2.4 Alkenes
1/33
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
34 Terms
What are alkenes
Unsaturated hydrocarbons
General formula
CnH2n
Double bond features
Centre of high electron density, so alkanes more reactive
What is a diene
diene is an alkene with two C=C bonds in the main carbon chain

Sigma (σ) Bond
A covalent bond formed by the linear overlap of atomic orbitals resulting in free rotation along the bond axis.
Рі (п) Bond
Рі (п) Bond
A covalent bond formed by the sideways overlap of p-orbitals, which restricts rotation along the bond
Alkene structure diagram

Pi bond freely rotate
Cannot freely rotate as there is restricted rotation about the carbon carbon double bond,
What bond C=C or C-C is stronger?
C=C stronger as contains both sigma and pi bonds
C-C only sigma
What bond C=C or C-C is longer
C=C shorter bond length than C-C
Why are alkenes more reactive than alkanes
The C=C bond in alkenes is a region of high electron density which makes it more susceptible to attack by electron deficient species (Electrophiles). This explains why alkenes are more reactive than alkanes and the pi bond is broken in addition reactions.
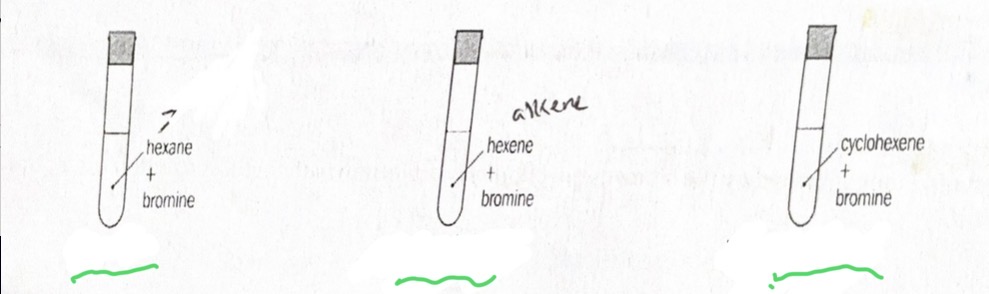
Bromination with bromine water use and results
Additional reaction, test for saturation as unsaturated hydrocarbons decolourise yellow bromine water

What is addition reaction
Addition reaction involves pi bond of a double covalent bond being broken and two species adding on across the double bond.

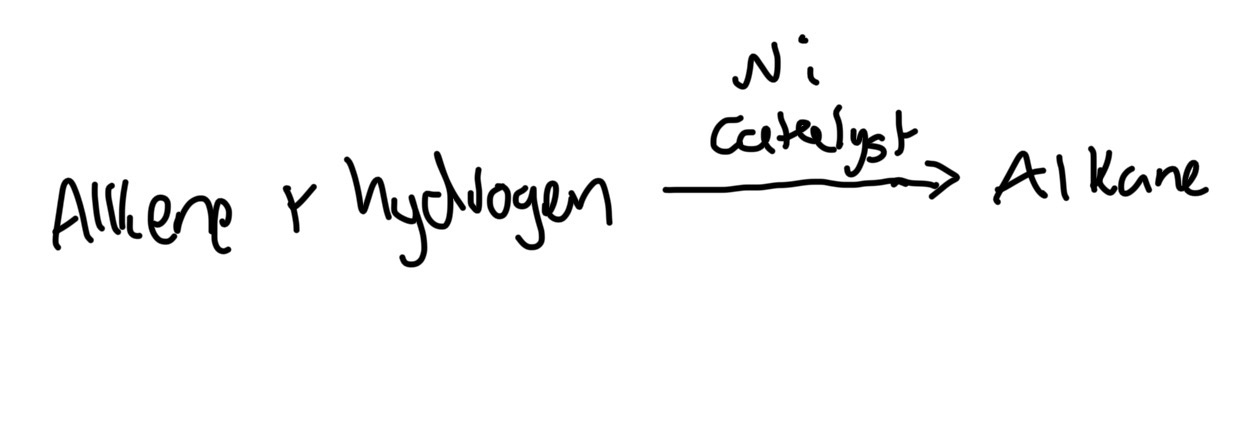
Hydrogenation alkene-alknane reaction conditions
Finely divided solid nickel, 180oC, 4atm
Catalyst increases surface area
hydrogenation reaction use
Manufacture of margarine, oils unsaturated runny, hydrogen increases molecular mass,
Hydrogenation formula
C2H4 + H2 → C2H6
Hydration formula and meaning
Addition of water as steam produce alcohol
C2H4 + H2O(g) → C2H5OH
Conditions for hydration
concentrated phosphoric acid (H3PO4), 300°C, 60 atm.
Electrophile definition
An ion or molecule which attacks regions of high electron density
Addition of a hydrogen halide to an Alkene mechanism ethene and hydrogen bromide
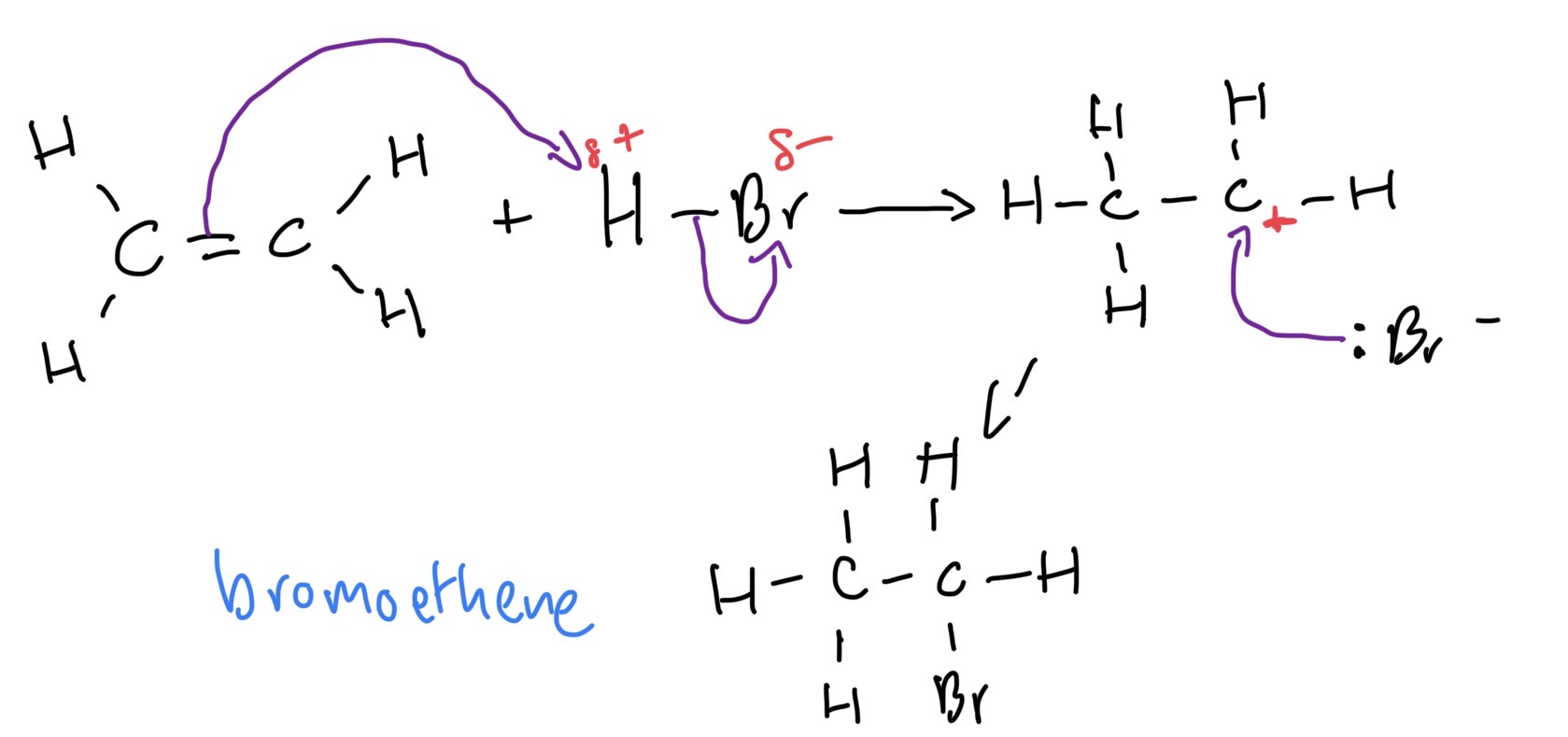
Heterolytic fission
Bond breaking in which the shared electrons go to one atom
Carbo cation definition
A positive ion with the charge centred on a carbon atom
Carbocation reactivity
Highly reactive, reacts instantly, bromoethane formed instantly
Electrophilic addition fro non symmetrical alkenes produces
Major and minor product
What is a primary carbocation
One alkyl group attached to the carbon with the positive charge
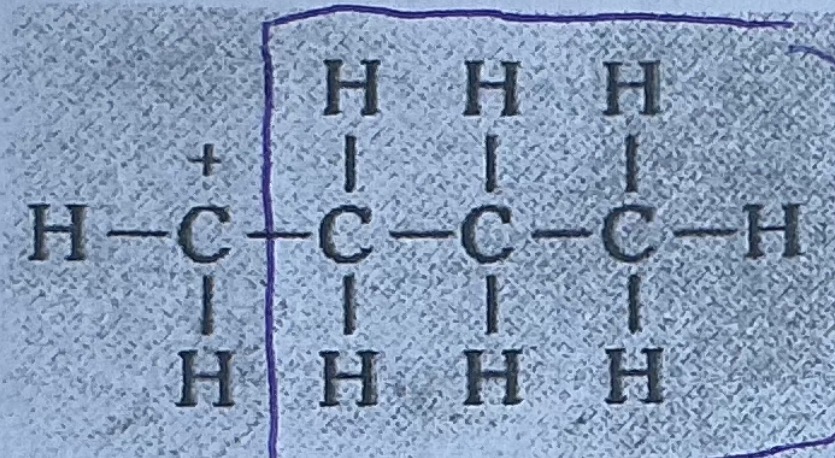
What is secondary carbocation
2 alkyl group attached to the carbon atom with the postive charge
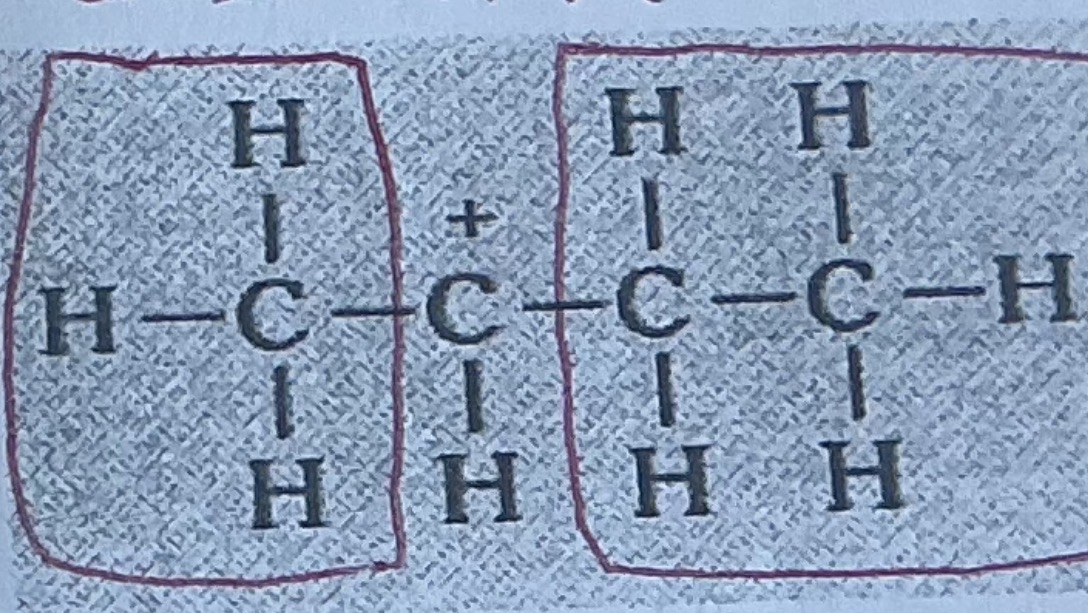
Reactivity of carbo cation primary/sepcmdary and reasons
3o most stable> 2o> 1o least stable
Alkyl groups tend to donate electrons carbon atom attach to.
Markovnikov's Rule:
When we add an HX molecule to an alkene, the H atom will join on to the carbon that already has more Hatoms.
Why are products not equal amounts
Carbocations intermediates produced, tertiary carbocations macro stable than secondary carbocation leads to formation major product
So Br and Cl will join to carbon inside
What are polymers
(Polymers are long chain molecules made up from joining together small molecules, called monomers. which are linked together.
What is polymerisation
Polymerisation is the joining together of small molecules to form a large molecule, addition reaction
Ethene polymerisation
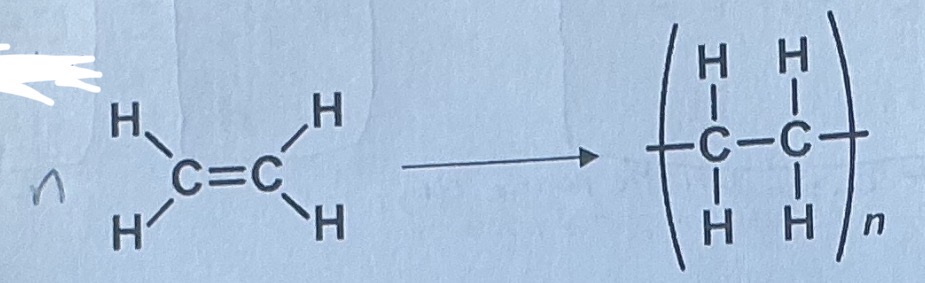
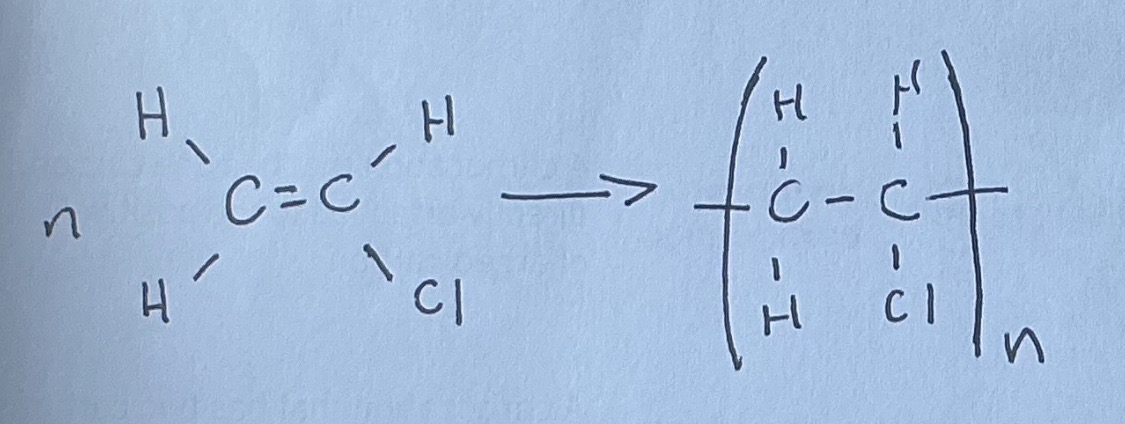
chloroethane: Polymerisation
polyvinyl chloride (PVC)
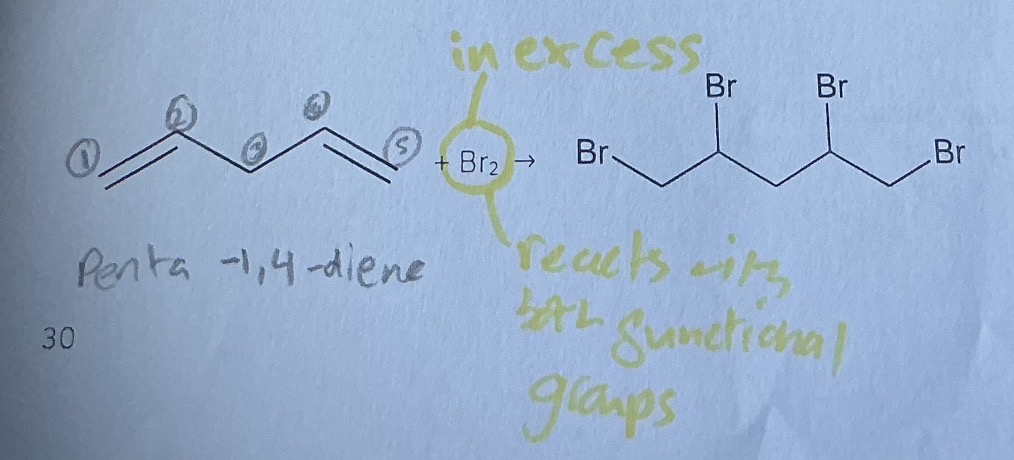
Excess reagent of bromine with penta-1,4-diene