TRIPLE: Esters
1/17
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
18 Terms
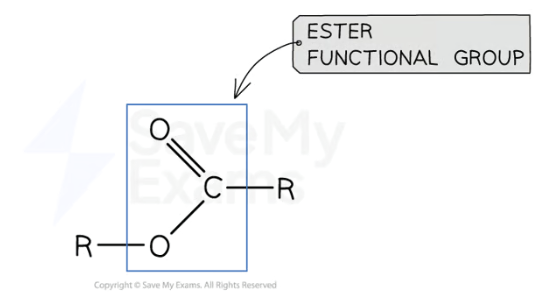
What are esters? Functional group?
Esters are organic compounds (it doesn’t say in the textbook but google says it is a homologous series) formed by the reaction of an alcohol with a carboxylic acid
The functional group sits in the middle so the ‘R’ represents that
—COO—
What can esters be used for? Why? Properties
They can be used for food flavourings and perfumes
They have a distinct and unique smell (in savemyexams: sweet-smelling oily liquids)
Properties
They have distinctive smells
They are volatile (they turn to vapour easily)
How are esters formed? How is this reaction called? RESPOND TO THIS QUESTION
Esters are made by heating an alcohol and a carboxylic acid with a catalyst of concentrated sulfuric acid
Esterification
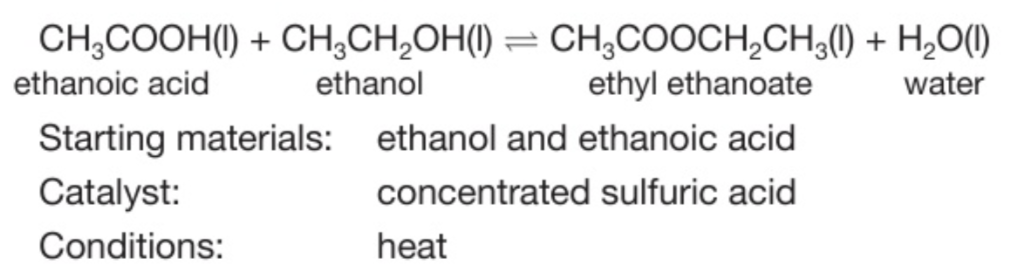
How do you produce the ester ethyl ethanoate? Word and chemical equation?
Indicate the reagents, catalyst and conditions
Heat a mixture of ethanoic acid and ethanol with a few drops of concentrated sulfuric acid.
Why is the concentrated sulfuric acid not written into the equation? Why is heat required?
Because it is a catalyst and it isn’t consumed in the reaction.
To accelerate the reaction
What type of reaction is this (aside from esterification)? Why? Another name for this kind of reaction basically.
Condensation because water is made when two molecules are joined together
This is a reversible reaction so how do we maximise the yield of ethyl ethanoate? Why?
To maximise the yield, we use pure ethanol (called glacial ethanoic acid, it is en cursiva en el textbook) and ethanoic acid
Because if water is added to the reactants the system would want to shift to the left to consume the water and therefore decrease the amount of ethyl ethanoate made
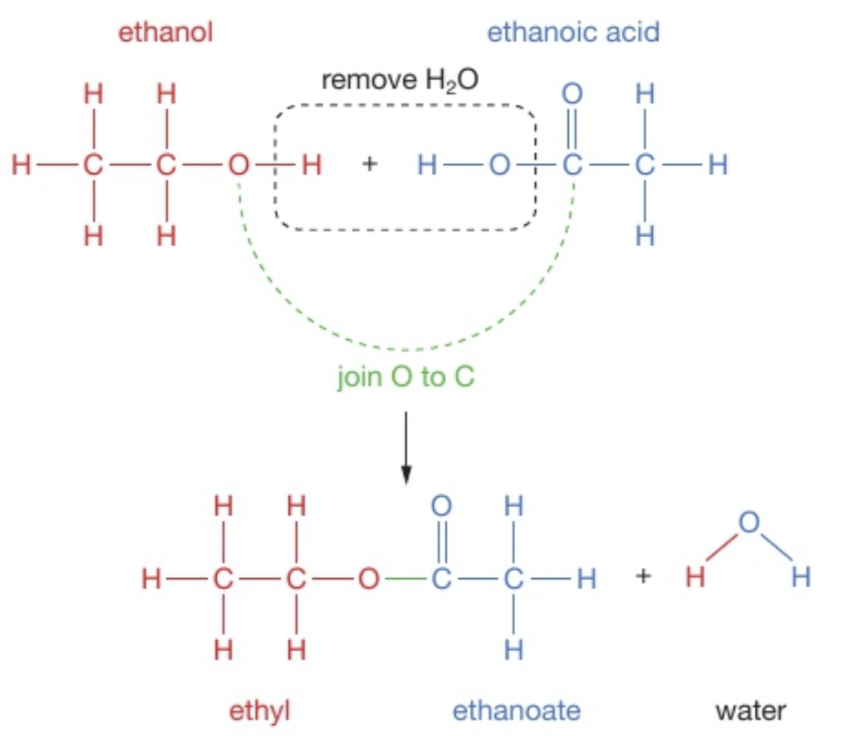
Drawing displayed formula for esters
Draw the displayed equation for ethanol and ethanoic acid
Draw both structures and remove the H2O. It is easier to draw the OH parts next to each other, but idk
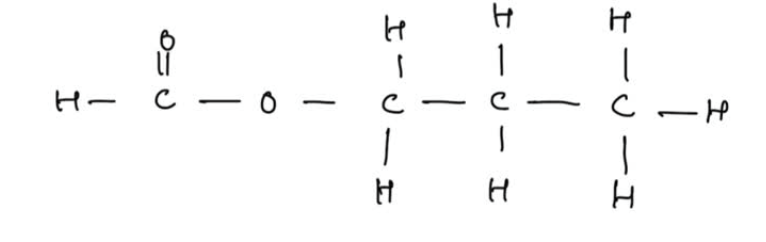
We will also need to be able to work out which carboxylic acid and alcohol reacted together to formed the given ester. How do we do this? What reagents reacted to form this ester?
This is the ester ‘propyl methanoate’ so made from reacting propanol and methanoic acid.
The image tells you how to properly do it
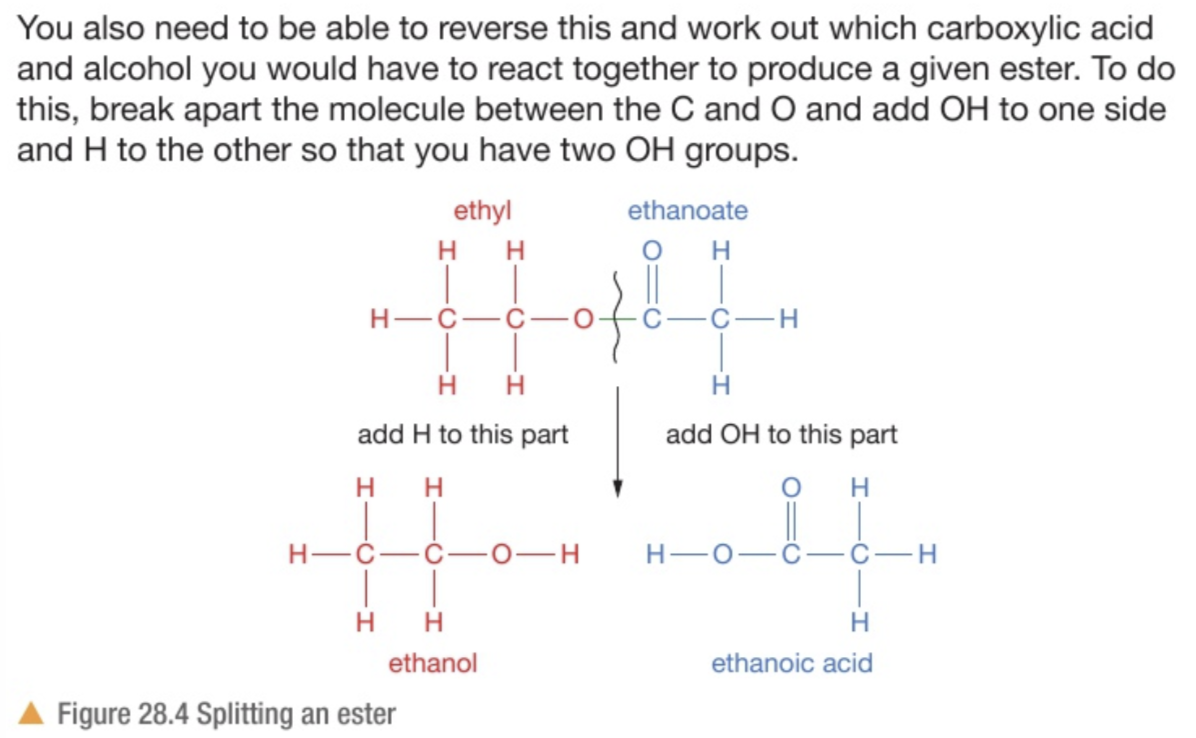
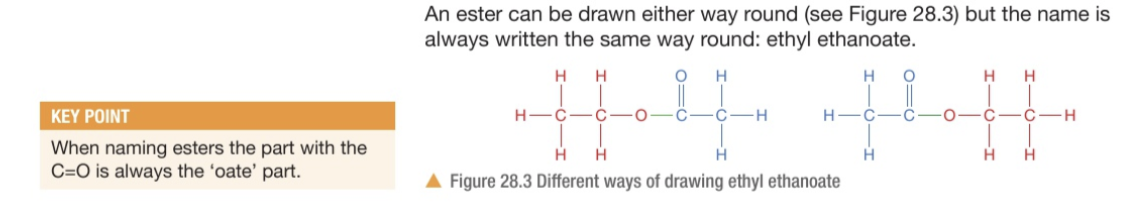
How do we know which part from the displayed formula or the name tells us the alcohol and the carboxylic acid?
The name: ‘-yl’ tells you the alcohol while the ‘-oate’ tells you the carboxylic acid
The displayed formula: the ‘C=O’ is the ‘-oate’ part so it is the carboxylic acid
Practical: making and testing the smell of some esters in the lab
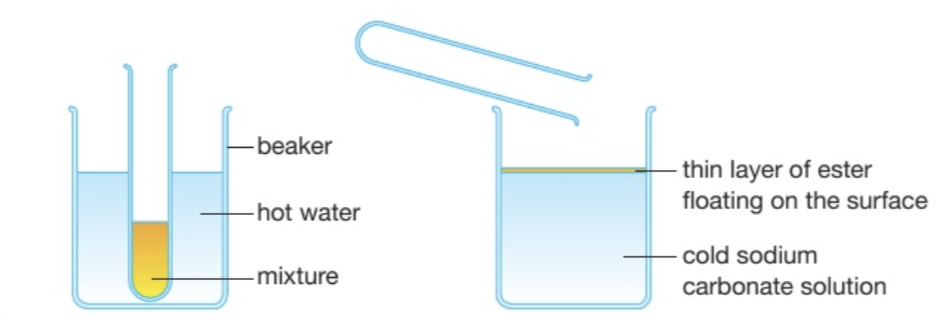
Method to prepare ethyl ethanoate?
Put 1 cm³ of ethanoic acid and 1 cm³ of ethanol into a boiling tube
Mix well and carefully add concentrated sulfuric acid
Place boiling tube into a hot water bath
Allow the contents of the tube to cool
Pour the mixture into a beaker half-full of sodium carbonate solution
Waft the odour towards your nose with your hand
The mixture has to be heated. Why in a water bath and not by a direct Bunsen flame?
Why are the contents added to sodium carbonate solution? How does it do this? What will you see?
Contents of the tubes are flammable
To be able to smell the ester better.
Not much ester is likely going to be formed in such a short time and the smell will be masked by the smell of the carboxylic acid
So we add NaCO3 to react it with excess acid.
We will therefore see some effervescence (carbon dioxide). It should be added until the mixture stops fizzing
Savemyexams: what should you add to remove ethanol?
Calcium chloride solution
At the end, there are two diff layers. What is the top one? What about the bottom one? Why?
A layer of ester will separate and float on top. The bottom layer is a solution of sodium carbonate.
This is because the acid and alcohol will mostly dissolve in the water, but the ester won’t
The small amount we produce at the lab will float on the water, which makes them easy to smell
What does butyl butanoate and propyl ethanoate smell like?
Butyl butanoate: pineapple
Propyl ethanoate: pear
