HW 11-14 Conceptual Biochem
1/227
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
228 Terms
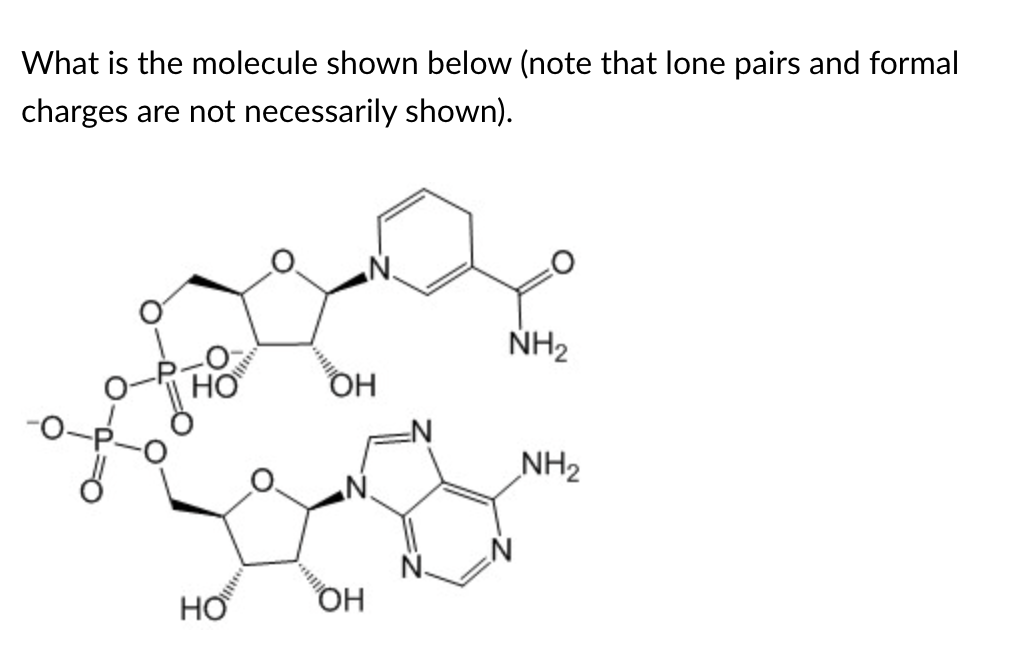
NADH
Anabolic reactions generally involve the synthesis of complex biomolecules from relatively simple precursor molecules.
True
Several copies of __________________ are added to a protein as a “tag” to mark the protein for degradation by the proteasome.
ubiquitin
The molecule that is the “transamination cousin” of alpha-ketoglutarate is ____________________. (Note that any choices that are amino acids are assumed to be L-amino acids.)
glutamate
The biochemical standard free energy change (ΔG°′) for the complete oxidation of glucose is −686 kcal/mol. What is the value of the corresponding equilibrium constant for this process? Assume a temperature of 37 °C.
1.0 × 10484
The free energy (ΔG°′) of hydrolysis of ATP to ADP, Pi, and H+ is −7.3 kcal/mol. The free energy (ΔG°′) of hydrolysis of glucose 1-phosphate to glucose and Pi is −5.0 kcal/mol. Given this information, which of the two species (ATP or glucose 1-phosphate) has the higher (or greater) phosphoryl group transfer potential?
ATP
This vitamin is required by the enzyme that catalyzes γ-carboxylation reactions of glutamate residues in the Gla domain of prothrombin.
Vitamin K (phylloquinone)
Birds of the Order Galliformes (i.e., fowl, pheasant, grouse, quail, & turkey) tend to have relatively light-colored breast meat, whereas birds of the Order Anseriformes (i.e., ducks and geese) tend to have relatively dark-colored breast meat. You will learn why during the course of the semester. Let us say you are hunting for pheasant and you flush one. How long (in seconds to the nearest ones) can it fly given that its ATP production is limited by the rate at which glycogen can be broken down to glucose-1-phosphate, which is 120 micromoles per minute per gram of flight muscle, which contains about 0.34% glycogen by weight. Assume that a glucose residue in glycogen has a MW of 162.
10
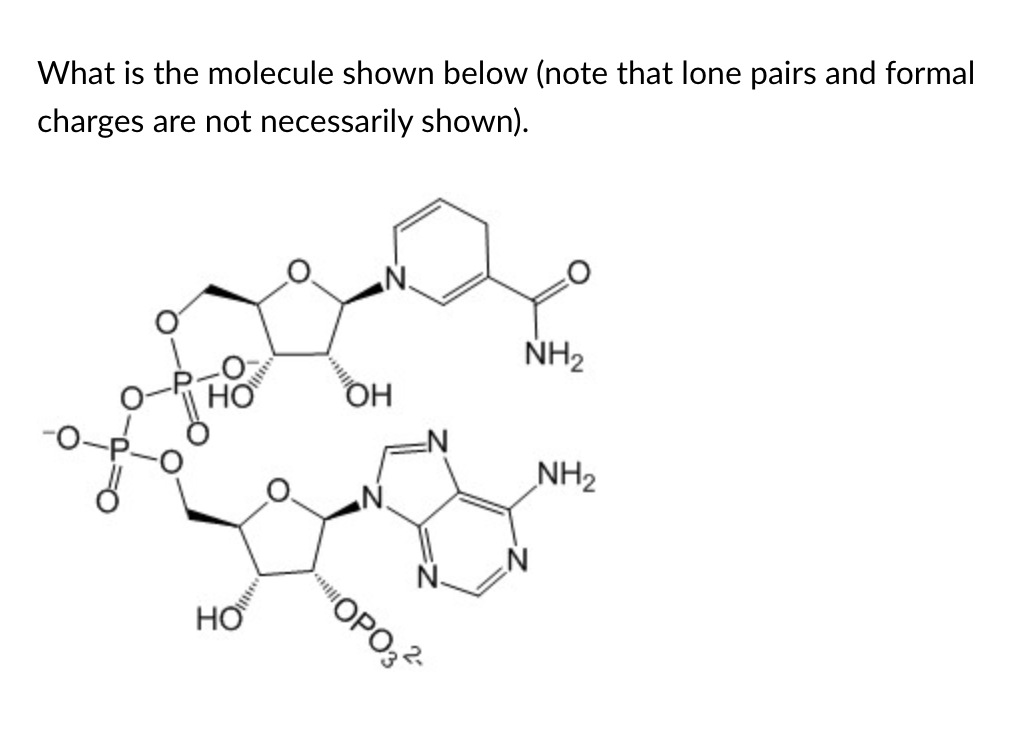
NADPH
Catabolic reactions generally involve the oxidation of fuel molecules.
True
If the amino acid L-aspartate entered the active site of an aminotransferase, it would depart as ___________________.
oxaloacetate
The active form of this vitamin helps promote the absorption of dietary calcium (as Ca2+ ions) across the intestinal lining.
Vitamin D
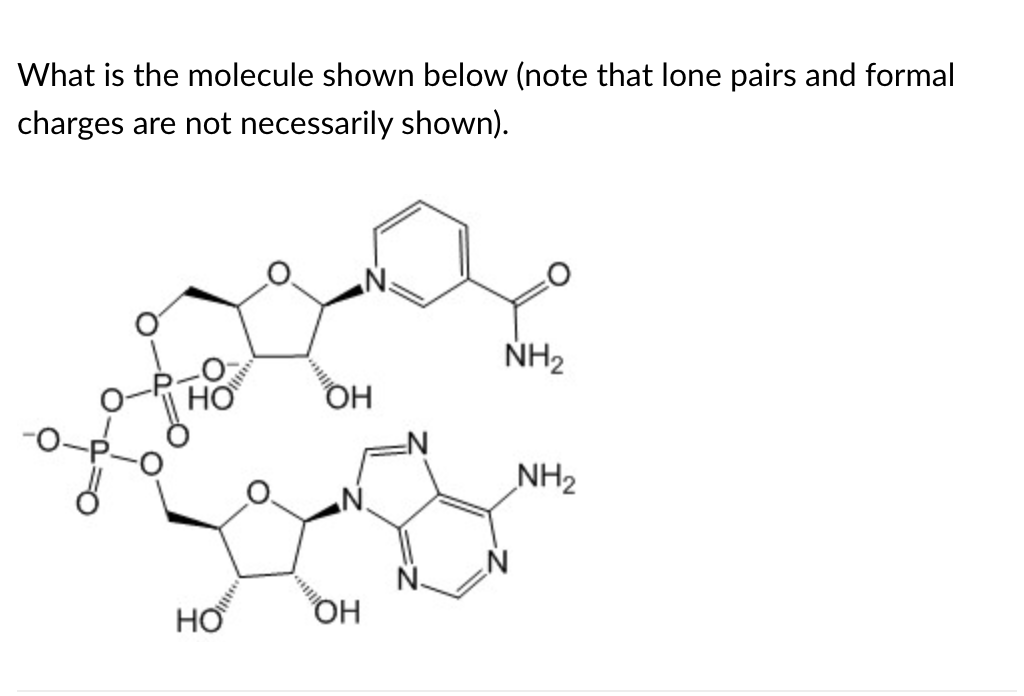
NAD+
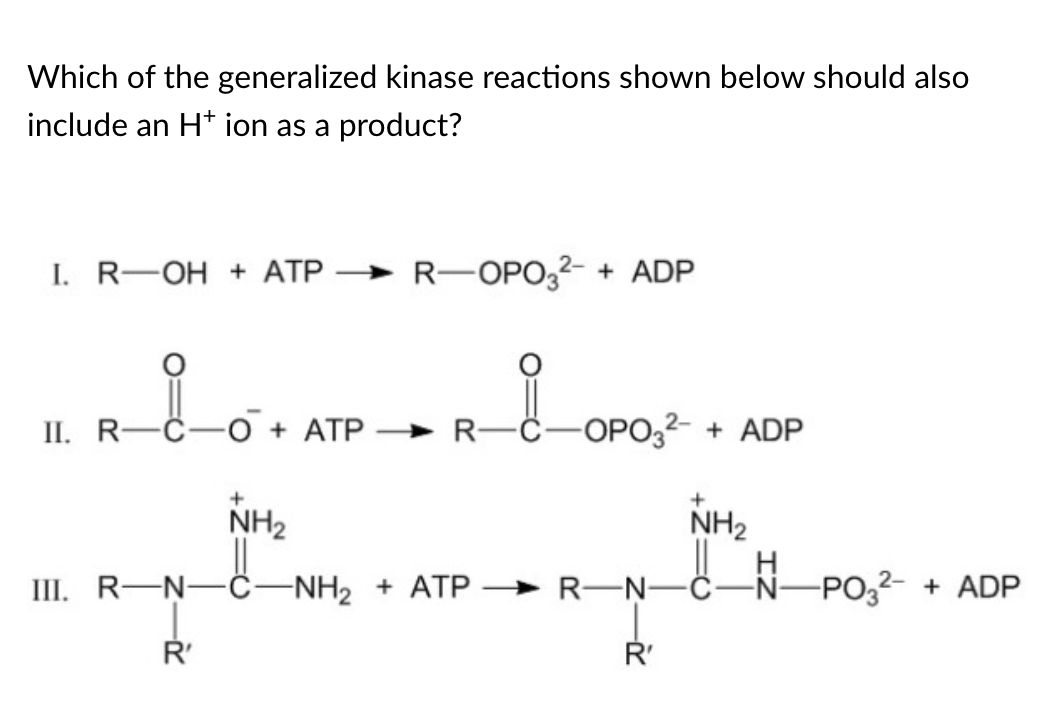
I and III
If the amino acid L-glutamate entered the active site of an aminotransferase, it would depart as ___________________.
alpha-keto-glutarate
This vitamin forms part of a dinucleotide cofactor that is involved in many catabolic redox reactions (the unphosphorylated form of the cofactor), and in many anabolic redox reactions (the phosphorylated form of the cofactor). These redox reactions often involve the oxidation of an alcohol to a carbonyl (or vice versa).
Vitamin B3 (niacin or nicotinic acid)
What is the advantage of breaking down glycogen via phosphorolysis rather than hydrolysis?
The glucose 1-phosphate thus liberated can be converted to glucose 6-phosphate without the need to “spend” and ATP
The molecule that is the “transamination cousin” of pyruvate is ____________________. (Note that any choices that are amino acids are assumed to be L-amino acids.)
alanine
This vitamin comprises several related compounds, one of which is the most abundant form, and people who take this vitamin do so because it is considered to be a general antioxidant.
Vitamin E (α-tocopherol)
Catabolic reactions generally involve the reduction of fuel molecules.
False
Ubiquitin is linked to the protein that it marks for degradation through _____ linkage to a _______ residue on the protein.
amide, lysine
This vitamin helps maintain the proper redox state of the enzyme that catalyzes the hydroxylation reaction of proline residues in collagen.
Vitamin C
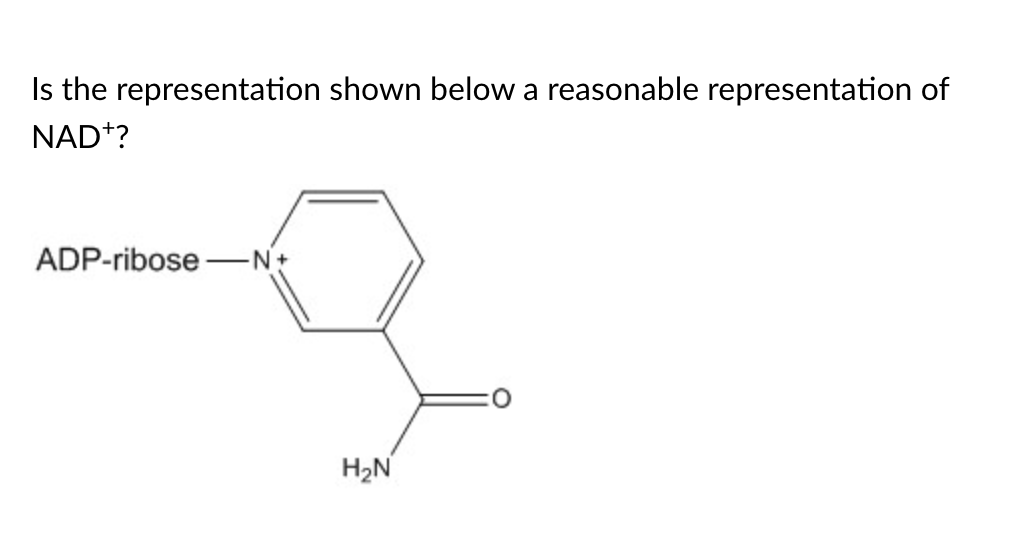
yes
If the amino acid L-alanine entered the active site of an aminotransferase, it would depart as ___________________.
pyruvate
The active form of this vitamin helps promote the absorption of dietary calcium (as Ca2+ ions) across the intestinal lining.
Vitamin D
Which of the following are major 2- and 3-carbon participants/precursors in several metabolic pathways? Select all that apply.
the acetyl group of acetyl-CoA
glyceraldehyde 3-phospate
pyruvate
The molecule that is the “transamination cousin” of oxaloacetate is ____________________. (Note that any choices that are amino acids are assumed to be L-amino acids.)
aspartate
The active form of this vitamin helps promote the absorption of dietary calcium (as Ca2+ ions) across the intestinal lining.
Vitamin D
__________________ is a prominent type of reaction that helps to break down many different types of food molecules.
Hydrolysis
This vitamin is derived from β-carotene, and, once it is oxidized to its “working” form, is involved in the signal transduction process that is vision.
Vitamin A (retinol)
Match the vitamin below to its biological role.
Vitamin A
Vitamin B3 (niacin or nicotinic acid)
Vitamin C
Vitamin D
Vitamin K
Vitamin E
Vitamin A - required for steps in vision
Vitamin B3 (niacin or nicotinic acid) - required as an essential part of a cofactor that is involved in many redox reactions in the cell
Vitamin C - required for post-translational reactions that occur with the protein collagen
Vitamin D - required for absorption of dietary calcium ions
Vitamin K - required for reactions that occur with the protein prothrombin
Vitamin E - required as a general antioxidant
How many NADHs are required for the first part of glycolysis (i.e., the “pump-priming” phase), and how many NADHs are produced in the second part of glycolysis (i.e., the energy yielding phase)?
0; 2
What is the name of the glycolytic enzyme that catalyzes the formation of a molecule whose predominant form is the keto form? In fact, the enol-to-keto tautomerization of this molecule helps to “drive” this reaction. Hint: this enzyme catalyzes one of the “irreversible” reactions of this pathway.
pyruvate kinase
If a sample of glucose labeled with 14C at carbon-1 (C1) and carbon-6 (C6) was used as the sole glucose source for glycolysis, at what carbon (carboxylate, ketone, or methyl) of the resulting pyruvate would the label appear?
the methyl carbon of all of the resulting pyruvate molecules
Fructose is converted to fructose 6-phosphate in muscle cells by ______________________, and it is converted to fructose 1-phosphate in liver cells by ________________________.
hexokinase; fructokinase
What is the name of the pentose phosphate pathway (PPP) enzyme that catalyzes the hydrolysis of a bisphosphorylated intermediate to a monophosphorylated compound and inorganic phosphate? Hint: this enzyme also functions in gluconeogenesis.
fructose 1,6-bisphosphatase
In the glycogen synthase reaction, the UDP moiety departs, which leaves a planar oxonium ion intermediate, which in turn undergoes nucleophilic attack from below by the 4′-hydroxyl at a non-reducing end of glycogen.
True
If the phosphorus in 3PG is labeled as 32P, will the first 2PG that is formed as a result of the phosphoglycerate mutase reaction contain 32P?
No, since no radiolabel (32P) will be transferred to the initial 2PG that is formed because the phosphoryl group on C3 of 3PG is not directly transferred to the hydroxyl on C2.
What is the net oxidation state of the carbons in glyceraldehyde 3-phosphate, and what is the net oxidation state of the carbons in 1,3-bisphosphoglycerate? By net oxidation state is meant the sum of the oxidation states of the carbons that make up the molecules.
The net oxidation state for the carbons in glyceraldehyde 3-phosphate is 0, and the net oxidation state for the carbons in 1,3-bisphosphoglycerate is +2.
How many ATPs are required for the first part of glycolysis (i.e., the “pump priming” phase), and how many ATPs are produced in the second part of glycolysis (i.e., the energy yielding phase)?
2; 4
What is the name of the glycolytic enzyme that catalyzes the ATP-dependent phosphorylation of a six-carbon monosaccharide to make a six-carbon monophosphorylated compound?
hexokinase
If a sample of glucose labeled with 14C at carbon-6 (C6) was used as the sole glucose source for glycolysis, at what carbon: aldehyde (-CHO), hydroxymethylene (>CHOH), or phosphomethyl (-CH2OPO32−) of the intermediate glyceraldehyde 3-phosphate would the label appear?
the phosphomethyl carbon of half the glyceraldehyde 3-phosphate molecules
What “feeder” pathway involves an epimerization reaction in which NAD+ is temporarily reduced to NADH and then re-oxidized back to NAD+ as one monosaccharide moiety is epimerized to another monosaccharide moiety?
The galactose feeder pathway
During the pentose phosphate pathway, fructose 1,6-bisphosphate is converted into fructose 6-phosphate and inorganic phosphate. What is the name of the enzyme that catalyzes this transformation? Hint: this enzyme also functions in gluconeogenesis in one of the “bypass” reactions.
fructose 1,6-bisphosphatase
In glycogen synthesis, glucose 1-phosphate reacts with uridine triphosphate (UTP) to make UDP-glucose and inorganic pyrophosphate (PPi). What is the fate of the PPi?
It is hydrolyzed to two molecules of inorganic phosphate (Pi) in a reaction catalyzed by the enzyme inorganic pyrophosphate.
What are the net yields of ATP, NADH, and H+
that are produced through glycolysis? Assume we start with glucose and end with the formation of two pyruvates.
2 ATPs; 2 NADHs; 2 H+
What is the name of the glycolytic/gluconeogenic enzyme that catalyzes the interconversion of 2-phosphoglycerate (2PG) and phosphoenolpyruvate (PEP)? (Note that water is liberated from 2PG as it is converted to PEP, and water is added to PEP as it is converted to 2PG.)
enolase
If a sample of glucose labeled with 14C at carbon-1 (C1) was used as the sole source of glucose for glycolysis, at what carbon (carboxylate, ketone, or methyl) of the resulting pyruvate would the label appear?
the methyl carbon of half of the resulting pyruvate molecules
What is the name of the pentose phosphate pathway (PPP) enzyme that catalyzes the oxidative decarboxylation of a six-carbon phosphorylated compound to a five-carbon phosphorylated ketose and carbon dioxide with the concomitant reduction of dinucleotide cofactor?
6-phosphogluconate dehydrogenase
In the glycogen synthase reaction, the UDP moiety departs, which leaves a planar oxonium ion intermediate, which in turn undergoes nucleophilic attack from below by the 4′-hydroxyl at a non-reducing end of glycogen.
True
How many FADH2s are required for the first part of glycolysis (i.e., the “pump-priming” phase), and how many FADH2s are produced in the second part of glycolysis (i.e., the energy yielding phase)?
0; 0
What is the name of the glycolytic enzyme that catalyzes the interconversion of a six-carbon phosphorylated aldose and a six-carbon phosphorylated ketose in glycolysis and gluconeogenesis?
phosphohexose isomerase (aka phosphoglucose isomerase)
If a sample of glucose labeled with 14C at carbon-2 (C2) and carbon-5 (C5) was used as the sole glucose source for glycolysis, at what carbon: aldehyde (-CHO), hydroxymethylene (>CHOH), or methyl-phosphate (-CH2OPO32−) of the intermediate glyceraldehyde 3-phosphate would the label appear?
the hydroxymethylene carbon of all the glyceraldehyde 3-phosphate molecules
The feeder pathway for the metabolism of fructose in the liver bypasses an important regulatory enzyme of glycolysis. Which of the following is the important regulatory enzyme of glycolysis that is bypassed?
phosphofructokinase-1 (PFK1)
What is the name of the pentose phosphate pathway (PPP) enzyme that catalyzes the transfer of a dihydroxyacetone moiety from a ketose donor to an aldose acceptor?
transaldolase
In glycogen synthesis, glucose 1-phosphate reacts with uridine triphosphate (UTP) to make UDP-glucose and inorganic pyrophosphate (PPi).
True
If a sample of glucose labeled with 14C at carbon-2 (C2) and carbon-5 (C5) was used as the sole glucose source for glycolysis, at what carbon (carboxylate, ketone, or methyl) of the resulting pyruvate would the label appear?
the ketone carbon of all of the resulting pyruvate molecules
What is the name of the enzyme that catalyzes the ATP-dependent phosphorylation of galactose to make galactose 1-phosphate as part of galactose metabolism (i.e., the galactose feeder pathway)?
None of the other choices is correct
What is the name of the pentose phosphate pathway (PPP) enzyme that catalyzes the interconversion of the pair: xylulose 5-phosphate and erythrose 4-phosphate with the pair: glyceraldehyde 3-phosphate and fructose 6-phosphate?
transketolase
Imagine that a liver cell has “run” gluconeogenesis to the point where glucose 6-phosphate has been produced. Instead of “running” the glucose 6-phosphatase reaction and exporting free glucose to the bloodstream, imagine that the glucose 6-phosphate is going to be used for glycogen synthesis. Before the glucose 6-phosphate can be used as a substrate for glycogen synthesis, it must first be converted to glucose 1-phosphate. What is the name of the enzyme that catalyzes this conversion?
Phosphoglucomutase
How many ATPs are required for the first part of glycolysis (i.e., the “pump priming” phase), and how many ATPs are produced in the second part of glycolysis (i.e., the energy yielding phase)?
2; 4
If a sample of glucose labeled with 14C at carbon-4 (C4) was used as the sole glucose source for glycolysis, at what carbon (carboxylate, ketone, or methyl) of the resulting pyruvate would the label appear?
the carboxylate carbon of half of the resulting pyruvate molecules
The feeder pathway for the metabolism of fructose in the liver bypasses an important regulatory enzyme of glycolysis. Which of the following is the important regulatory enzyme of glycolysis that is bypassed?
phosphofructokinase-1 (PFK1)
What is the name of the pentose phosphate pathway (PPP) enzyme that catalyzes the interconversion of a three-carbon phosphorylated aldose and a three-carbon phosphorylated ketose? Hint: this enzyme also functions in glycolysis and gluconeogenesis.
triose phosphate isomerase
In glycogen synthesis, ____________________ reacts with uridine triphosphate (UTP) to make UDP-glucose.
glucose 1-phosphate
What is the name of the glycolytic enzyme that catalyzes the interconversion of glucose 6-phosphate and fructose 6-phosphate in glycolysis and gluconeogenesis?
phosphohexose isomerase (aka phosphoglucose isomerase)
If a sample of glucose labeled with 14C at carbon-3 (C3) was used as the sole glucose source for glycolysis, at what carbon: aldehyde (-CHO), hydroxymethylene (>CHOH), or phosphomethyl (-CH2OPO32−) of the intermediate glyceraldehyde 3-phosphate would the label appear?
the aldehyde carbon of half the glyceraldehyde 3-phosphate molecules
What is the name of the pentose phosphate pathway (PPP) enzyme that catalyzes the ATP-dependent phosphorylation of a six-carbon aldose to make a six-carbon phosphorylated aldose? Hint: this enzyme also functions in glycolysis.
hexokinase
In glycogen synthesis, glucose 6-phosphate reacts with uridine triphosphate (UTP) to make UDP-glucose and inorganic pyrophosphate (PPi).
False
What is the net yield of ATPs that are produced in glycolysis?
2
What is the name of the glycolytic/gluconeogenic enzyme that catalyzes a retro-aldol addition reaction in glycolysis and an aldol addition reaction in gluconeogenesis?
aldolase
If a sample of glucose labeled with 14C at carbon-1 (C1) and carbon-6 (C6) was used as the sole glucose source for glycolysis, at what carbon (carboxylate, ketone, or methyl) of the resulting pyruvate would the label appear?
the methyl carbon of all of the resulting pyruvate molecules
In glycogen synthesis, glucose 1-phosphate reacts with uridine triphosphate (UTP) to make UDP-glucose and inorganic pyrophosphate (PPi). What is the fate of the PPi?
It is hydrolyzed to two molecules of inorganic phosphate (Pi) in a reaction catalyzed by the enzyme inorganic pyrophosphate.
What is the name of the glycolytic/gluconeogenic enzyme that catalyzes the interconversion of a three-carbon compound that is phosphorylated at the 2-position and a three-carbon compound that is phosphorylated at the 3-position?
phosphoglycerate mutase
What is the name of the glycolytic/gluconeogenic enzyme that catalyzes the interconversion of 2-phosphoglycerate (2PG) and 3-phosphoglycerate (3PG)?
phosphoglycerate mutase
If a sample of glucose labeled with 14C at carbon-2 (C2) was used as the sole glucose source for glycolysis, at what carbon (carboxylate, ketone, or methyl) of the resulting pyruvate would the label appear?
the ketone carbon of half of the resulting pyruvate molecules
What is the name of the pentose phosphate pathway (PPP) enzyme that catalyzes the hydrolysis of a cyclic ester?
lactonase
What is the name of the glycolytic/gluconeogenic enzyme that catalyzes the interconversion of glyceraldehyde 3-phosphate (GAP) and dihydroxyacetone phosphate (DHAP)?
triose phosphate isomerase
What are the net yields of ATP, NADH, and H+
that are produced through glycolysis? Assume we start with glucose and end with the formation of two pyruvates.
2 ATPs; 2 NADHs; 2 H+
If a sample of glucose labeled with 14C at carbon-3 (C3) and carbon-4 (C4) was used as the sole glucose source for glycolysis, at what carbon: aldehyde (-CHO), hydroxymethylene (>CHOH), or methyl-phosphate (-CH2OPO32−) of the intermediate glyceraldehyde 3-phosphate would the label appear?
the aldehyde carbon of all the glyceraldehyde 3-phosphate molecules
In the “feeder” pathway that allows galactose to enter glycolysis, an epimerization reaction occurs in which NAD+/NADH serves as a true cofactor (as opposed to a REDOX cofactor). By “true cofactor” is meant that this cofactor is used catalytically and thus does not undergo a net change during the overall reaction.
True
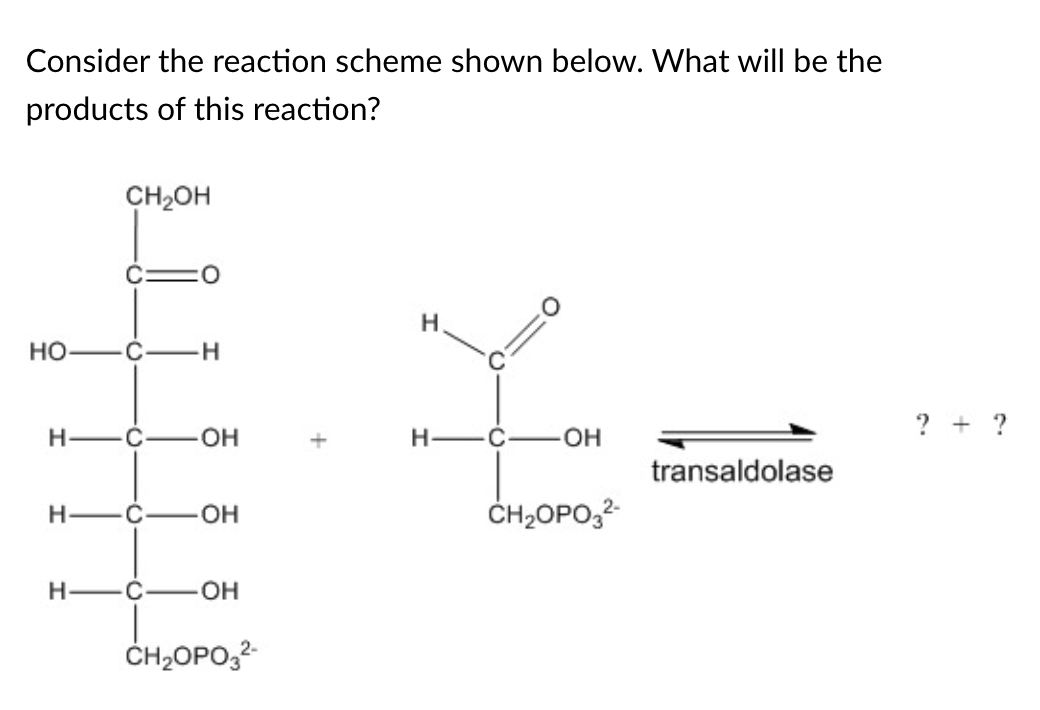
Erythrose 4-phosphate and fructose 6-phosphate
If a sample of glucose labeled with 14C at carbon-4 (C4) was used as the sole glucose source for glycolysis, at what carbon: aldehyde (-CHO), hydroxymethylene (>CHOH), or phosphomethyl (-CH2OPO32−) of the intermediate glyceraldehyde 3-phosphate would the label appear?
the aldehyde carbon of half the glyceraldehyde 3-phosphate molecules
What is the name of the pentose phosphate pathway (PPP) enzyme that catalyzes the transfer of an aldehyde moiety from a ketose donor to an aldose acceptor?
transketolase
In glycogen synthesis, glucose 1-phosphate reacts with uridine triphosphate (UTP) to make UDP-glucose and inorganic pyrophosphate (PPi).
True
If the inorganic phosphate used in the glyceraldehyde 3-phosphate dehydrogenase reaction had all four oxygens labeled with 18O, how many labeled oxygen atoms would appear in the ATP that is formed in the subsequent (i.e., phosphoglycerate kinase) glycolytic reaction?
3
As part of the galactose feeder pathway, the enzyme galactose-1-phosphate uridylyl transferase catalyzes _______________________________.
the formation of glucose 1-phosphate and UDP-galactose from galactose 1-phosphate and UDP-glucose
If a sample of glucose labeled with 14C at carbon-1 (C1) was used as the sole source of glucose for glycolysis, at what carbon (carboxylate, ketone, or methyl) of the resulting pyruvate would the label appear?
the methyl carbon of half of the resulting pyruvate molecules
If mannose is to enter glycolysis, it must undergo an enzyme-catalyzed reaction that uses a UDP-sugar (uridine diphosphate-sugar) cofactor.
False
What is the name of the pentose phosphate pathway (PPP) enzyme that catalyzes the interconversion of the pair: glyceraldehyde 3-phosphate and sedoheptulose 7-phosphate with the pair: fructose 6-phosphate and erythrose 4-phosphate?
transaldolase
If a sample of glucose labeled with 14C at carbon-2 (C2) was used as the sole glucose source for glycolysis, at what carbon: aldehyde (-CHO), hydroxymethylene (>CHOH), or phosphomethyl (-CH2OPO32−) of the intermediate glyceraldehyde 3-phosphate would the label appear?
the hydroxymethylene carbon of half the glyceraldehyde 3-phosphate molecules
What is the name of the glycolytic/gluconeogenic enzyme that catalyzes a REDOX (oxidation reduction) reaction in these two pathways?
glyceraldehyde 3-phosphate dehydrogenase
If a sample of glucose labeled with 14C at carbon-5 (C5) was used as the sole glucose source for glycolysis, at what carbon (carboxylate, ketone, or methyl) of the resulting pyruvate would the label appear?
the ketone carbon of half of the resulting pyruvate molecules
What is the name of the pentose phosphate pathway (PPP) enzyme that catalyzes the interconversion of the pair: ribose 5-phosphate and xylulose 5-phosphate with the pair: glyceraldehyde 3-phosphate and sedoheptulose 7-phosphate?
transketolase
If a sample of glucose labeled with 14C at carbon-1 (C1) and carbon-6 (C6) was used as the sole glucose source for glycolysis, at what carbon: aldehyde (-CHO), hydroxymethylene (>CHOH), or methyl-phosphate (-CH2OPO32−) of the intermediate glyceraldehyde 3-phosphate would the label appear?
the methyl-phosphate carbon of all the glyceraldehyde 3-phosphate molecules
What is the name of the glycolytic/gluconeogenic enzyme that catalyzes the formation of ATP and a monphosphorylated three-carbon compound from ADP and a bisphosphorylated three-carbon compound? The reverse happens in gluconeogenesis.
phosphoglycerate kinase
If a sample of glucose labeled with 14C at carbon-1 (C1) was used as the sole glucose source for glycolysis, at what carbon: aldehyde (-CHO), hydroxymethylene (>CHOH), or phosphomethyl (-CH2OPO32−) of the intermediate glyceraldehyde 3-phosphate would the label appear?
the phosphomethyl carbon of half the glyceraldehyde 3-phosphate molecules
Mannose is phosphorylated by hexokinase (in an ATP-dependent manner) prior to its entry into glycolysis.
True