Carriage of carbon dioxide
1/5
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
6 Terms
How is carbon dioxide carried
5% is dissolved in plasma
10% as carbaminohaemoglobin, forms when CO2 combines with haemoglobin
85% as hydrogen carbonate ions, HCO3,-
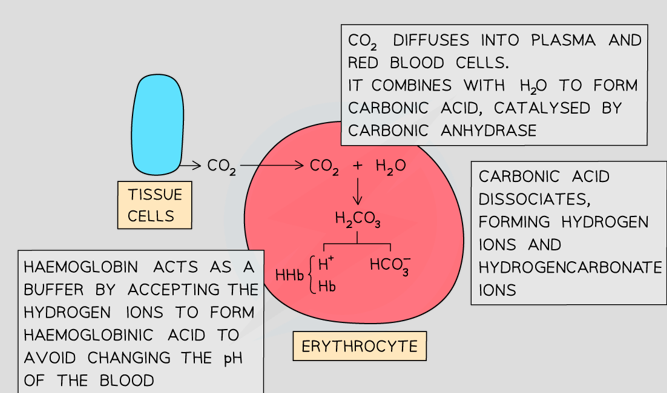
How do the hydrogen carbonate ions form
CO2 diffuses from plasma to RBC and in RBC combines with H2O to form H2CO3 (carbonic acid) CO2 + H2O ⇌ H2CO3
Carbonic acid dissociates readily into H+ and HCO3- ions H2CO3 ⇌ HCO3– + H+
Carbonic anhydrase catalyses this reaction
What is the chloride shift
Carbonate ions, which are negatively charged, are transported out of RBC via transport proteins. To prevent an electrical imbalance negatively charged chloride ions are transported into RBC via the same transport proteins.
What would happen if there was no chloride shift
There would be a build up of H+ ions decreasing the pH of blood which would affect enzyme activity
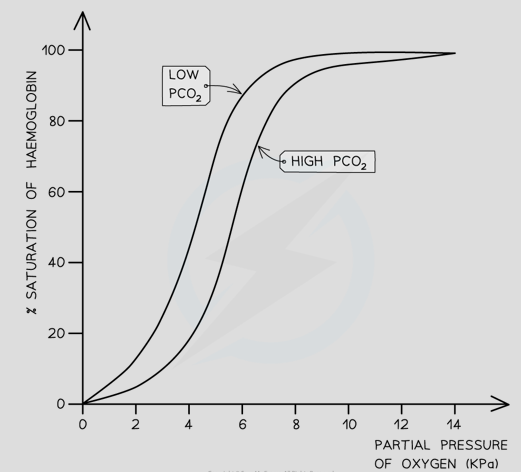
What is the Bohr shift
This occurs when partial pressure of carbon dioxide in blood is high. H+ ions which are formed from carbonic acid change quaternary structure of haemoglobin, so as a result they have a lower affinity for oxygen. This is useful for respiring tissue as oxygen is released by haemoglobin more readily
How does the Bohr shift affect the dissociation curve
Dissociation curve shifts to the right meaning at any given partial pressure of oxygen, the % saturation of haemoglobin is lower at higher levels of CO2