PHAN SPECTROPHOTOMETRY
1/74
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
75 Terms
Spectroscopy
Interaction of Radiation with a sample
Spectroscopy
The study of molecular or atomic structure of a substance by observation of
its interaction with electromagnetic radiation
Spectroscopy
measurement of spectra
QUANTITATIVELY
Spectroscopy For determining the amount of material in a sample
QUALITATIVELY
Spectroscopy For identifying the chemical structure of a sample
Spectrometry
Radiation reacts with matter àreflected, scattered, absorbed, fluoresce and cause photochemical reaction like bond breaking.
Spectrometry
Position (wavelength) in the spectrum where interaction takes place
Measurement of these interactions
Light
is electromagnetic radiation within a certain portion of the electromagnetic spectrum.
Light
The word usually refers to visible light, which is visible to the humaneye and is responsible for the sense of sight.
wavelength (UNITS: micrometer, nanometer, angstrom)
The length of a complete wave or cycle from peak of wave to the peak of the next
Wavenumber
number of waves per centimeter, equivalent to 1/wavelength(cm)
number of waves per centimeter, equivalent to 1/wavelength(cm)
Wavenumber is the
FREQUENCY
complete cycle which pass a given point per second (units: cps/ hertz)
cps/ hertz (1 hertz = 1cps)
units of FREQUENCY
Radiation Energy
The strength of the radiation energy will interact with the molecules in different ways
X-Ray, γ Rays,…
High energy sources produce breaking of bonds
UV / VISIBLE Spectroscopy
Medium energy sources excite electrons
Infrared Energy
Low energy sources produce vibrations in chemical bonds
Microwaves and Radio waves
Very low energy sources produce rotation of the chemical bonds
optical methods
are physicalchemical methods of analysis that are based on the optical range of the scale of electromagnetic waves
220 – 380 nm
range of wavelength of UV
380 – 780 nm
range of wavelength of Visible
780 – 3000 nm
range of wavelength of Near IR
3.0 – 15 um
range of wavelength of Medium
15 – 300 um
range of wavelength of Far IR
cm, µm (10-4 cm) , nm (10-7 cm), Å (10-8cm)
The ranges of the wavelength of radiant energy are measured in
Radio waves
Microwaves
Infrared rays
Visible light
Ultraviolet rays
X-rays
Gamma rays
Types of Radiation from Low to High frequency
Spectrophotometry
is a branch of spectrometry which embraces the measurement of the absorption by chemical species of radiant energy of definite and narrow wavelength approximating monochromatic radiation.
Colorimetry
is branch of spectrophotometry in which absorption takes place in the visible region
VISIBLE SPECTROSCOPY
Absorbance and reflectance
When a sample only absorbs light of a single wavelength the eye sees COMPLEMENTARY colours.
WHITE
Visible Spectroscopy
If the material completely REFLECTS all light it appears
GREY
Visible Spectroscopy
If the material absorbs a constant fraction of the light across the spectrum it appears
BLACK
Visible Spectroscopy
If the material completely ABSORBS all the light it appears
What is the fundamental principle regarding how substances interact with radiant energy?
Selective Absorption. Every substance absorbs or transmits certain wavelengths but not others, depending on its molecular structure.
Chromophore
functional group which absorbs radiant energy in the UV or Vis regions
Ethylene
Ketones
Aldehydes
Acetylene
Organic acids
Azomethines
Example of chromophore
concentration
Intensity of the transmitted radiant energy is a function of the
transverse wave
Light transmitted is in the form of ___ at an enormous velocity
wave
Energy maybe transmitted as
Amplitude
is the distance of half height of the wave
Wavenumber (ṽ)
is the number of waves per centimeter and is equal to 1/wavelength (cm).
photon
Energy maybe absorbed as
photons or quanta
Radiant energy absorbed is in the form of discrete parcels of energy known as
frequency of the radiation
The energy E of photon depends upon the
increases
As frequency (ν) increases, energy (E) of light
Electronic (UV, Vis)
Energy can be transferred to the ff forms
Associated with the overall motion of electrons around the nuclei
Vibrational (IR)
Energy can be transferred to the ff forms
Associated with the motion of atoms within the molecule
Rotational (IR, Microwave)
Energy can be transferred to the ff forms
Associated with the overall rotation of the molecule
Translational (not quantized)
Energy can be transferred to the ff forms
Associated with the motion of the molecule as a whole
Absorbed energy
A molecule may exist only in certain permitted energy states and cannot possess any arbitrary amount of energy
Electronic energy (UV, Vis)
Involves the transfer of electrons between different electronic state à ground state to the excited state
fluorescence
When UV is absorbed an electron is raised from the ground state to excited state, loses its vibrational energy, decays to ground state, emitting UV or visible light →
phosphorescence
When the transition back to the ground state is delayed it results to
Stretching
distance between two atoms increases or decreases
Bending (deformation)
position of the atom changes relative to the original bond axis
Transmittance
Defined as the ratio of the intensity of light emerging from the solution (I) to that of incident light entering (Io)
Absorbance
Also called optical density, absorbancy, extinction coefficient
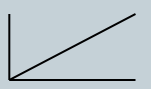
Absorbance α concentration graph
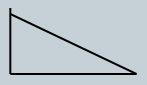
Transmittance 1/ α to concentration and absorbance
Beer’ s Law
states that the power of a transmitted radiant beam decreases exponentially as the concentration of the solution containing the absorbing chemical species increases arithmetically.
Beer’ s Law
Absorbance is directly proportional to concentration
Lambert or Bouguer’s Law
states that the power of a transmitted radiant beam decreases exponentially as the thickness of the solution containing the absorbing chemical species increases arithmetically
Beer–Lambert or Beer–Bouguer’ s Law
is a combination of the above law and relates the power of the incident and the transmitted radiant beam to the thickness and concentration of the solution containing the absorbing chemical species.
Spectrophotometer
These instruments may either be manually operated or automatic recording instruments
Instruments have
1.) radiant power source
2.) radiant energy dispersing device
3.) a sample compartment
4.) the associated electronics which permit the measurement of radiant power transmitted by the sample.
SA curve
spectral absorbance curve, plot of absorbance values against wavelength
Beer’s plot
plot of absorbance values against a series of known solute concentration