Retail and Ermergancy Supplies - exemptions
1/17
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
18 Terms
5 exemptions to restrictions on sale, supply and administration
Sale of GSLs without supervision of a pharmacist e.g in supermarket
Patient Specific Direction
Patient Group Direction
Optometrist/Podiatrist or podiatrist signed patient orders
Supply of medicines to school - signed order
PSD
written instruction from a doctor, dentist or non medical prescriber for a medicine to be supplied or administered to a named patient after having been assessed on an individual basis
Organisations can choose to limit who is authorised to supply / administer medicines under a PSD
PSD must contain enough info for person to safely administer the medicines - verbal,written
PGD
written direction that allows the supply/administration of a specified medicine by named authorised health professionals to a well defined group of patients requiring treatment for a pacific condition
E.g baby immunisation/ seasonal vaccines or oral contraception
Pharamcy Frst supply of nitrofurantoin or uncomplicated UTI
What limitations are there to the administration and supply of medicine under PGD and PSD ?
Administration : P or GSL medicines can be administered without PGD or PSD
Supply: P medicines require PGD or PSD unless an exemption applies or supply is made from a registered pharmacy under supervision of a responsible pharmacist - pre packed GSL medicines can be supplied without a PGD
Optometrist or Podiatrist signed patient orders
Optometrists and podiatrist cannot authorise supplies of POMs by writing prescriptions unless they are additionally qualified as independent or supplementary prescribers
Pharmacist working in a registered pharmacy can supply certain POMs directly to patients in accordance with a signed patient order from any registered podiatrist or optometrist
Medicine must be from a list of medicines that can be legally sold by the op/pod rather than just one they can administer
E.g chloramphenicol 0.5% eye drops for conjunctivitis or miconazole for athletes foot
Supply of Medicines to schools - signed order
adrenaline/salbutamol from a pharmacy via a signed order
Emergency supply
Signed order must contain : school name, product details, strength, purpose, total quantity, signature of head teacher, letter headed paper
Must retain SO for 2 years from the date of supply
The exemption of Naloxone
Naloxone - an opioid/opiate antagonist which can completely or partially reverse the CNS depression - used in the treatment of suspected opioid overdose
Individuals employed or engaged in lawful, recognised drug treatment centres can obtain Naloxone from a wholesaler and make direct supplies to patients without the need of a prescription, PGD or PSD
Wholesaling exemption
anyone trading medicines other than to a patient must :
have a wholesale license
Comply with Good Distribution Practice Standards and pass inspections
Have a suitable RP to ensure suitable procurement, storage and distribution of medicines
Serious Shortage Protocol (SSP)
2019 amendment to the Human Medicines regulations 2012 to introduce SSPs
Sale or supply by or under the supervision of a pharmacist in accordance with a SSP - in the event of a shortage of a POM
Allows different strengths, quantities or form of POM to that prescribed
Different brand or POM
Supervising Pharmacist must use professional judgement and skill to determine that the substitution is appropriate
Patient also needs to agree to the switch
Emergancy Supply at the Request of a Prescriber
To supply POM in the absence of a Prescription - in an Emergancy at the request of a patient or prescriber
case by case basis - professional judgment - best interest of the patient
Criteria :
Written prescription cannot be provided immediately due to an Emergancy event
Prescription must be provided however within 72 hours after request
Medicine must be supplied in accordance with directions given by the prescriber
NO CDs except Phenobarbital
NO. Sch1,2,3 CDs
Emergancy Supply at request of prescriber Records must contain …
date POM supplied
Name of medicine inc strength and form and quantity
Name and address of prescriber requesting medicine
Name and address of patient
Date of prescription
Date of when prescription was received
Emergency Supply at request of Patient
case by case basis
Professional Judgement
Criteria :
interview patient
Must demonstrate immediate need for POM and evident that prescription is not practical to obtain
previous treatment evidence - UK, EEA or Swiss health professional
Dose - summary care records
Max length of Emergency Supply at request of patient
Sch 4 or 5 CDs : max 5 days
POM : Max 30 days
Insulin, ointment, cream, inhaler - smallest pack possible
Oral contraceptive - full treatment cycle
Antibiotic - smallest quantity or full treatment
Records - Emergency supply at Patient’s request
made in POM register on the day of supply
Must include :
date of POM supply
Name and quantity of medicine supplied
Name and address of the patient
Info on the nature of the emergency
Emergency supply label must be added to dispensing label
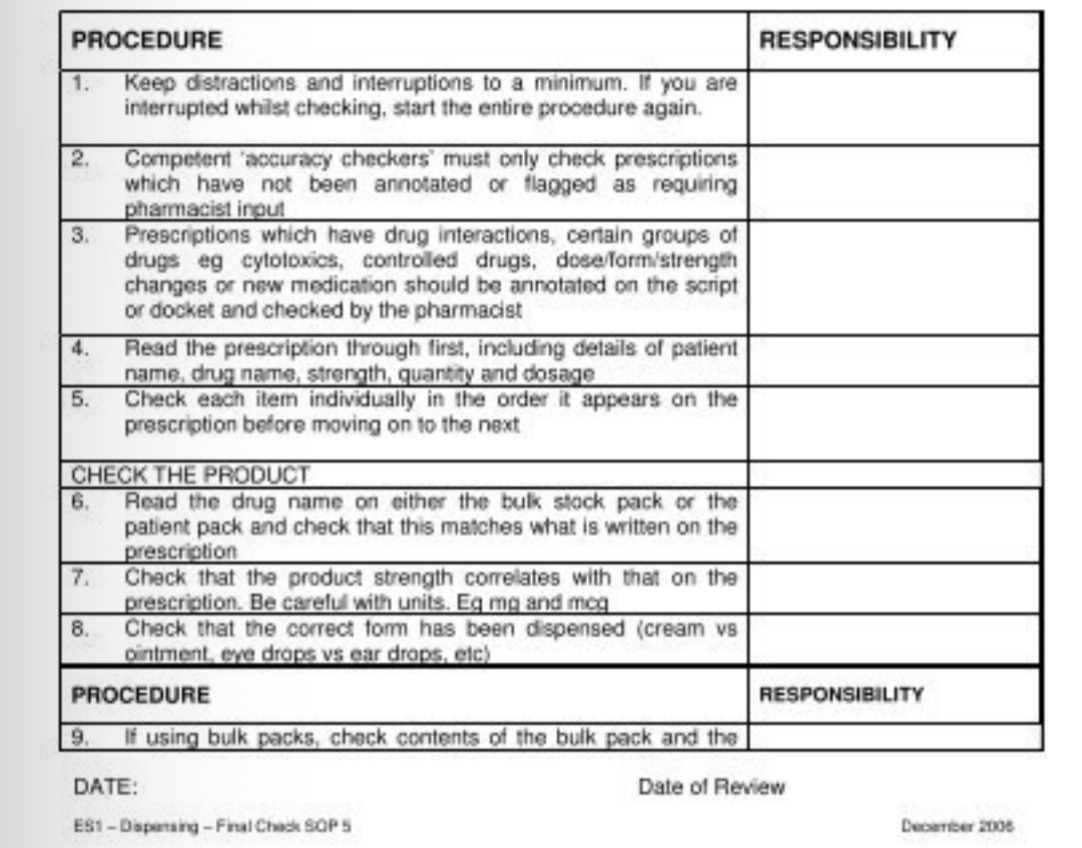
Standard Operating procedures - SOPs
written instructions on how to carry out a routine task - when, where and by whom
Clinical governance - management of risk and harm minimisation
Benefits of SOPs
Standardisation of processes and practice towards a specific outcome
Maintains consistency
Improve quality assurance and safety
Saves time/training needs/cognitive workload
RPS Error reporting Guidlines
Take steps to let the patient know promptly
Make things right - contacting prescriber
Offer an apology
Let colleagues involved in the error know

Criteria for justified legal defence for honest dispensing error
Dispensed in registered pharmacy
Dispensed under supervision of registered pharmacist
Supplied against prescription, PGD or direction of prescriber requesting medicine
Promptly notified to patient once pharmacy team are aware of error