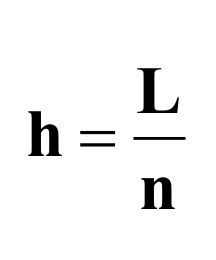CHEM 4631 Exam 4 Lecture Slides 24
1/17
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
18 Terms
What is the definition of Chromatography?
A method used primarily for separation of the components of a sample where the components are distributed between two phases.
What is the First Rule of Chromatography?
Like dissolves like
What happens in Adsorption?
Solute and mobile phase molecules compete for active sites on the surface of the solid stationary phase (the adsorbent).
What happens during a Partition?
Partitioning of a solute between two immiscible liquids but one liquid is held stationary on a solid support. A solute in contact with two immiscible liquids will distribute itself between them according to its distribution coefficient, K
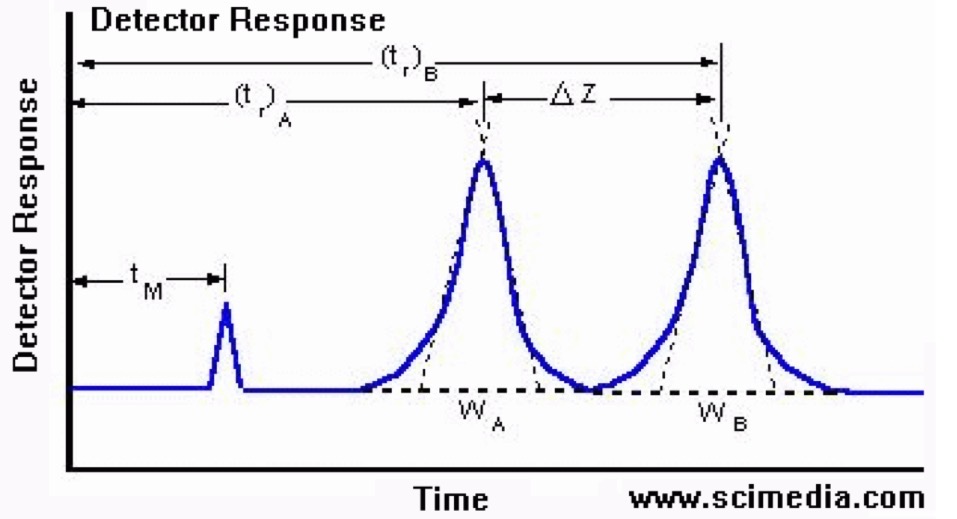
What is Tm according to the Detector Response Graph?
The time it takes a solute to pass through the space occupied by the mobile phase (dead space, column void volume).
Represents no separation process
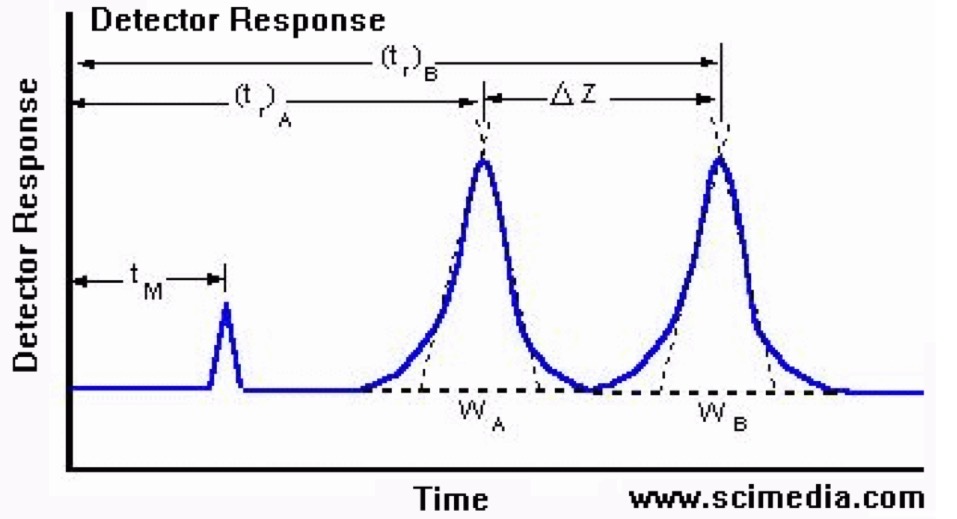
What is Tr according to the Detector Response Graph?
Represents the time the analyte spends retained by the stationary phase.
Represents the separation process or interaction of analyte with stationary phase.
What is the Partition Coefficient K, and what are the variables?
K: Conc. of analyte in the Stationary and Mobile Phase
Cs: Conc. of analyte in Stationary Phase
Cm: Conc. of analyte in Mobile Phase
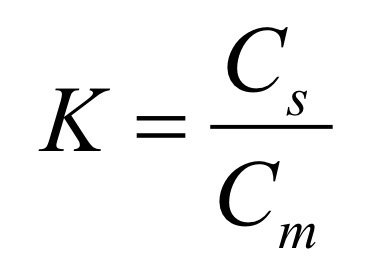
What is the Capacity Factor and what does it measure?
It is the ratio of the total amount of a solute in the stationary phase to the amount in the mobile phase at equilibrium.
Mostly used in HPLC
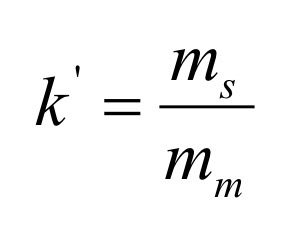
What is the relationship between k’ and K?
Relates equilibrium distribution of analyte within the column to the thermodynamic coefficient, k’
The ratio of volumes of the mobile and stationary phases is called β (Openness of the column)
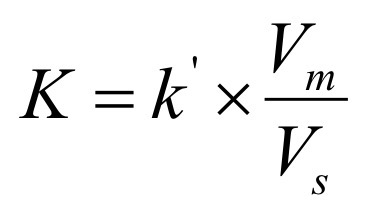
Instead of measuring volumes, which equation relates the probability to time?
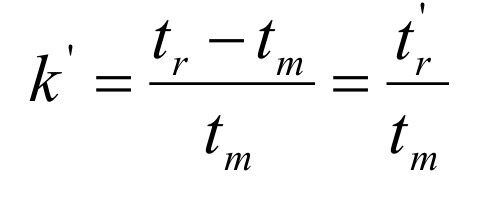
What is the Goal of Chromatography, and what is the term for it?
To achieve sharp symmetrical peaks, thus optimizing analyte separation and improving detection.
Column Efficiency
What does the Sharpness of the Peaks represent?
The efficiency of the chromatographic column/system.
Why is Flow Rate controlled?
Determines the time taken for the sample to go through the column
Too slow, peaks broaden
What are two general approaches to measuring Column Efficiency?
1) Plate Theory
2) Rate Theory
What is Plate Theory?
Models a column as a series of narrow, discrete sections called theoretical plates.
Assume that at each plate equilibrium of the analyte is established between mobile and stationary phase
Movement of analyte and mobile phase is viewed as a series of transfers from one plate to the next. Efficiency of a column increases as the number of theoretical plates increases.
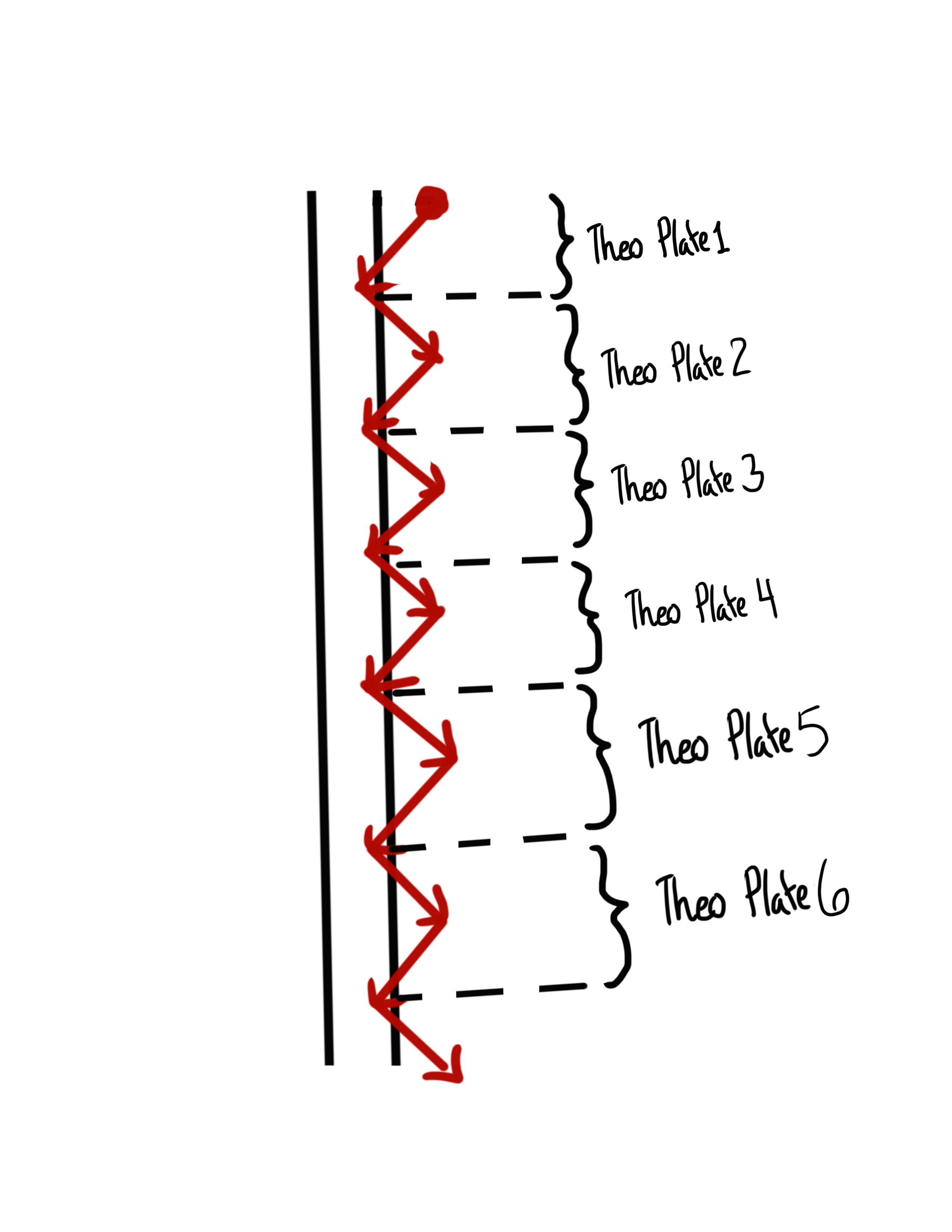
What can be said about Column Efficiency in regard to Plate Theory?
Efficiency of a column increases as the number of theoretical plates increases.
What is the equation for the Number of Plates N (Effective Plate Number)?
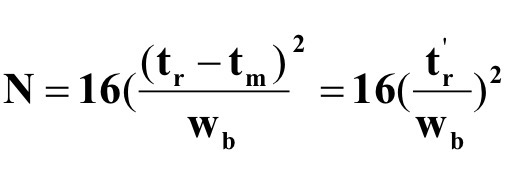
What is the (Effective) Plate Number dependent on in Plate Theory? What variable is included because of this?
Depends on the length of the column
Height of the plate (h) is included