L4 - Major Element Geochemistry
1/47
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
48 Terms
How are Major Element compositions usually reported?
Percent by weight
Atomic Proportion
Percent by volume
Define a Major Element
Has an abundance >1.0 wt. %
Define a Minor Element
Has an abundance of 0.1-1.0 wt. %
Define a Trace Element
Has an abundance of < 0.1 wt. %
What are 3 uses of Major Elements in Geochemistry?
Used to classify rocks
Used to study the chemical control of magmas by the separation of crystals
Control the mineralogy, crystallization, and melting behavior in igneous systems
How are Minor Elements incorporated into the crystal lattice?
Commonly substitute for an element in a principal mineral
Example: Mn2+ for Fe2+ and Mg2+ in olivine and pyroxene
Where are you likely to find minor elements within a rock?
Present in small quantities of accessory minerals.
Zr in Zircon
P in Apatite
Ti in titanite, rutile, or ilmenite
Sr in Plag
How are Trace Elements incorporated into a rock? Why?
Trace Elements substitute for major and minor elements, because they are too dilute to form a separate phase
What is another name for trace elements and why?
Often called Passive Tracers
Their concentration, ratios, and distributions are extremely powerful tools in Earth Science
How are Whole Rock major element concentrations (wt%) typically analyzed?
Analyzed using X-Ray Fluorescence (XRF)
How are mineral major element concentrations typically analyzed?
Analyzed by electron microprobe (EMPA)
What minerals are technically minor in most igneous rocks?
TiO2
MnO
Na2O
K2O
P2O5
What oxide starts a typical major element table? Why?
SiO2 - because it is the most abundant (standard format)
How were most major analyses done until the 1960s?
Major element analyses were determined by wet chemical methods, where ppts (mostly oxides) were formed and then weighed.
Why are Major Elements presented on a table listed as oxides?
Most Cations in rock-forming minerals are bonded to oxygen, so reporting analyses as “oxides“ is reasonable.
Why is it inconvenient to have major elements reported as wt.%?
Differences in major element abundances can be obscured by atomic masses (inclusion of different atoms)
Some workers convert wt% to atom % for a more direct comparison
What is 1wt% equivalent to in ug/g?
1wt% = 10,000 ug/g (ppm)
What are the two different names for iron, and what do they represent?
Ferric = Fe3+ - Fe2O3
Ferrous = Fe2+ - FeO
Represents the two valence states of the major element Iron
What does a higher Ferric/Ferrous ratio represent?
High Fe3+/Fe2+ = Oxidized conditions
What does a lower Ferric/Ferrous ratio represent?
Low Fe3+/Fe2+ = Reduced conditions
One the Major element table, what does H2O+ represent?
Represents the water in the lattice of silicate minerals
released above 110 degrees Celsius
One the Major element table, what does H2O- represent?
Water present as dampness in a rock powder and driven off by heating (not important constituent of a rock)
What is Loss-on-Ignition (LOI) mean?
The total volatile content of a rock is determined by ignition at 800-1000 degrees Celsius
Example: H2O+, CO2, F, Cl, S; can also be, K, Na (if heated for too long)
What is the process of anhydrous renormalization?
Subtract LOI (or H2O) from total
divide each oxide by the new anhydrous total
multiply by 100
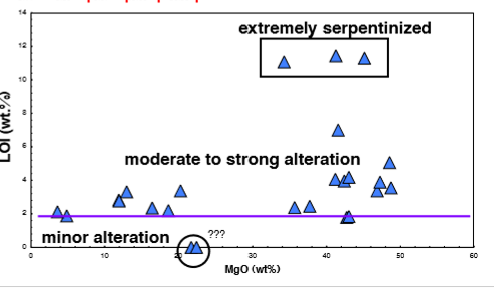
How much H2O% is considered serpentine?
12-14 wt.%
Why is it important to do anhydrous renormalization?
For proper comparison of all major elements
Plot on a TAS Diagram
Image shows plot before normalization
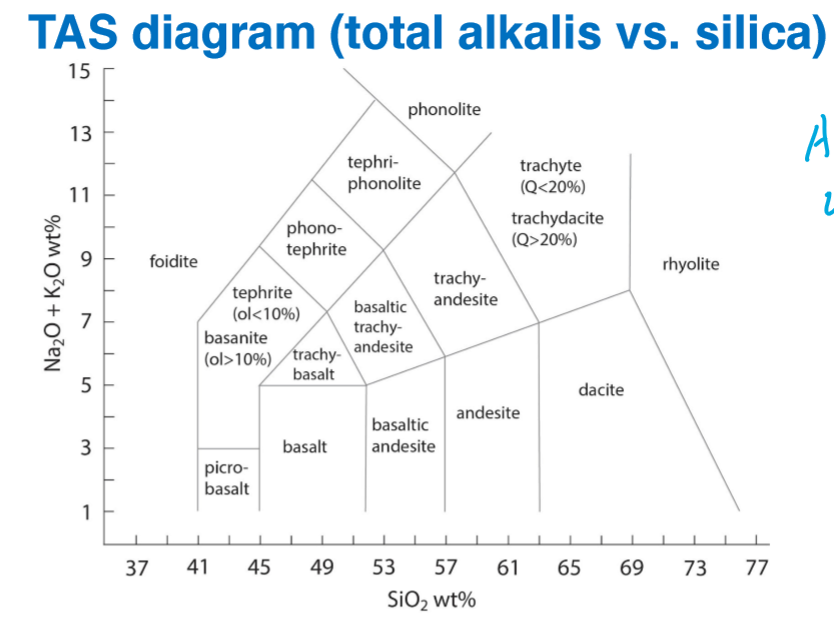
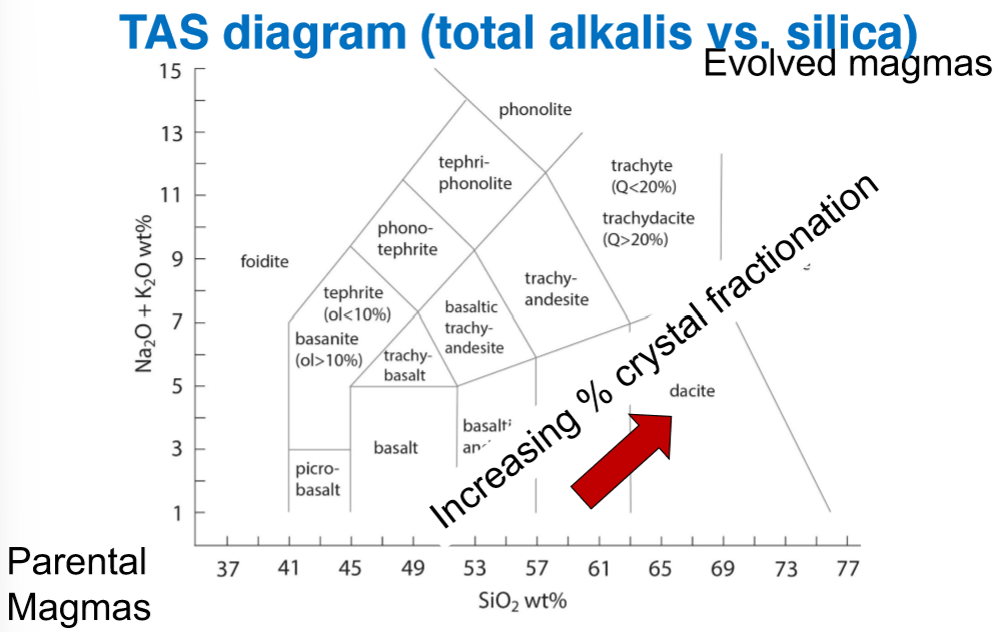
Where are the following located on the TAS Diagram?
Parental Magmas
Evolved Magmas
The trend during increasing crystal fractionation
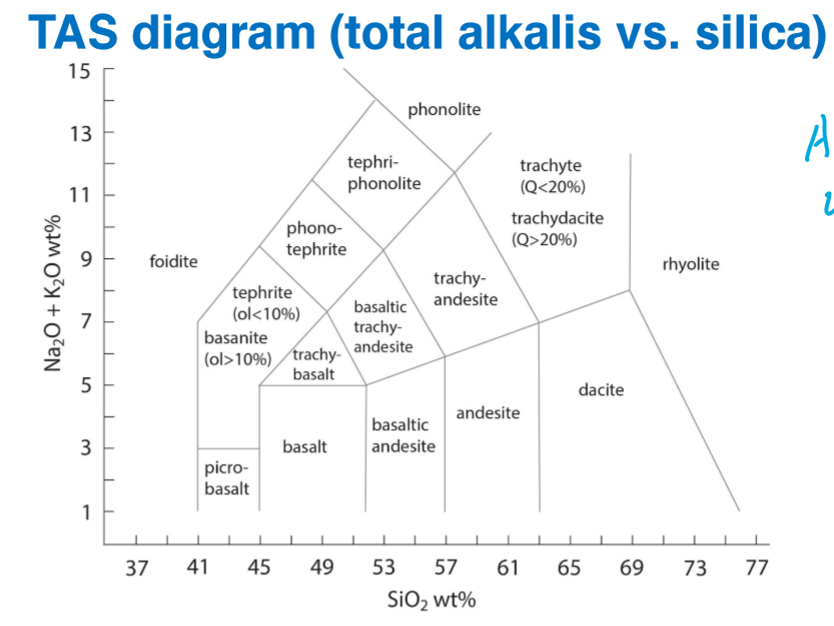
Where are the following located on the TAS Diagram?
Alkaline Rocks
Tholeiitic Rocks
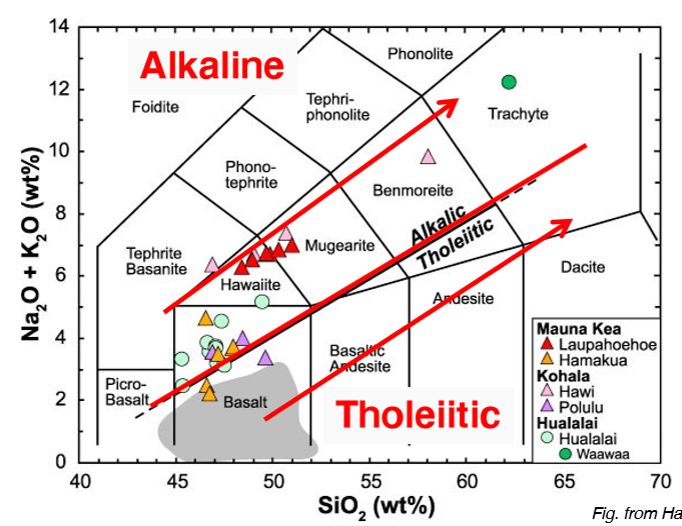
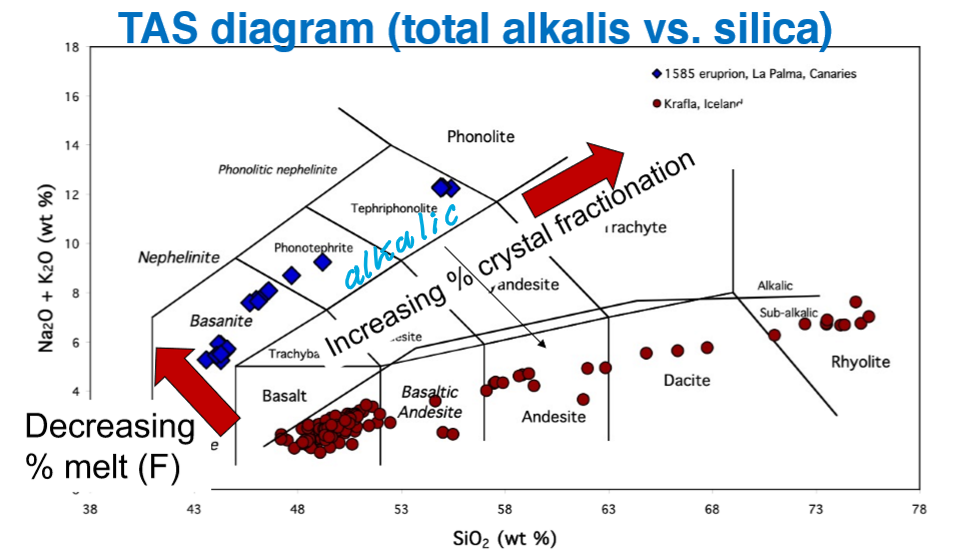
What are the trending directions for:
Decreasing% melt(F)
Increasing % crystal fractionation
What is a Bivariate Plot?
A standard way to examine the major element compositional variability of a dataset
similar to Harker Diagrams, but no SiO2 on the x-axis
What is commonly be plotted on the x-axis in mafic rocks?
MgO (wt%)
What effect do analytical methods involving driving off volatiles at 800-1000 degrees Celsius have on iron?
All the Fe2+ is oxidized in the presence of air to Fe3+; therefore, the results will be reported as Fe2O3(T)
T = total
FeO from sample can then be determined using a titration method/calculation.
Define Loss-On-Ignition (LOI). What temperature needs to be reached?
LOI - The volatile content of a rock is determined by ignition at 800-1000 degrees Celsius that is expressed on a table.
What are some examples of volatile compounds?
-H20
CO2
F
Cl
S
and can also be Na and K, if heated for too long
What are the added compounds for LOI?
O2
Allows for oxidation. Example: FeO → Fe2O3
What oxide should be chosen to be placed on the X-axis?
The oxide used to show the variation of a dataset, and therefore usually the oxide with the greatest absolute range.
What part of the rock represents the major element composition?
The glass or a fine-grained groundmass in volcanic rocks
Define a Co-genetic lava suite.
a suite of lavas that have a common primary melt
What is the best way to test a specific hypothesis in co-genetic lava suites?
Bivariate plots
Can determine melting, fractional crystallization, contamination, and mixing
Describe Fractional Crystallization
The crystallization and physical removal of different minerals occur as magma cools.
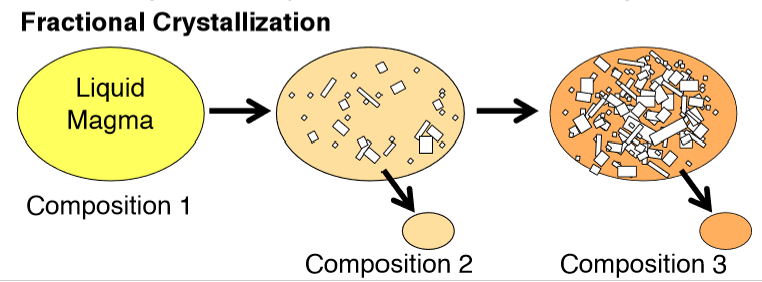
What is the basic principle of mass balance?
“The whole is the sum of its parts“
What are some examples of mass balance?
The major element composition of a sediment can be calculated from the known composition and abundance of clay and quartz fragments
The major element composition of basalt can be calculated from the known starting composition and the composition of the minerals that ppts.
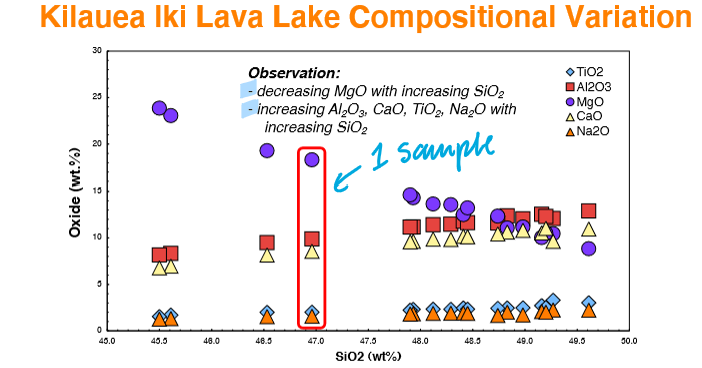
What does a wide range of MgO contents in a table suggest?
Suggests the involvement of one (or more) Mg-rich mineral phase(s) in the chemical variability of these lavas.
What does a very low volatile abundance and total close to 100% suggest?
Indicates that the samples are very fresh and do not have to be renormalized
What type of information can linear relationships of different oxides provide?
Provides important information about how different magmas evolved.
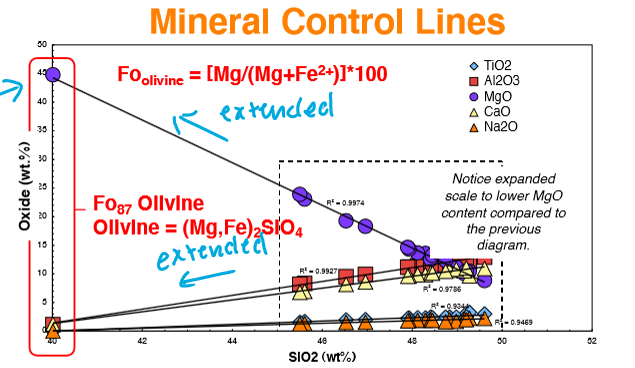
What are control lines, and what do they represent?
Control lines - linear arrays present in a bivariate plot that represent the accumulation of a single mineral phenocryst composition that controls the chemistry of the samples.
can also be considered as “mixing lines“ between the mineral and the magma (in equilibrium with each other)
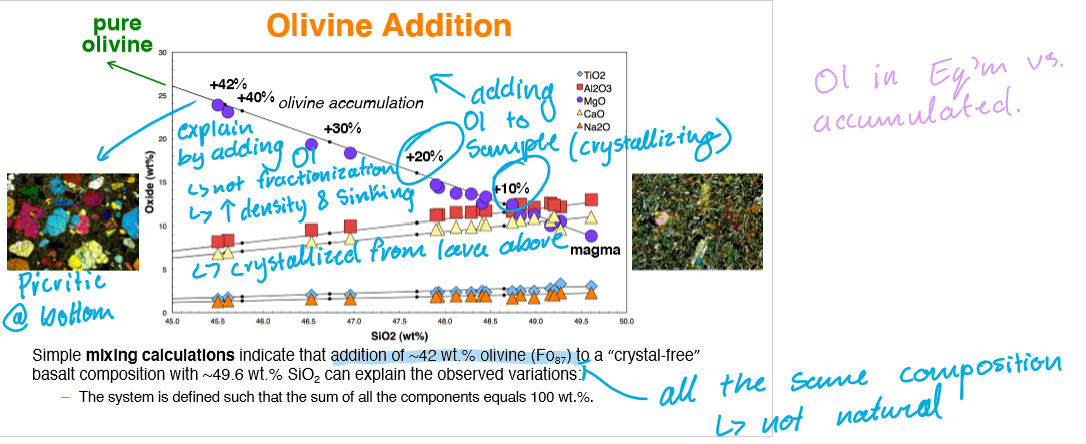
What explains the difference in Picrite (very Ol-rich rock) and Tholeiitic Basalt (Ol-Poor rock)?
Mixing calculations through Olivine addition can be used to explain the observed variations in the rocks.
Olivine accumulation occurs through the crystallization and sinking (high density) of olivine at the bottom of the Kilauea Iki Lava Lake. This causes a Ol-rich sample at the bottom and an Ol-poor sample near the top of the lake.
Note: Phenocryst compositions remain unchanged.
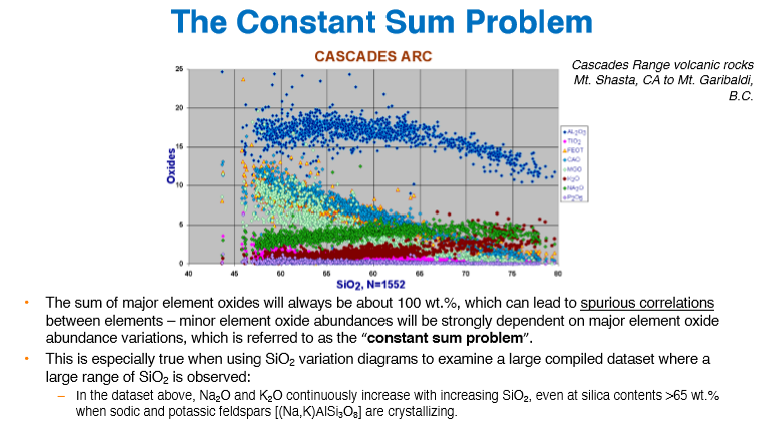
What is the “Constant Sum Problem“?
Minor element oxide abundances will strongly depend on major element oxide abundance variations, creating spurious correlations (since the sum will always be ~100%)