1. biological molecules
1/228
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
229 Terms
how can you test for reducing sugars?
(typical) Benedict’s test:
place 2 cm³ of liquid testing substance in a boiling tube (if not liquid, first crush w/ pestle and mortar and add distilled water, then use filter paper and funnel to create a filtrate)
add 10 drops of Benedict’s
place in a boiling water bath for 3-5 mins
if positive - blue → green/yellow/orange/brick red precipitate (depending on conc of reducing sugar)

how can you test for non reducing sugars?
(altered) Benedict’s test:
boil in diute HCl (to hydrolyse the non-reducing sugar)
add sodium hydrogen carbonate (to neutralise solution)
place 2 cm³ of liquid testing substance in a boiling tube (if not liquid, first crush w/ pestle and mortar and add distilled water, then use filter paper and funnel to create a filtrate)
add 10 drops of Benedict’s
place in a boiling water bath for 3-5 mins
if positive - blue → green/yellow/orange/brick red precipitate (depending on conc of non reducing sugar)

name the reducing sugars:
(all monosaccharides)
glucose
galactose
fructose
maltose
lactose
name a non reducing sugar:
lactose
what is the difference between a reducing and a non reducing sugar?
reducing sugars act as reducing agents in chemical reactions, whereas non reduicng sugars do not
how can you test for starch?
iodine test:
place 3 cm³ of liquid testing substance in a boiling tube (if not liquid, first crush w/ pestle and mortar and add distilled water, then use filter paper and funnel to create a filtrate)
add 1 cm³ of iodine solution (containing iodine and potassium iodide)
if positive - orange → blue black

how can you test for lipids?
emulsion test:
add ethanol then water and shake
if lipid present, white emulsion should form

why do we not filter the lipid solution when testing for lipids?
lipids may stick to filter paper
how can you test for proteins?
biuret test:
place 3 cm³ of liquid testing substance in a boiling tube (if not liquid, first crush w/ pestle and mortar and add distilled water, then use filter paper and funnel to create a filtrate)
add 3 cm³ of dilute NaOH solution and mix
add 10 drops of dilute copper (II) sulfate solution and mix
(this is biuret solution - may be premixed)
if positive - blue → violet

why would you get a negative result in the biuret test for a solution w/ just amino acids (and not proteins)?
tests for peptide bonds (which would not be present)
what is a calibration curve used for?
to demonstrate the conc of a substance in an unknown sample
creatinine detecting solution reacts w/ creatinine to produce an orange colour - how could you produce a calibration curve for creatinine?
use distilled water and creatinine solutio to produce a serial dilution
add creatinine detecting solution to each soltuion
use a known/specified/constant vol of a soltuion (e.g. diluted creatinine solution)
record absorbance/transmission of solution(s)using a colourimeter
plot dilution/conc of creatinine solution against absorbance/transmission
summarise how a colourimeter works
colourimeter detects how much light is absorbed by solution (how much of the light that is travelling through the solution is being stopped by the solution)
the more concentrated the solution, the more light it will absorb
we can use this to determine to conc of a solution
what is a macromolecule?
a molecule built up from much smaller molecules
why are all polymers macromolecules, but not all macromolecules monomers?
a polymer always consists of many monomers
whereas a macromolecule is simply a giant molecule which may or may not contain repeating units
what is a monomer? what is a polymer?
monomer: subunits of life - many can bond together to form a polymer
polymer: many monomers bonded together
what is a condensation reaction?
reaction which joins 2 molecules together with the formation of a chemical bond and involves the elimination of a molecule of water
enzymes are required!
what is a hydrolysis reaction?
reaction which breaks a chemical bond between two molecules and involves the use of a water molecule (e.g. when breaking polymers into monomers)
enzymes are required!
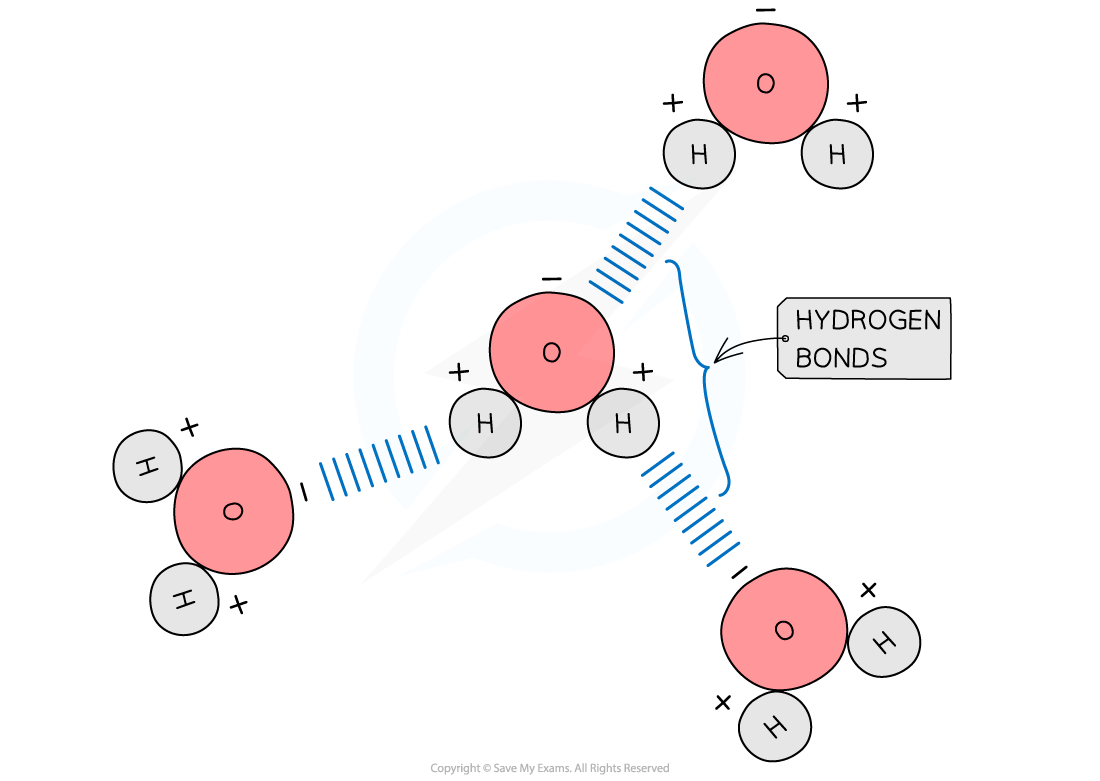
what is hydrogen bonding? how are hydrogen bonds formed?
type of intermolecular attraction (NOT a covalent bond!)
formed by a H atom covale ntly bonded to a very electronegative atom (such as an N, O or F atom) and another electronegative atom
this is made possible due to the partial charges of the atoms
single hydrogen bonds are weak, but many hydrogen bonds are strong
how do we represent partial +ve/-ve charge on an atom?
𝛿+/𝛿-
why is water a dipole?
charge of the water molecule is unevenly distributed
rank the strength of single bonds of hydrogen, covalent and ionic bonds
strongest to weakest: covalent, ionic, hydrogen
what are some examples of monosaccharides?
glucose, galactose, fructose
what is a disaccharide?
two monosaccharides joined together
what are some examples of disaccharides?
maltose
lactose
sucrose
what is a polysaccharide?
polymers, made up of repeating units of monosaccharides
joined by glycosidic bonds
formed by condensation reactions
H bonds between molecules of water
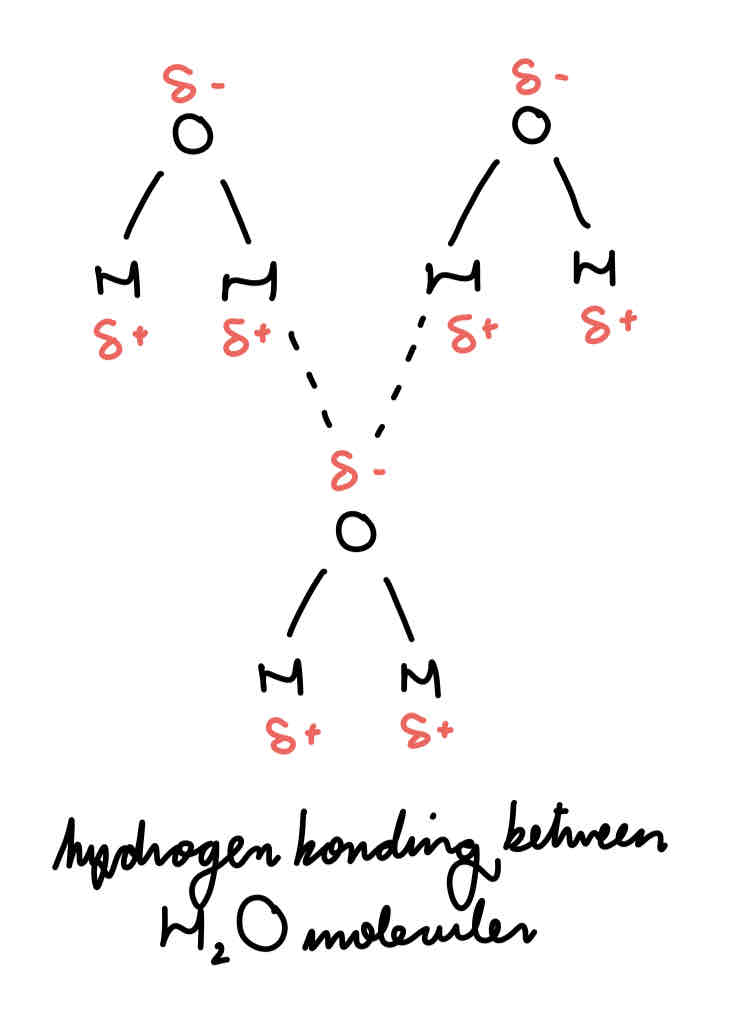
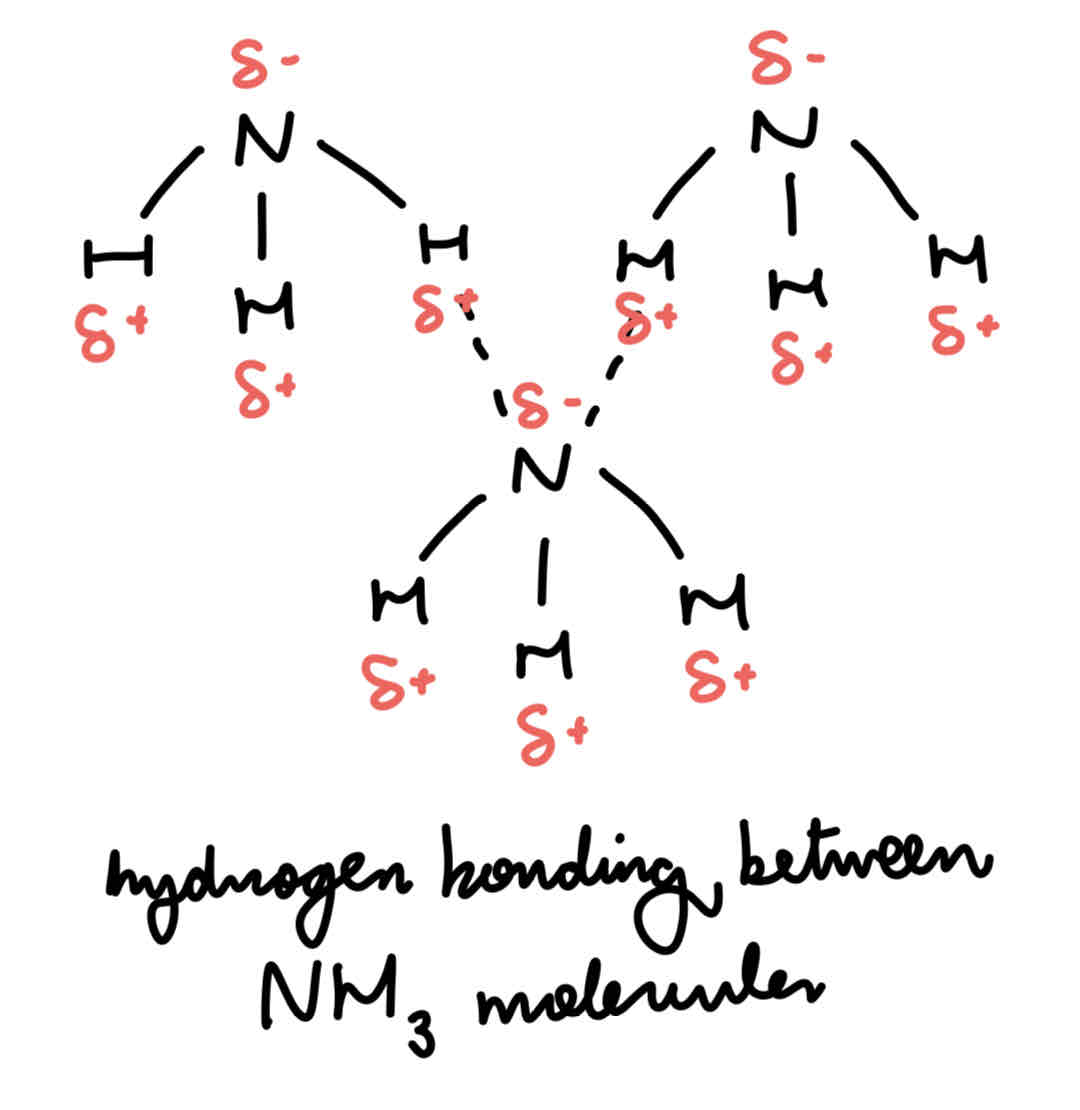
H bonds between molecules of ammonia
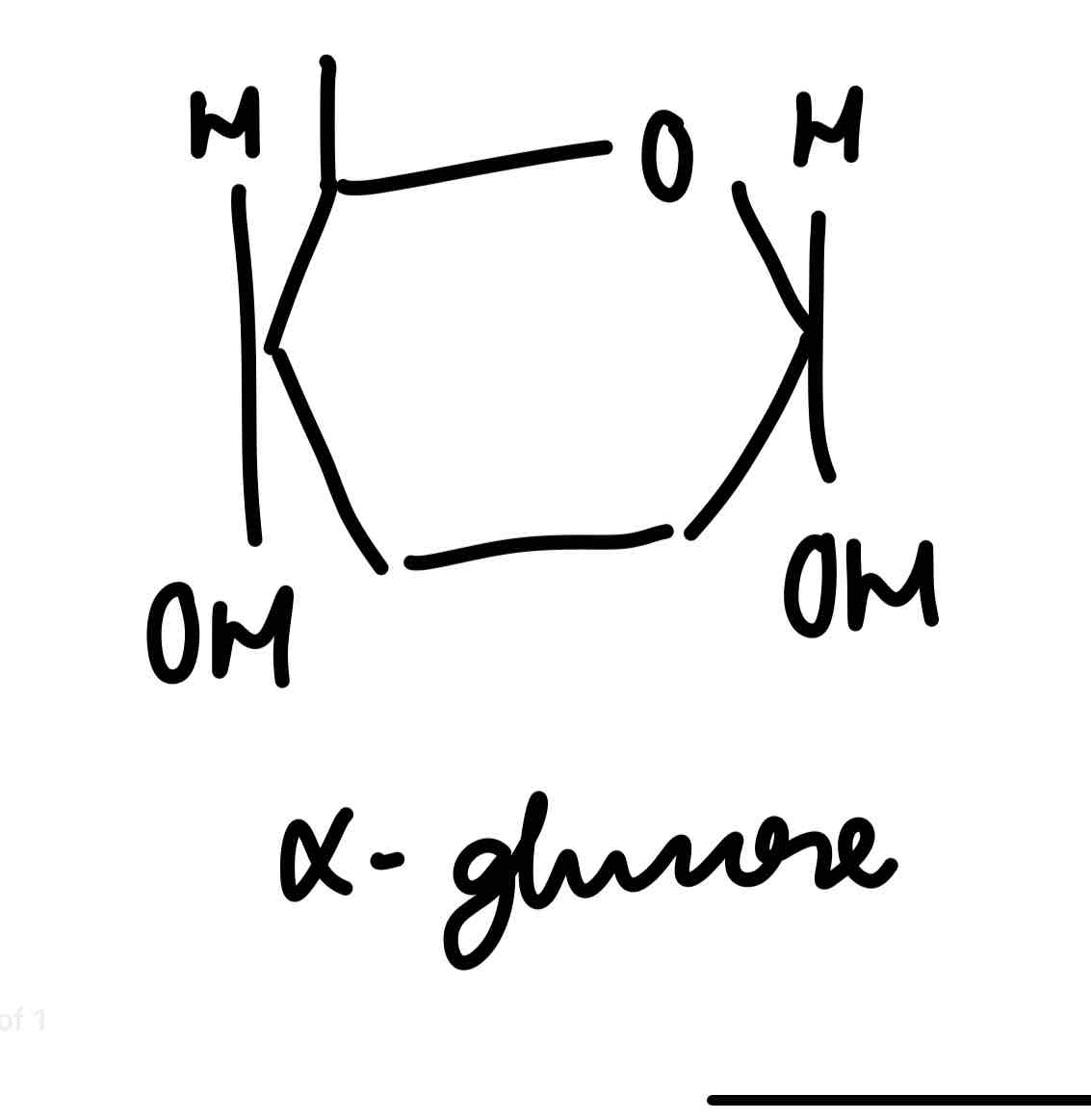
α-glucose
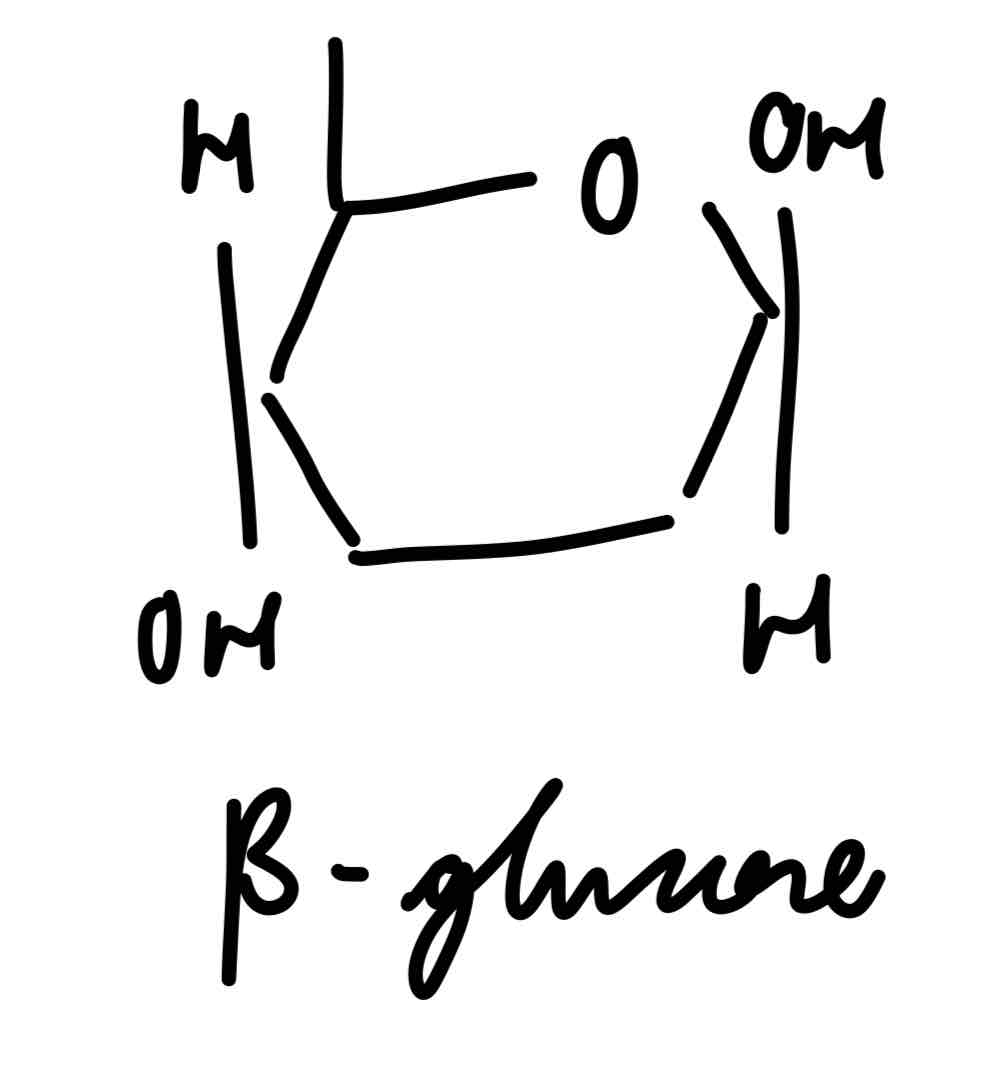
β-glucose
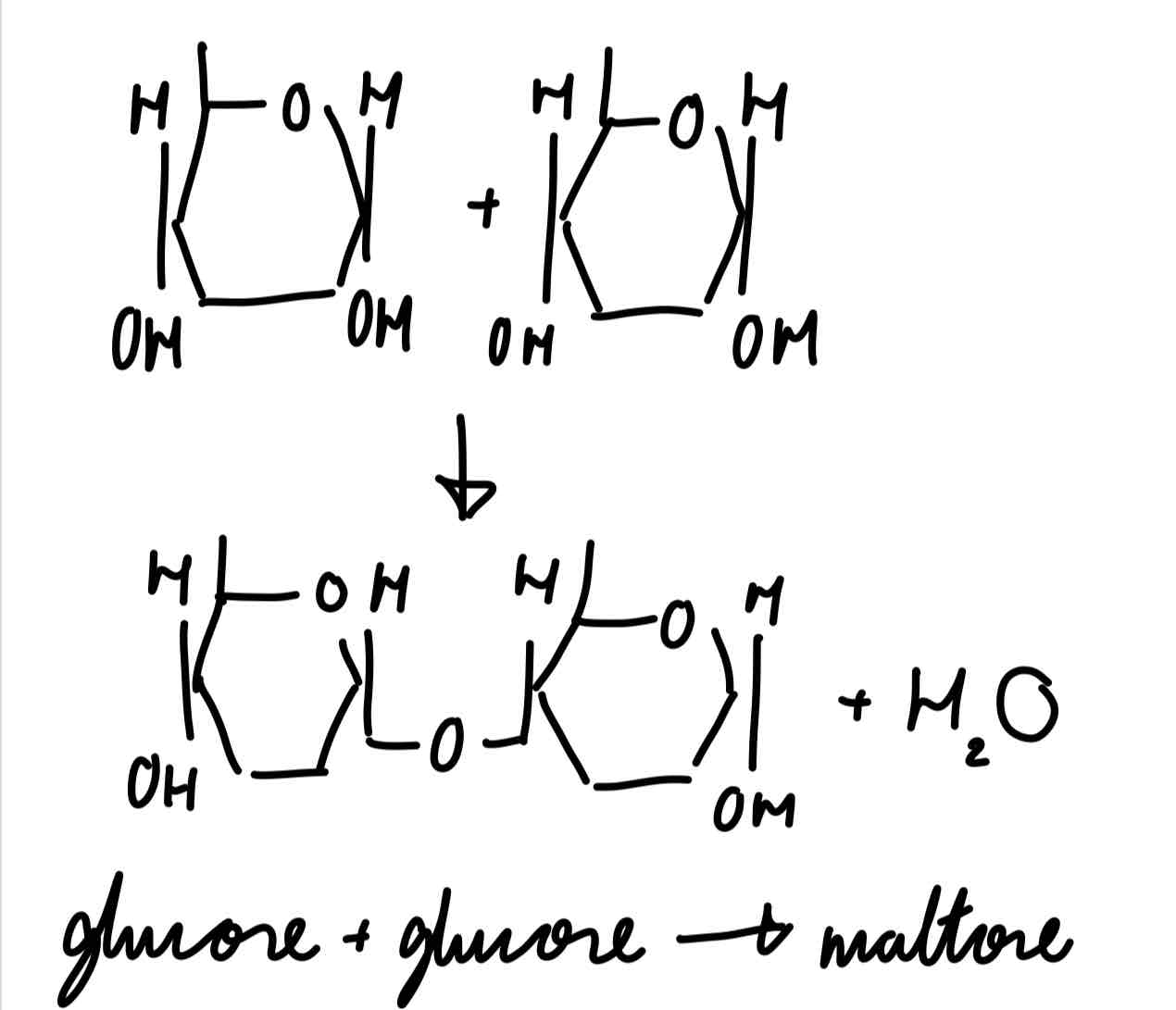
condensation reaction to form maltose
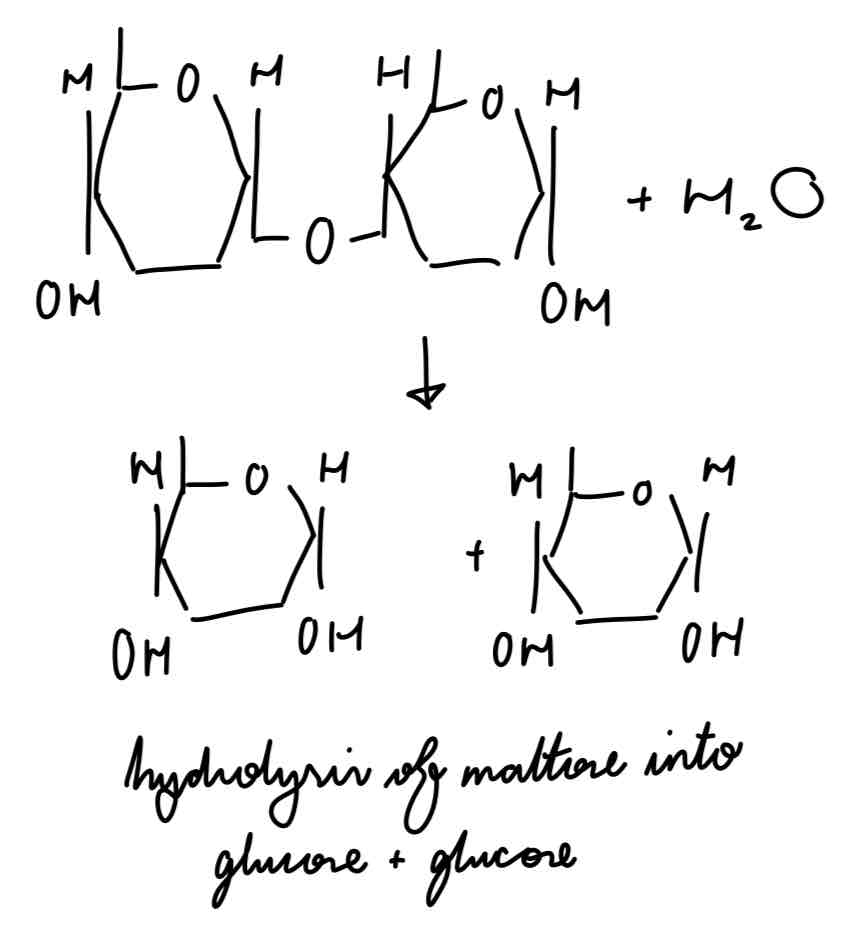
hydrolysis of maltose (w/ alpha-glucose)
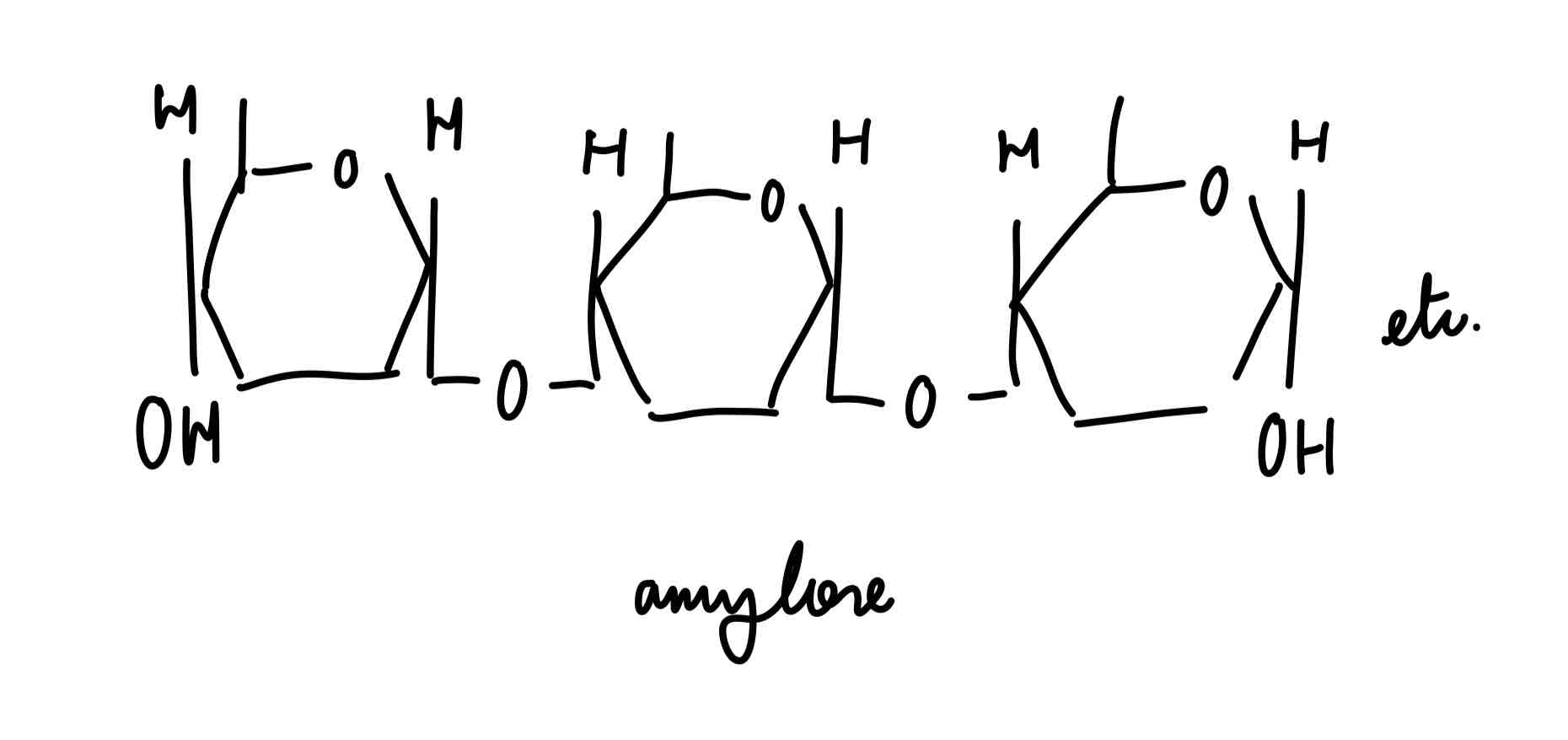
amylose
amylopectin
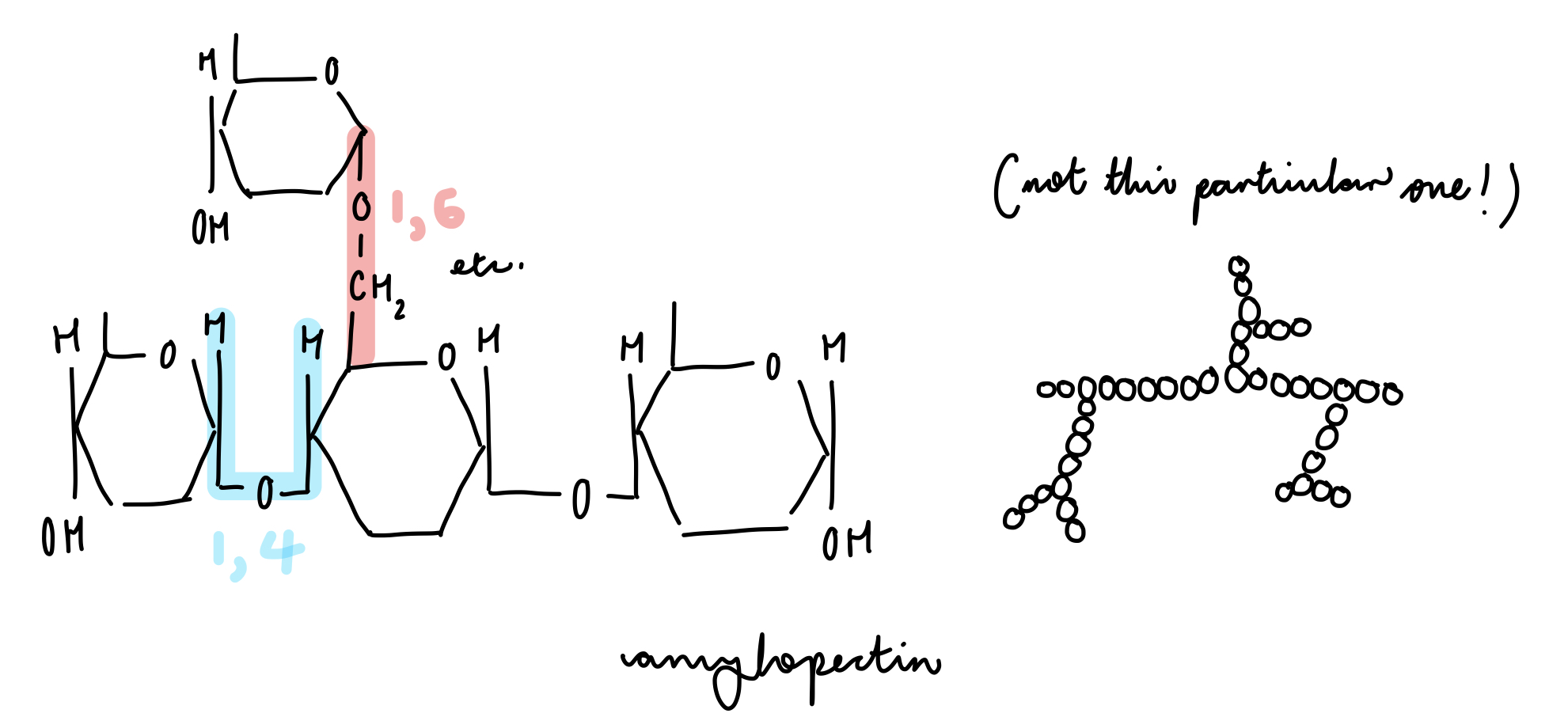
glycogen
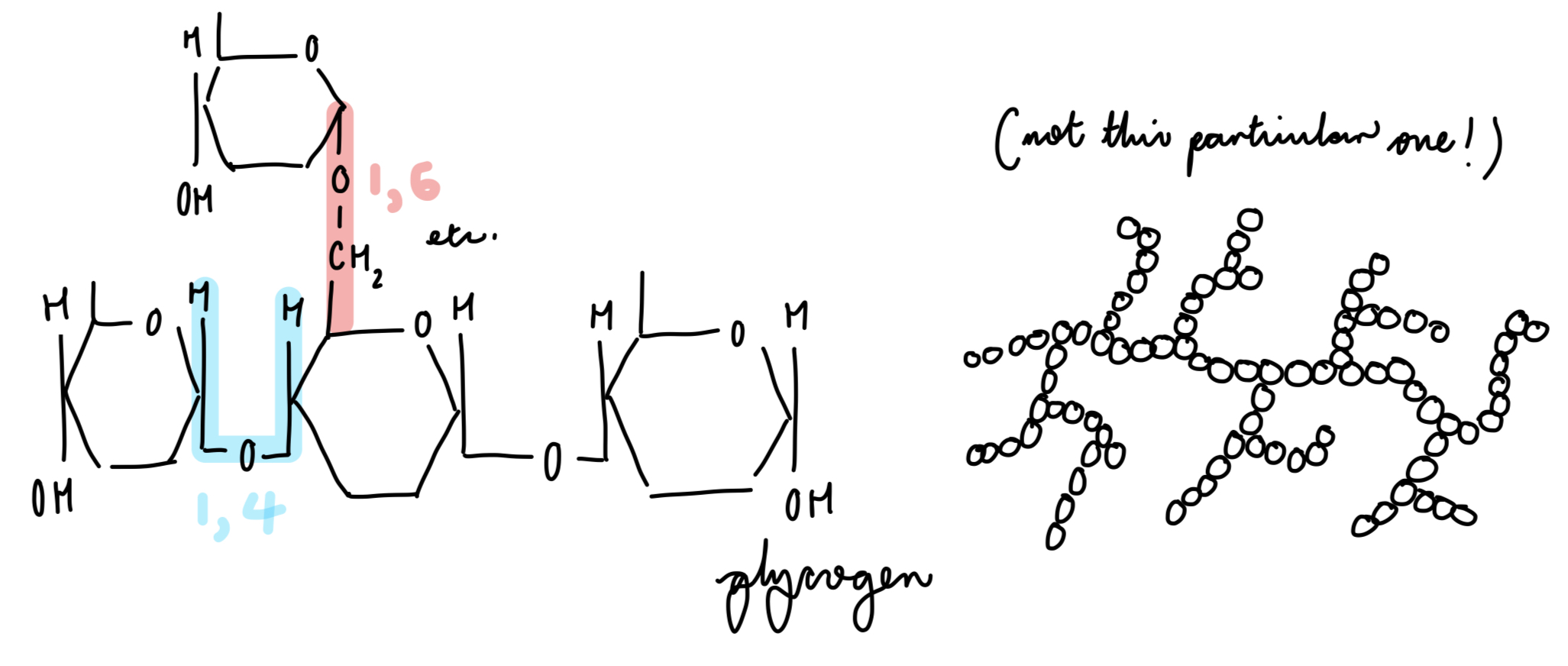
cellulose
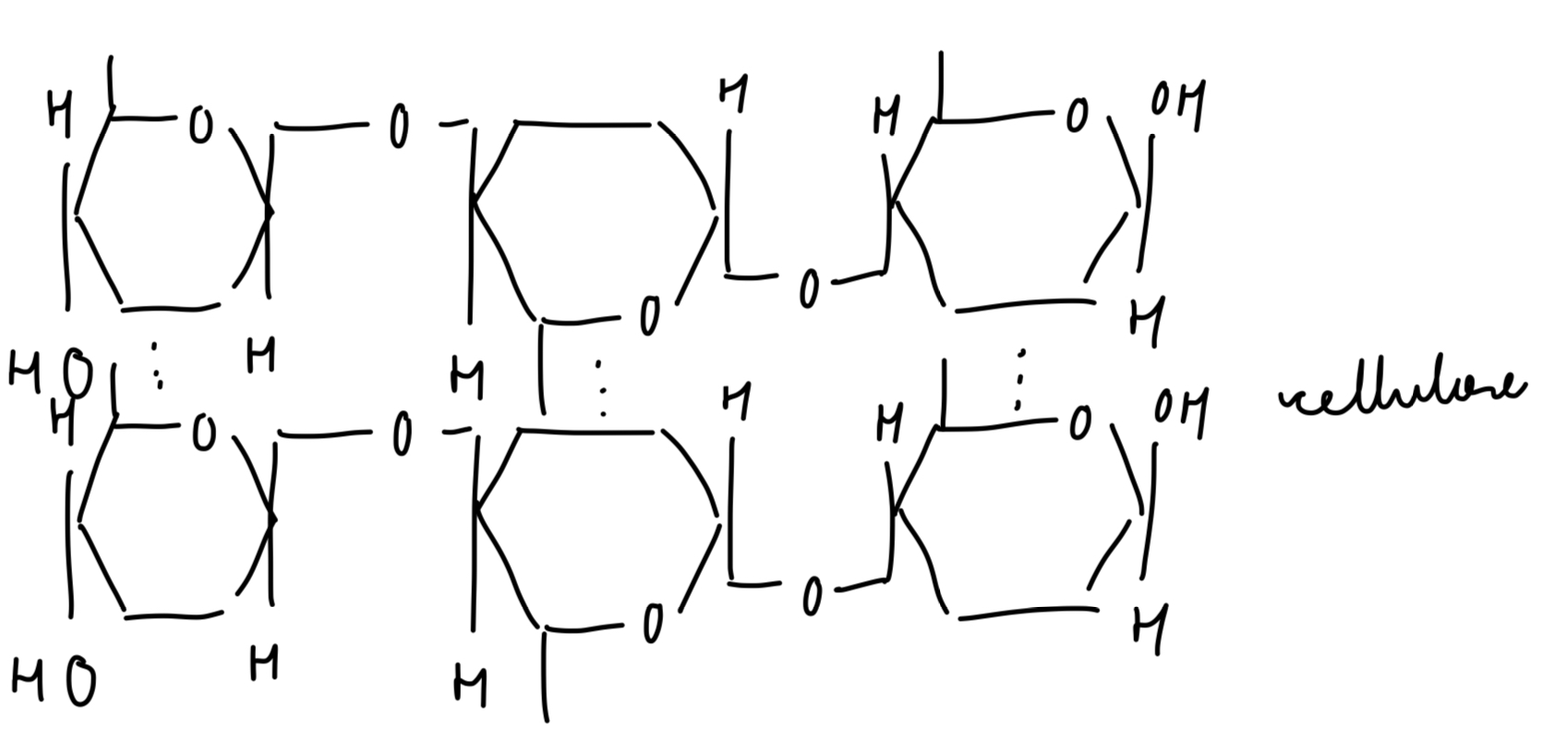
unsaturated triglyceride
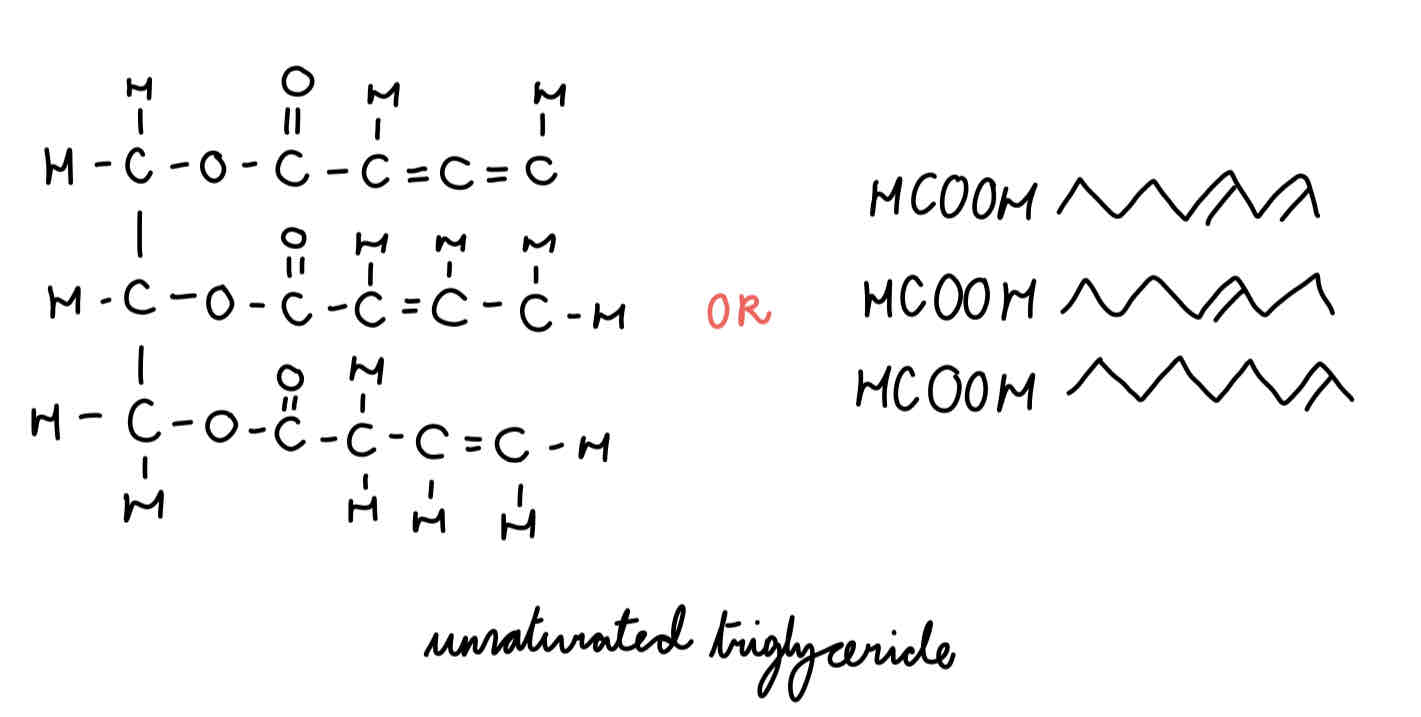
polysaturated triglyceride
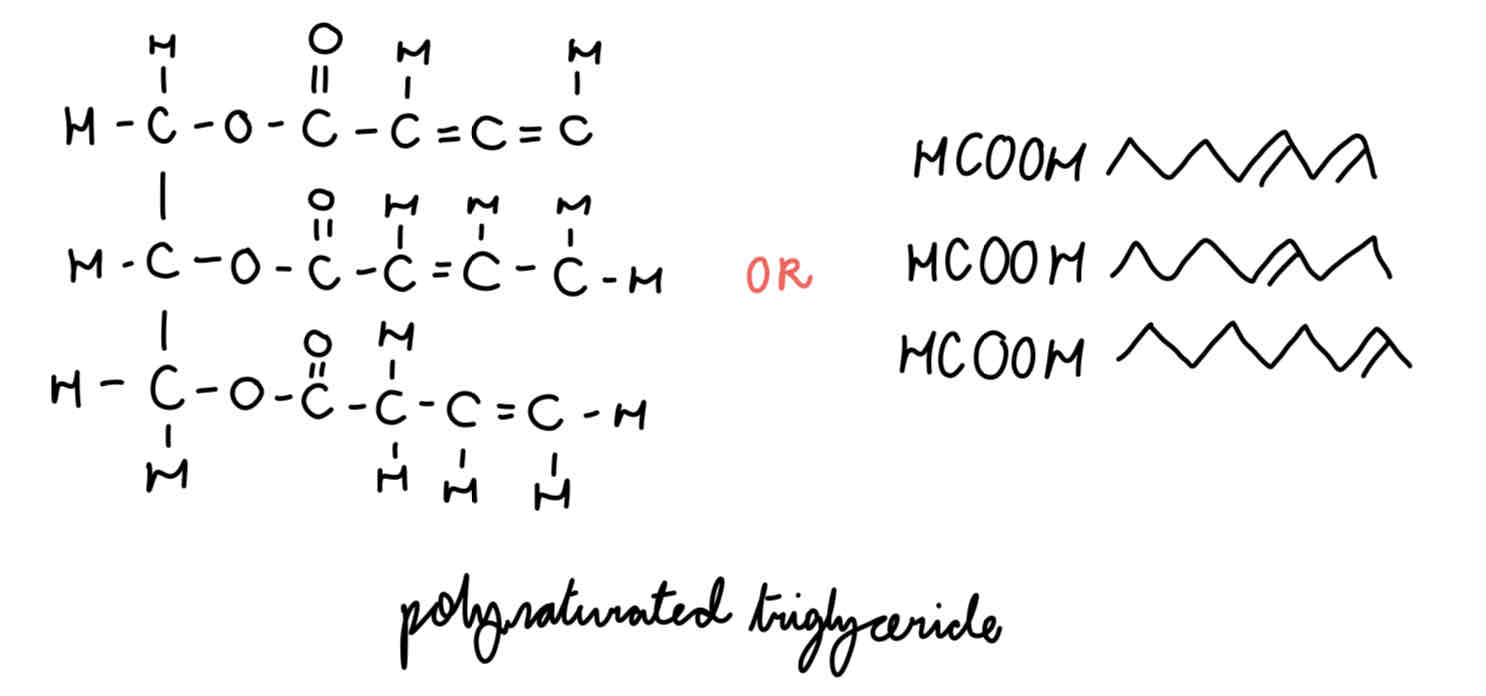
triglyceride condensation/hydrolysis
unsaturated phospholipid
polysaturated phospholipid
saturated fatty acid
unsaturated fatty acid
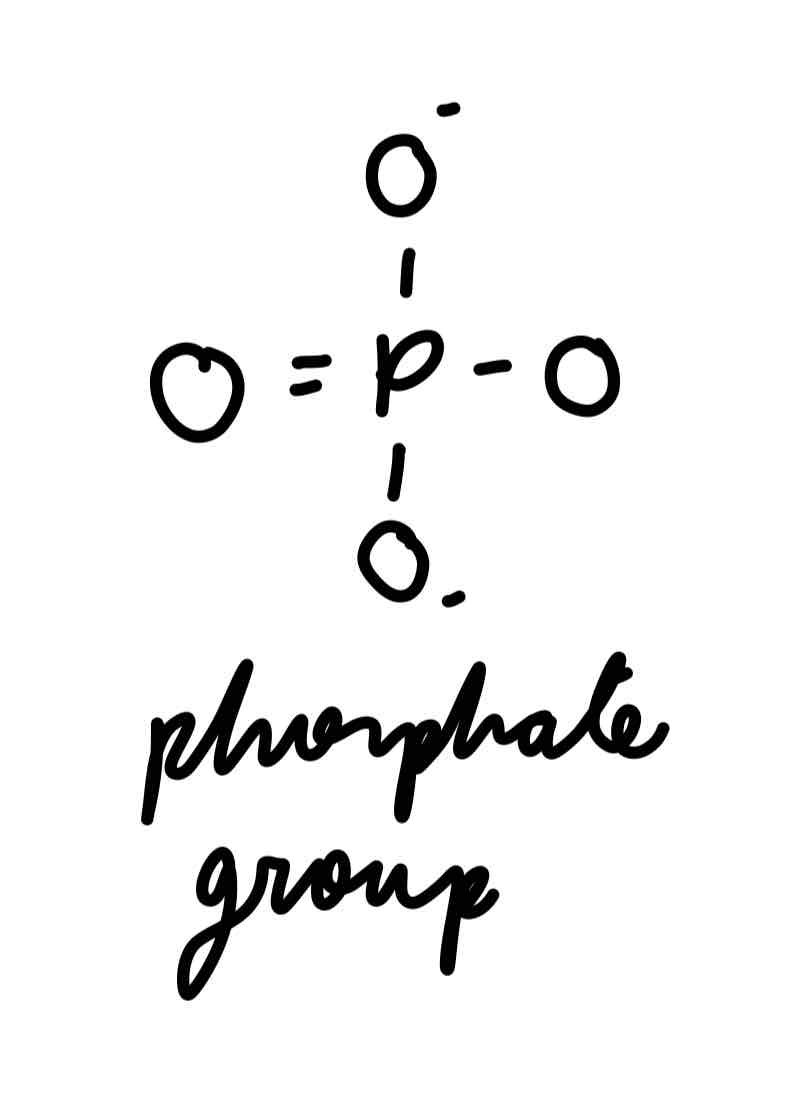
phosphate group
glycerol
amino acid (general structure)
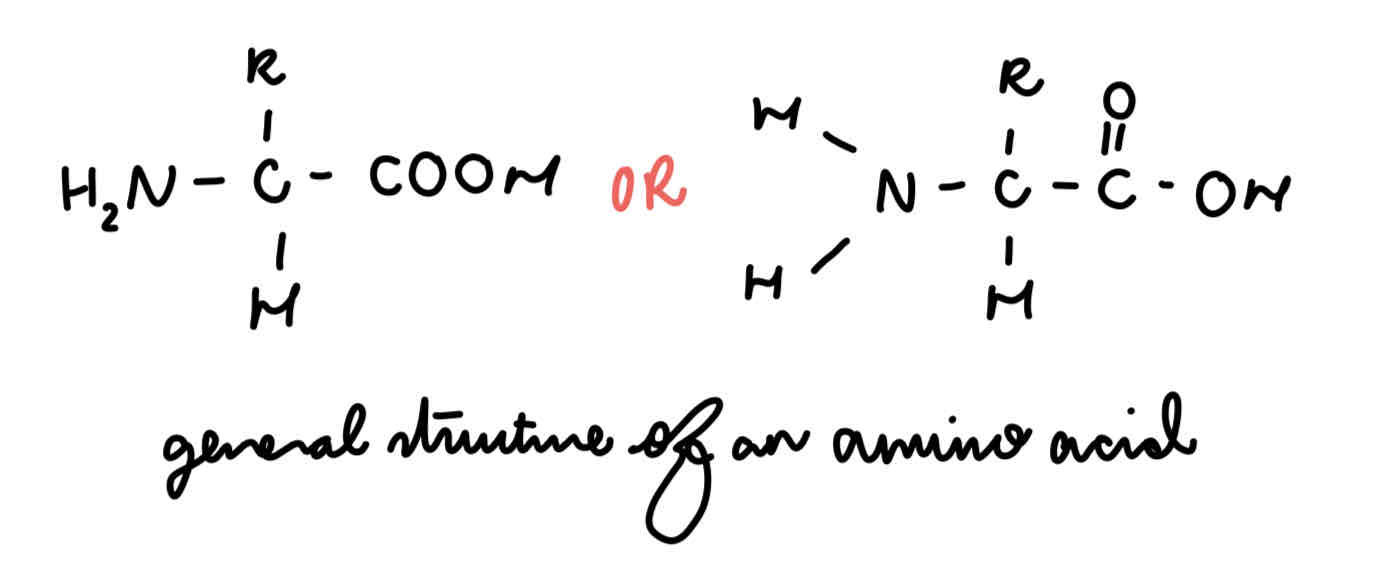
amine group
carboxyl group
dipeptide
polypeptide
give the equation for aerobic respiration (not including carrier molecules):
C6H12O6 + 6O2 → 6CO2 + 6H2O
give the equation for aerobic respiration (including carrier molecules):
C6H12O6 + 6O2 + 38ADP + 38Pi → 6CO2 + 6H2O + 38ATP
what is ATP?
adenosine triphosphate - energetic molecule w/ a high energy, unstable phosphoanhydride bond in which energy is released through respriation
energy from condensation reaction between ADP and Pi comes from breaking down glucose in respiration
phosphoanhydride bond broken through hydrolysis → Pi + ADP
why is ATP a good/bad energetic molecule?
releases relatively small amount of E
releases E instantaneously
phosphorylates other compounds
can be rapidly resynthesised
is not lost from/does not leave cells
BUT:
can’t be stored in last quantities as lowers water potential due to its -ve charges
this means it will attract water through osmosis, causing cells to lyse
a cell must have lots of mitochondria to release lots of ATP (as only a small amount can be released each time)

what is phosphorylation?
transferring/attaching Pi to target molecule, activating/energising target molecule and inducing a conformational change
what is dephosphorylation?
removing the Pi from the target molecule, causing the target molecule to become inactive
give some examples where phosphorylation may occur:
active transport pumps
sliding proteins in muscle cells (allowing muscles to contract
cytoskeleton proteins needed to move vesicles in cells
enzymes (e.g. protein synthesis)
making compounds more reactive (e.g. glucose is phosphorylated in first stage of respiration to synthesise starch)
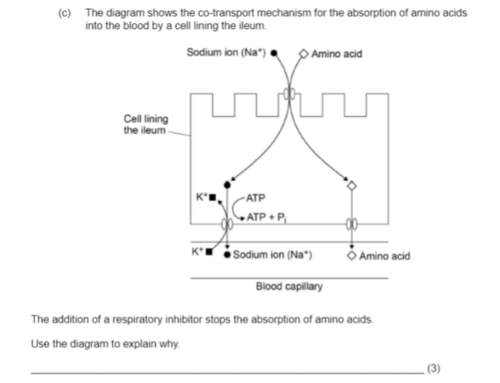
the addition of a respiratory inhibitor stops the absorption of amino acids - use the diagram to explain why (3)
no/less ATP produced → no/less active transport
Na+ not moved out of cell
no conc grad for Na+ to move into cell w/ amino acid
describe how an ATP molecule is formed from its component molecules (4)

what is DNA a polymer of?
nucleotides
what are the main functions of the genetic information in DNA?
acts as a info carrying molecule of inheritance
used to make proteins and functional RNAs
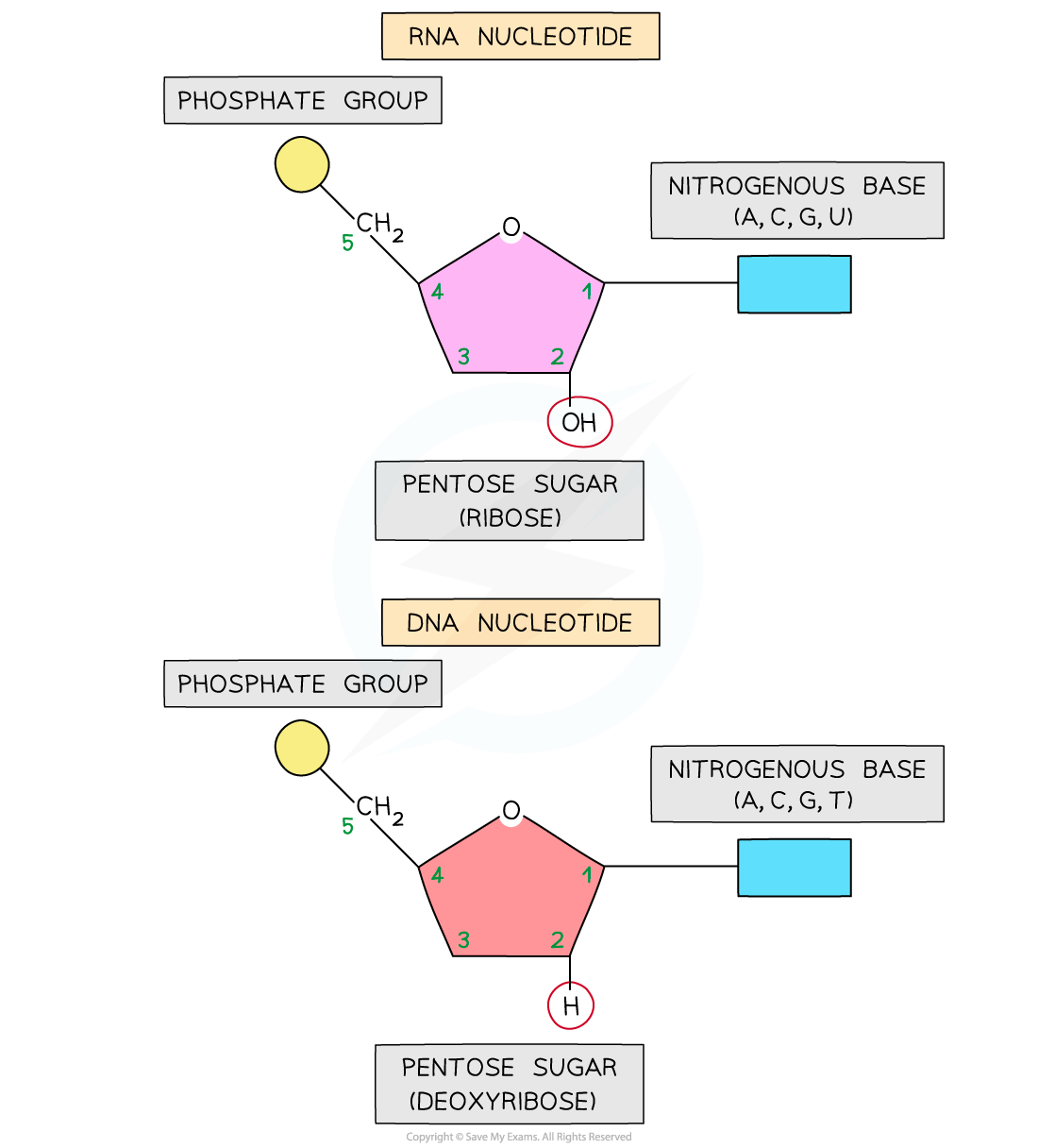
what is a nucleotide made up of?
(don’t need to be able to draw!)
deoxyribose
phosphate group
nitrogenous organic base
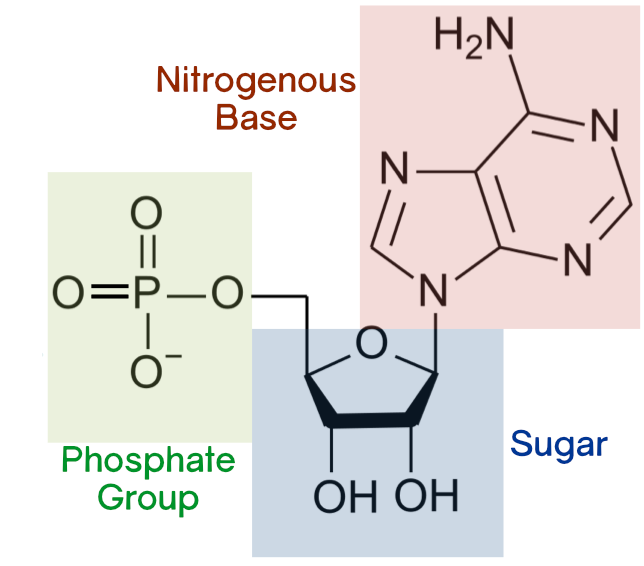
how do the 3 components of a nucleotide join together?
via condensation reaction
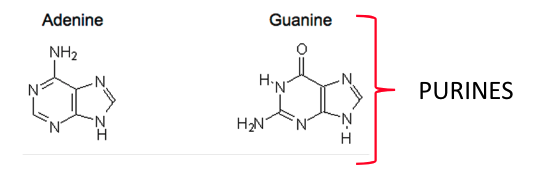
name the 4 nitrogenous organic bases present in DNA:
adenine
thymine
guanine
cytosine
what do adenine and guanine have in common (in terms of their structure)?
both purines
(this is why adenine only bonds to guanine)
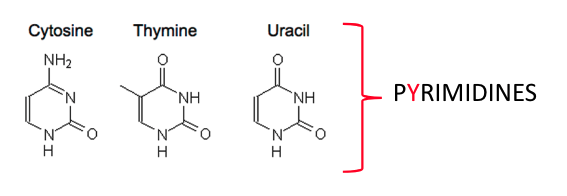
what do cytosine, thymine and uracil have in common (in terms of their structure)?
all pyramidines
how are the strands arranged in a dsDNA?
anti parallel
what is the bond between nucleotides called? how is it formed?
phosphodiester bond
condensation reaction
which bonds join DNA strands together? why is this significant?
H bonds
strong - lots of H bonds together are strong (although they are individually weak)
state Chargraff’s Rule
complementary base pairing rule - in a sample, amount of adenine = amount of thymine found in a sample and the amount of cytosine is always = to amount of guanine found in a sample
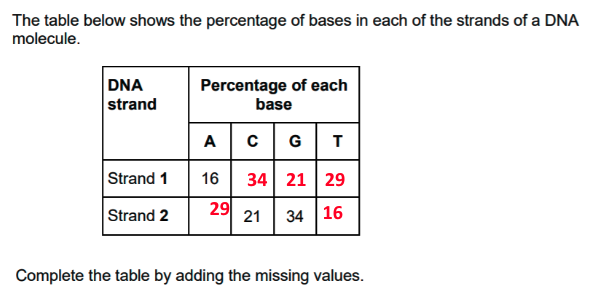
why did many scientists initially doubt that DNA carried genetic code?
its structure was relatively simple
what is RNA?
relatively short polynucleotide chain
what are the components of RNA?
ribose sugar, Pi group and organic nitrogenous base of adenine/uracil/guanine/cytosine
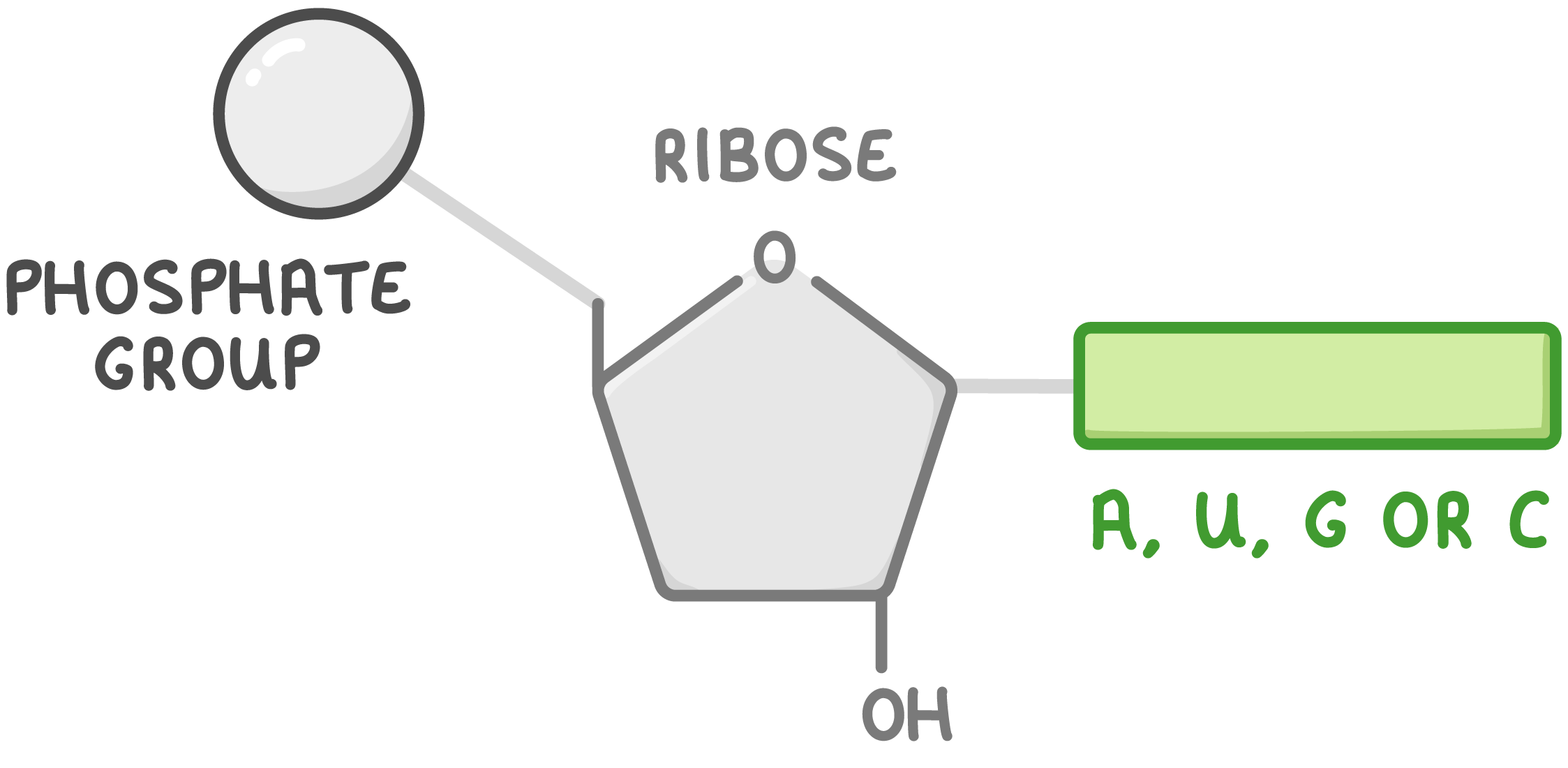
what is the overall function of RNA?
transfers genetic info from DNA to ribosomes
compare the structure of DNA to the structure of RNA:
DNA has the base thymine, RNA has the base uracil
DNA has a deoxyribose sugar, DNA has a ribose sugar
DNA is double stranded, RNA is single stranded
describe the structure of DNA and the structure of a chromosome (6)
polymer of nucleotides
nucleotide consists of deoxyribose, phosphate and organic/nitrogenous base
phosphodiester bonds between nucleotides
DNA double helix held by H bonds
complementary base pairing and H bonds between adenine, thymine pairs and cytosine, guamine pairs
DNA is associated w/ histone proteins
during mitosis (when visible) - chromosome consists of 2 chromatids joined at a centromere
name the two groups of lipids:
phospholipids
triglycerides
what is the main function of phospholipids?
make up cell (plasma) membrane
what is the main function of triglycerides?
act as fats and oils
what are the main features of lipids:
insoluble in water
soluble in organic solvents
contain C, H and O, but proportions are less than in carbs
what are the main roles of lipids?
energy source
waterproofing
insulation
waxy cuticle
protecting
how do lipids act as an energy source?
lipids provide more than 2x the energy as the same mass of carbohydrates
how do lipids act as a means of waterproofing?
insoluble in water
how does the lipid waxy cuticle aid a plant/insect?
helps to conserve water
how do lipids act as a means of insulation?
lipids are slow conductors of heat
so can help to retain body heat
how do lipids act as protection?
stored around delicate organs (e.g. kidneys)
how are triglycerides formed?
condensation of one molecule of glycerol and three molecules of fatty acid
ester bond and molecule of water formed (for every condensation reaction that occurs)
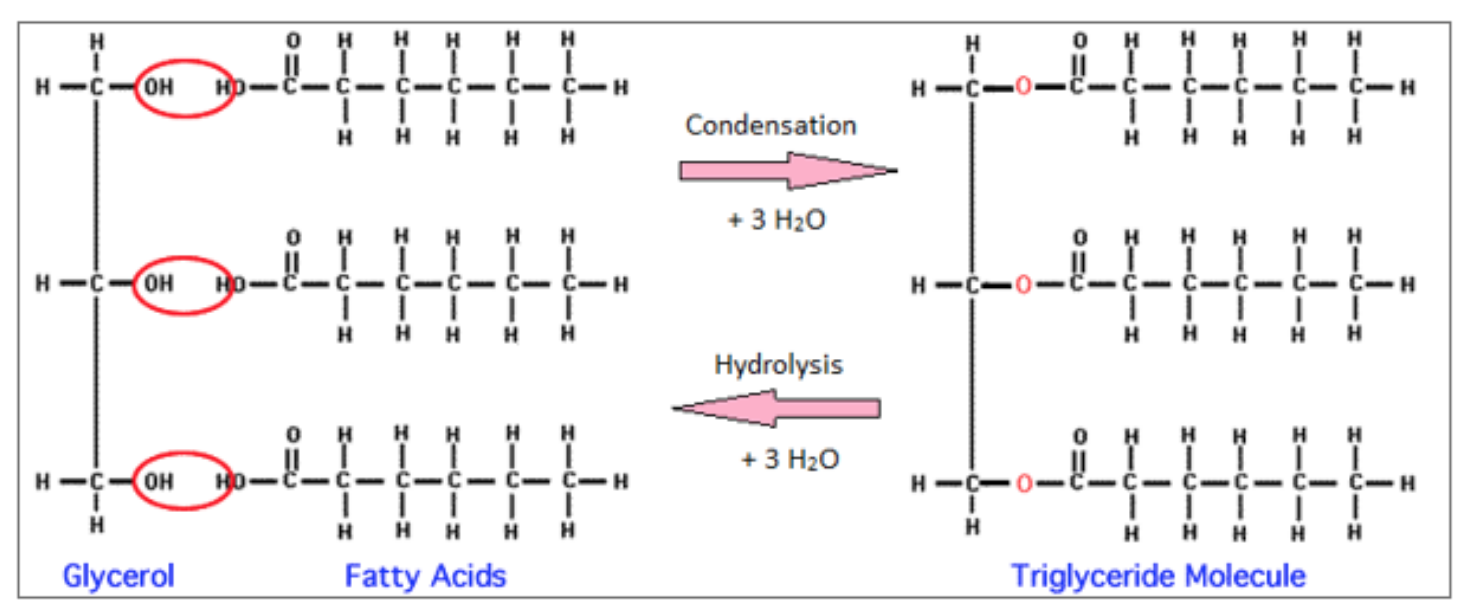
what are esters? why are they significant?
organic compounds formed by the reaction of an alcohol w/ a carboxylic acid
all have thefunctional group -COO
all lipids are esters
what are the two main properties of esters?
volatile
distinct fruity smell
what is glycerol?
an alcohol
C3H8O3
what is a fatty acid?
all have a carboxyl group (-COOH)
can vary in length and saturation/double bonds
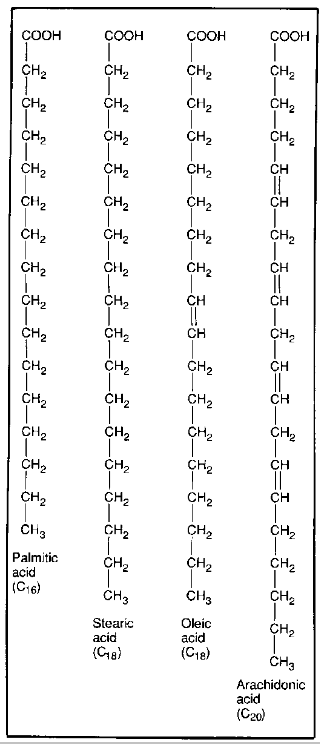
what is a saturated fatty acid?
have no double bonds (C-C)
so no of H bonds on fatty acid tail is maximised/saturated
higher melting point → solid at room temp
what is an unsaturated fatty acid?
have no double bonds (C=C)
so no of H bonds on fatty acid tail is not maximised/saturated
lower melting point → liquid at room temp
what are the main properties of triglycerides?
high ratio of C-H to C atoms
low mass to energy ratio
large, non polar molecules
why do triglycerides have a high ratio of C-H to C atoms?
so lots can be stored in a small volume
reduces mass that needs to be carried as they move
why do triglycerides have a low mass to energy ratio?
excellent store of energy
why are triglycerides large, non polar molecules?
so as not to affect water potential
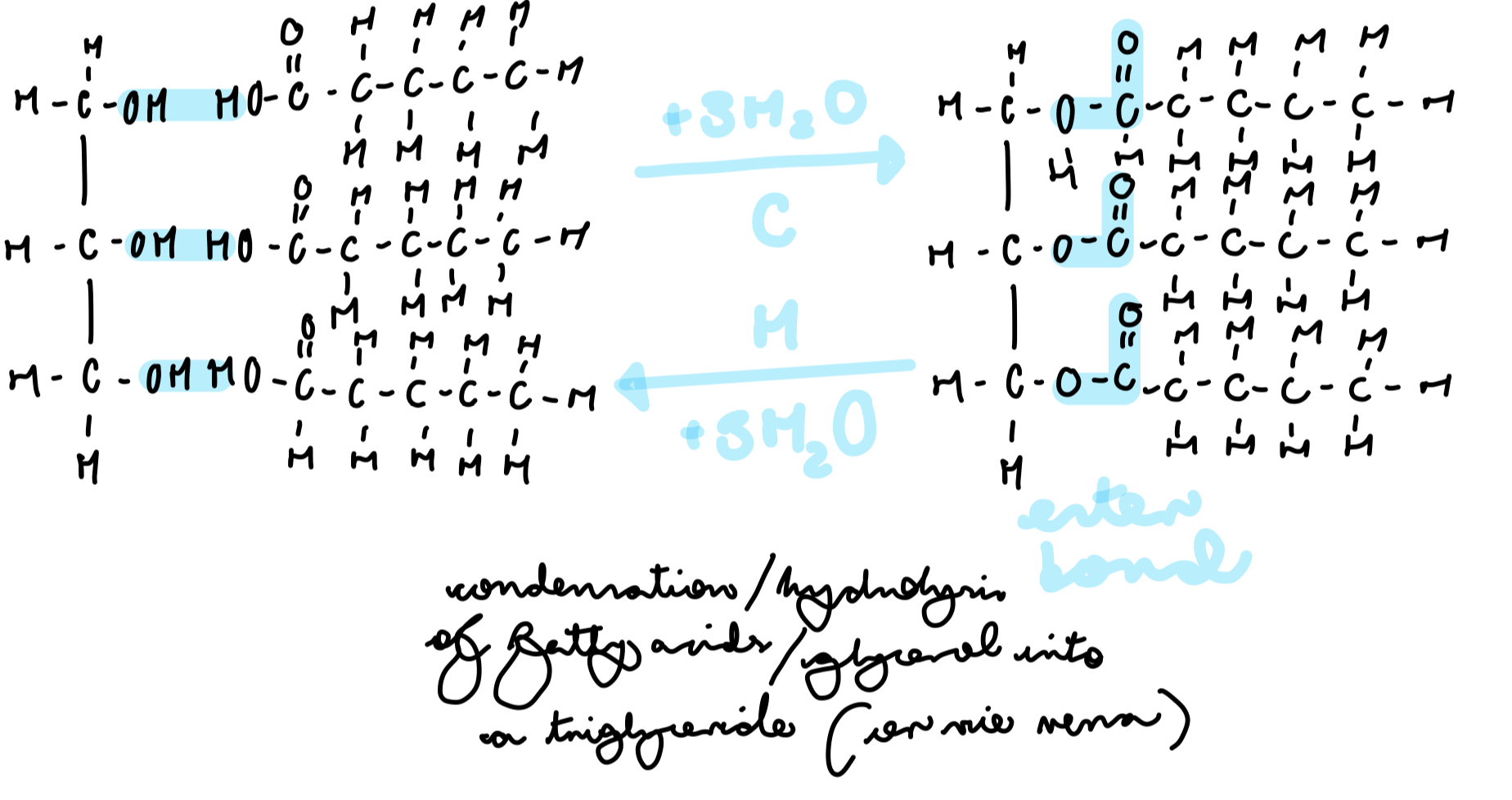
how are phospholipids formed?
condensation of one molecule of glycerol, two molecules of fatty acids and one phosphate group
ester bond and molecule of water formed (for every condensation reaction that occurs)
what are the polarities and affiliation to water of the different parts of a phospholipid?
hydrophilic, partially negative head
hydrophobic, partially positive tails
what is the general formula of a carbohydrate?
(CH2O)n