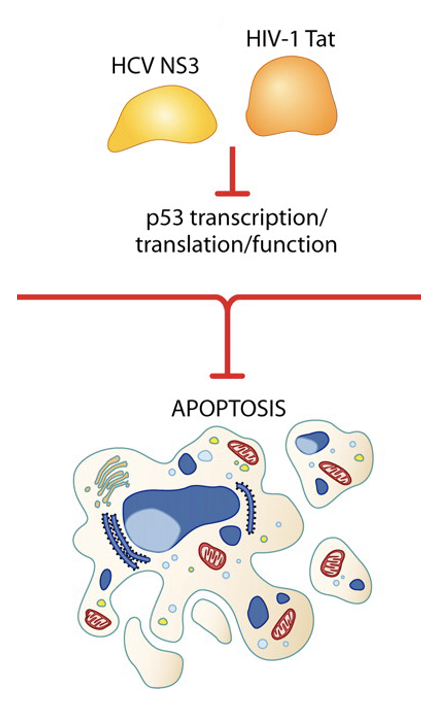
Example of Subversion of Cellular Apoptotic Pathways
Viruses inhibit the activity the cellular pro-apoptotic p53 protein
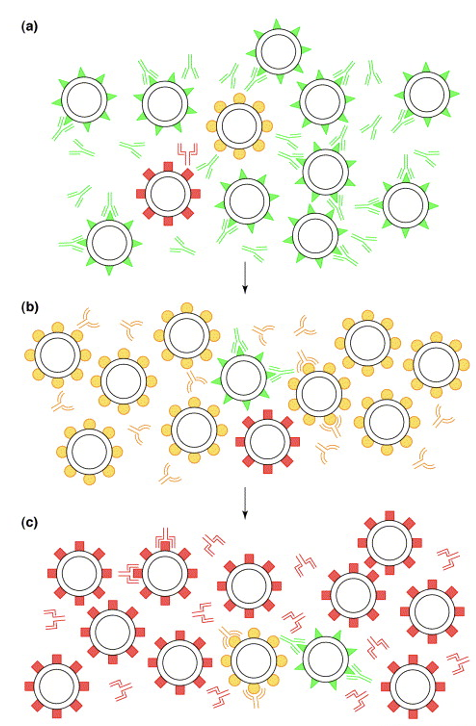
Avoidance of Clearance by the Immune System
RNA viruses exploit genetic variability to avoid recognition by the immune system
High mutation rate in RNA Viruses
Multiple escape mutants enable the virus to evade the immune response
Interferon Response
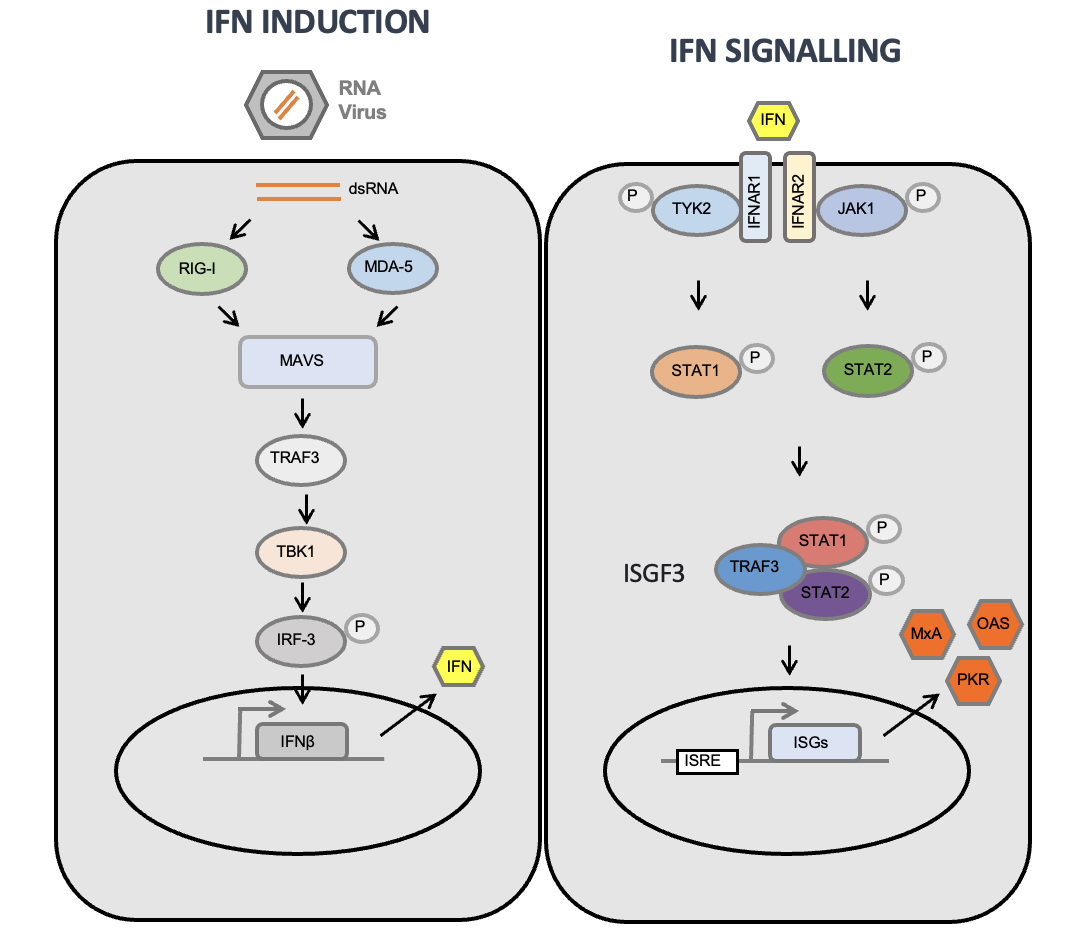
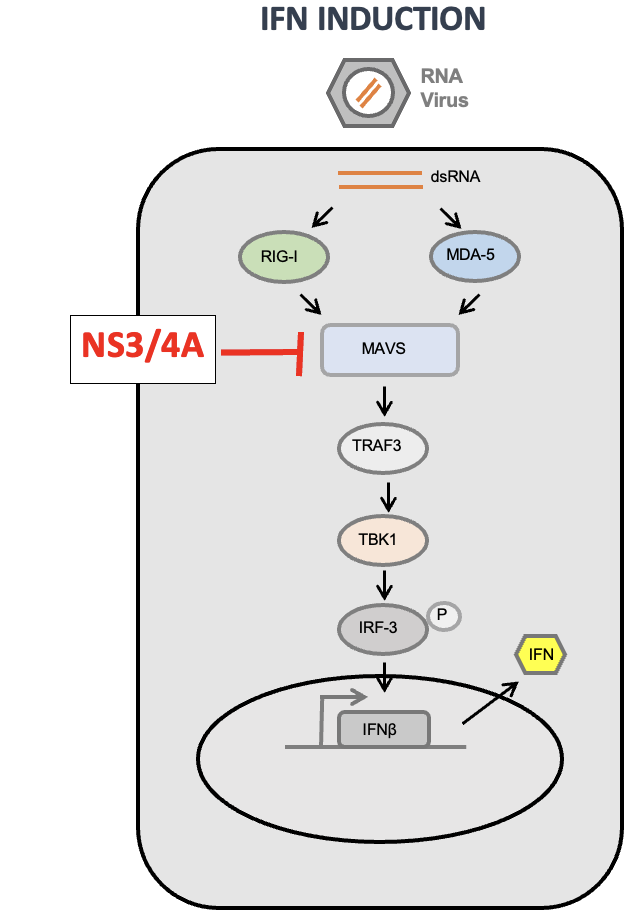
HCV Blocks in IFN Induction Pathway
HCV NS3/4A protein complex acts as a viral interferon antagonist.
NS3/4A proteolytically cleaves MAVS.
Cleavage of MAVS blocks the downstream signaling cascade and stops interferon expression
HCV Blocks IFN Signalling Pathway
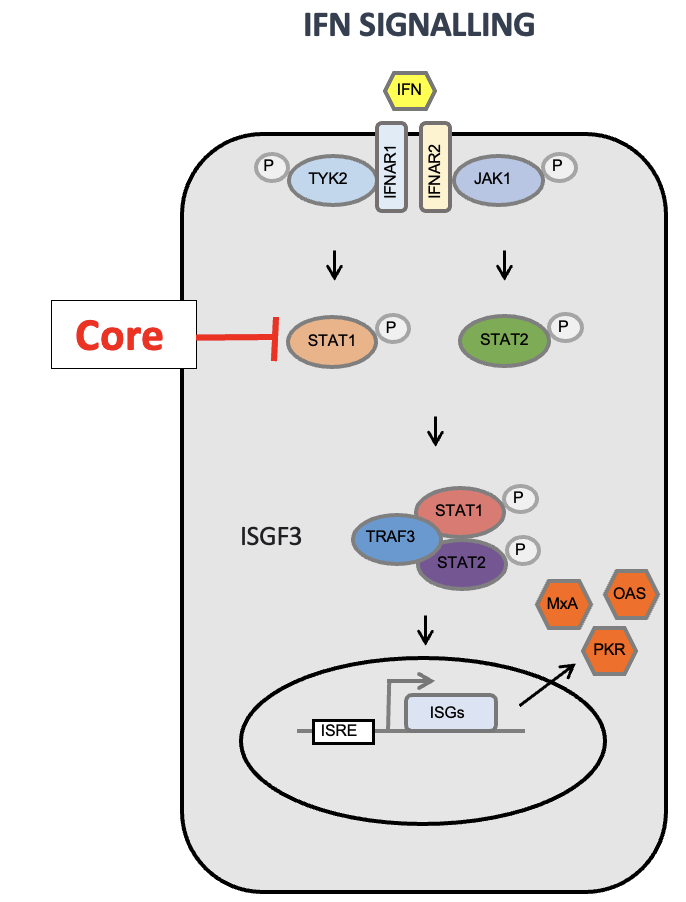
HCV core protein acts as a viral interferon antagonist.
Core inhibits STAT1 phosphorylation and, therefore, its activation.
Core also induces STAT1 degradation.
Inhibition of STAT1 prevents downstream expression of interferon-stimulated genes (ISGs).
HCV Employs Multiple Strategies to Evade the Innate Interferon Response
Disrupts the cellular signaling pathway that leads to interferon expression.
Disrupts the JAK-STAT pathway to limit the expression of interferon stimulated genes (ISGs).
Disrupts the antiviral activities of ISGs
Don’t forget that HCV also employs multiple strategies to also evade adaptive immunity.
HIV and Memory CD4+T Cells
Infection of activated CD4+T Cells results in cell death/apoptosis
DNA copy of the HIV genome incorporated into the nucleus of the host cell
CD4+T Cell can get infected —> allows HIV to persist for decades, integrated into the host cell genome
How does HIV establish latency in resting memory CD4+ T cells?
Accessibility of key host cell transcription factors
In resting memory CD4+ T cells, transcription factors are sequestered in the cytoplasm to suppress transcription.
Both host cell and HIV transcription are suppressed.
Position of the HIV provirus in the host cell genome
HIV preferentially integrates into actively transcribing genes.
Transcription can be suppressed by transcriptional interference via proximal promoters of host cell genes
How does HIV Establish Persistent Infection?
Continuous productive replication and latency
How does HCV establish persistent infection?
Continuous productive replication in hepatocytes