6.2 Hardenability and Heat treatment for Steel
1/26
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
27 Terms
Definition of hardenability
ability of hardening steel→ how easy it is to form marsentite
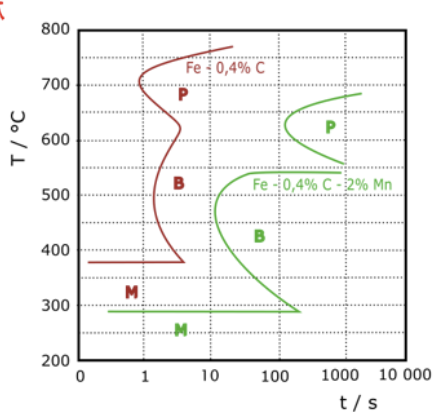
How to judge hardenability with TTT diagrams
for the same cooling rate, judge TTT diagram of different compositions to which one allows 100% marsentite to form
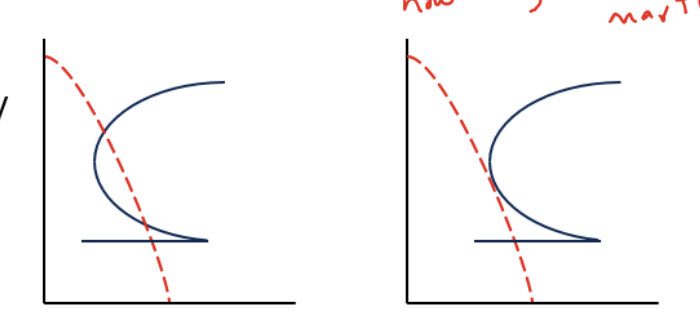
How does hardenibility changes with alloying additions
addition of such alloy leads to very high hardenability AKA low cooling rate still leads to 100% marsentite formation
(C curve translate down)
What is a CCT diagram
Continous cooling diagram
unlike TTT, which consider isothermal cooling, CCT plots for continuous cooling
C curve shifts down and right
(dotted line-TTT, solid line-CCT)
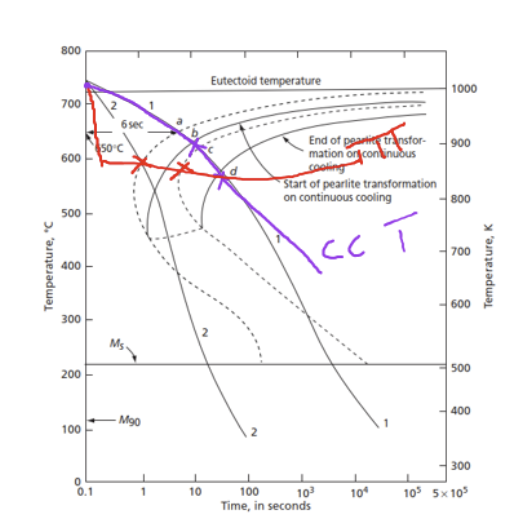
Reason for difference of CCT and TTT C curve
thermal activation energy decreases as time goes on
→ forces material to transform as temperature drop, causing curve to drop and take a longer time
Effect of annealing
remove resdual/manufacturing stress
remove work hardening and restore ductility
soften steel
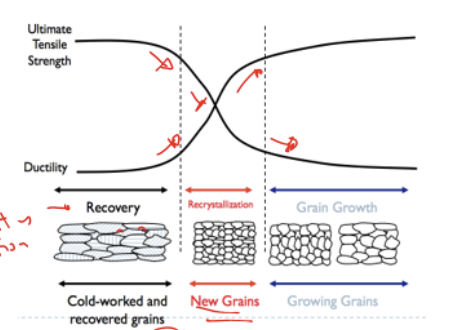
3 Steps of annealing
Recovery: dislocations entangles and DLCT density decreases
→oppositely oriented DLCT cancels out
Recrystallisation: new grains forms to form strain-free microstructure
→ strength decrease and ductility increases
grain growth
→ time-induced process that can be controlled to control final property
Condition of Temperature for process annealing to occur
Apply to pre-strained/work hardened auestenite that cools
Temp: around 100 degrees eutectic temperature
(refer to stress relief section of diagram)
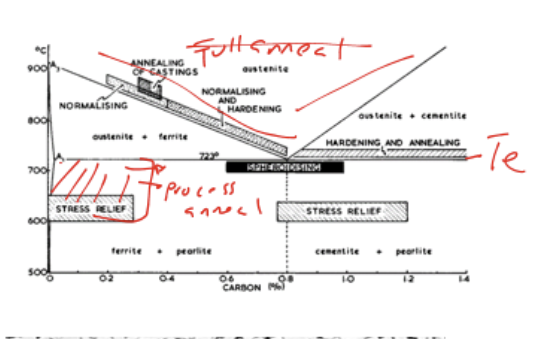
Effect of process anneal in terms of property and microstructurely
remove work hardening and restore ductility
→ only ferrite recrystallise while cementite remains its stretched shape
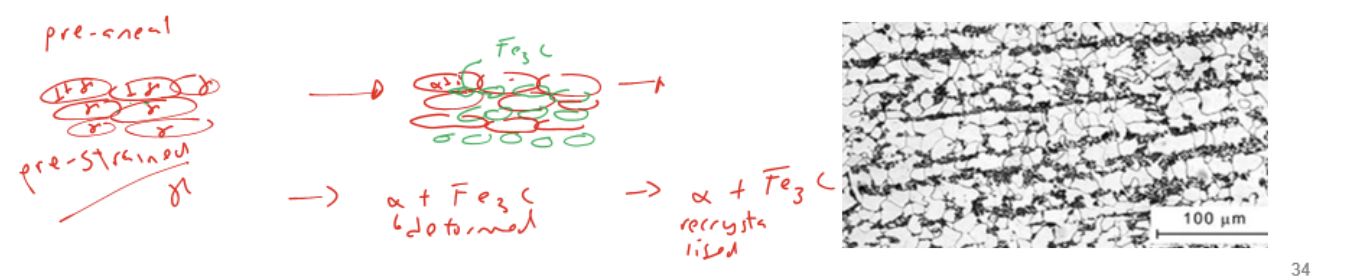
Effect of full anneal
restore equilibirum microstructure of hypoeutectoid steel
microstructure become equiaxed again
Temperature for full annealing
heated at austenite region and cooled quickly
Process of normalising
heat hypoeutectic steel at austenite region and cooled at a slowly (but faster rate than full aneal)
Effect of normalising
form finer equibilibrium of ferrite and pearlite

Process and effect of spheroidization
Process:
prolonged heating below eutectoid temp to change shape of cementite to spheres
→ temperature (thus driving force) high enough for cementite to from, but not grow lammenarly, therefore forming spheres
Effect: soften steel for easy machining
Process and effect of quenching
cooling quickly to form marsentite
Factors affecting end result of quenching
medium used (quenching speed: saltwater>water>oil)→ cracking could occur if quenching too fast
composition of steel (quenching rate plotted to different TTT used result in different microstructure)
size of component (inside cool slower than outside→ hardening varies in X section)

Reason for tempering after quenching
tempering improve poor property of steel it has after quenching
2 semi-quantitative methods to measure hardenability
Critical diameter/ruling section method
Jominy end-quench test
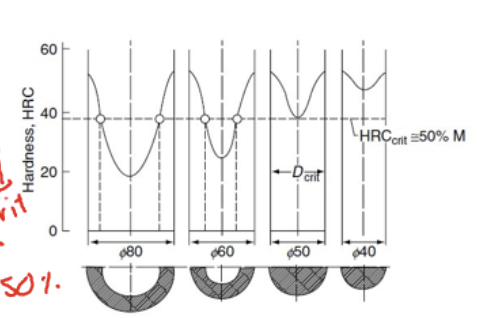
Principle of critical diameter method
Quench a solid steel cylinder and measure hardness across diameter
Critical diameter is when the minimum hardness value drop to 50% of the hardness at the end
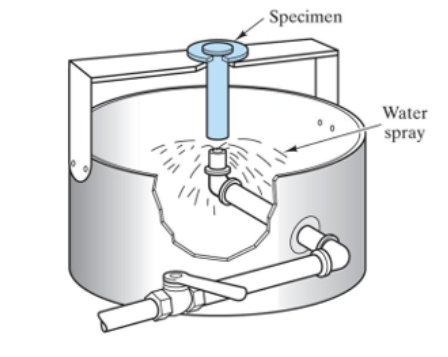
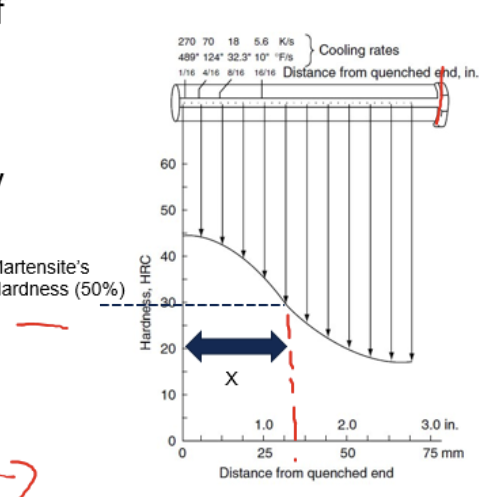
Principle of Jominy end-quench test
Quench the bottom of one cylinder and measure hardness along the length x
→ could be used to predict size of specimen than hardens successfully
→ critical distance is when hardness is 50% of max hardness
2x_crit=D_crit
Process and effect of tempering
Process:
reheat quenched martensite at a not too high temperature ( around 500 degrees)
hold
quench again
effect: reduce brittleness and retain toughness
4 steps in tempering martensite
initial formation of fine ε carbide (Fe5C2) precipitate (<200 degrees)
any retained austenite is converted to bainite (<350 degrees)
→ austenite may be present due to CCT crossing the C curve
Fe5C2 covert to course Fe3C (cementite) (>500)
spheroidisation of Fe3C (over tempering)
Final marsentite phase has no carbon content anymore
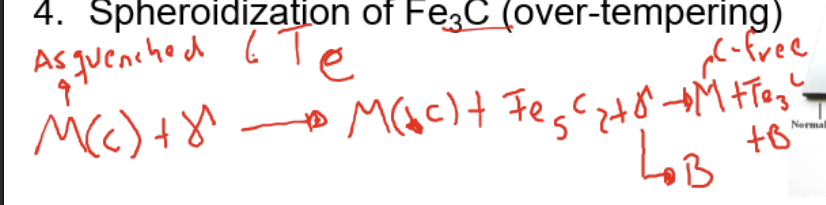
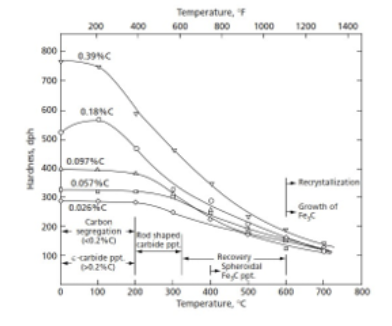
Importance of choosing the correct temperature of heat treatment
No matter the carbon content, as temperature increases, all steel hardness reduces and converges to similar value

Principle of case hardening
process: impart hardened tempered maternsite on surface of component while keeping core tough
Method of case hardening of low carbon steel
heating in a C/CO2 rich atmosphere (T>1000 degrees)
→ carbon diffuses and absorb slowly into centre of steel
form martensite
Method of case hardening for high C steel
apply induction/localised heating
→ quench and temper surface regions to form case
