AP BIO STUDY
1/45
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
46 Terms
What are the four Macromolueculs?
Proteins, carbohydrates, lipids, nucleic acids
What are Carbohydrate composed of? Ratio?
1 Carbon, 2 hydrogen, and 1 oxygen
Monomer for Carbohydrate? Examples?
Monosaccharides: simple sugars such as glucose and fructose that serve as the building blocks for carbohydrates.
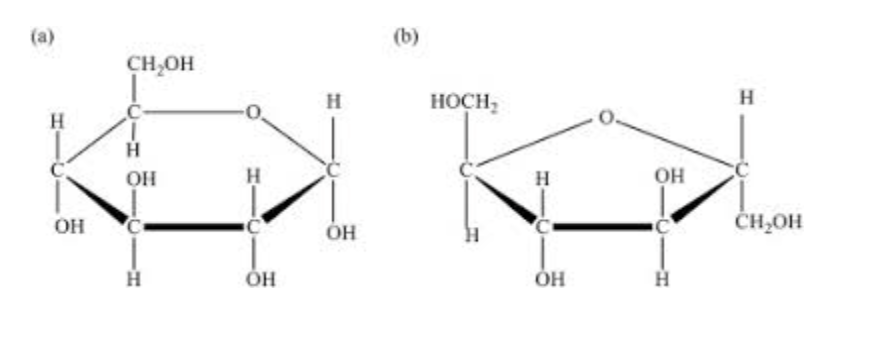
What are two monosaccharides together called? Exp?
Disaccharides; examples include sucrose and lactose.
What is the bond in a monosaccharide connects the sugar monomers in a carbohydrate called?
Glycosidic bond
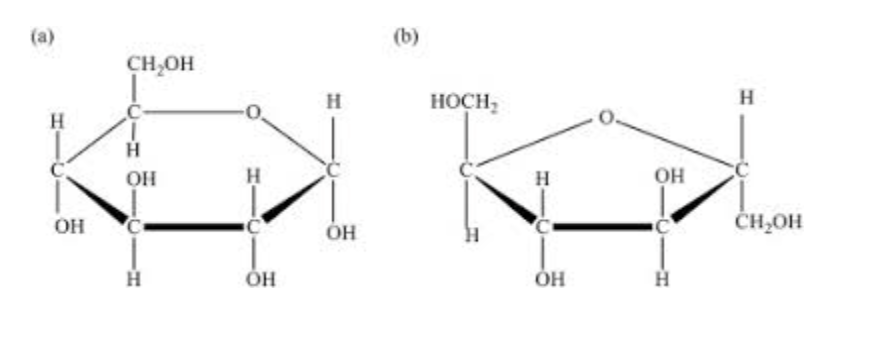
What is this?
Monosaccharides: simple sugars such as glucose and fructose that serve as the building blocks for carbohydrates.
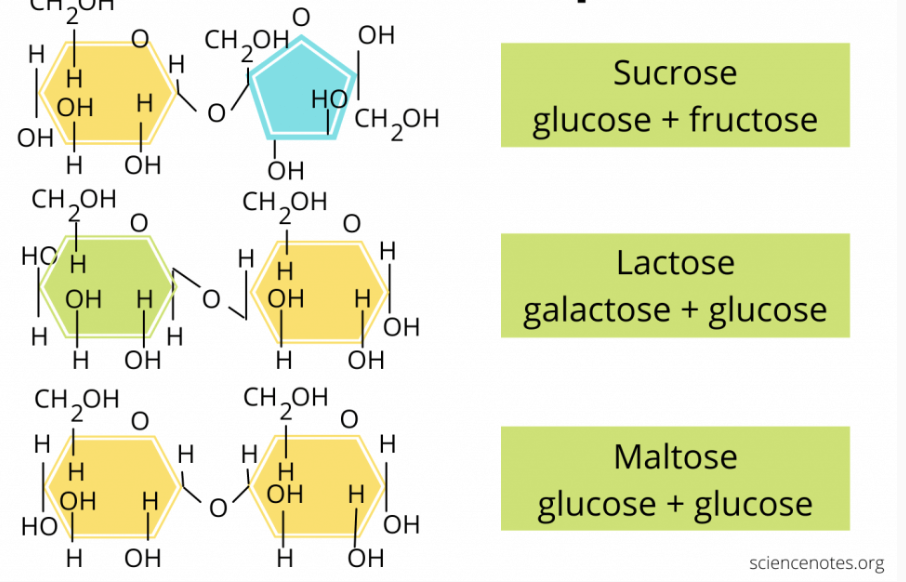
What is this?
disaccharide; the sugar that forms when two monosaccharides or simple sugars join via a glycosidic bond.
What are structural carbohydrates and where they are found?
Polysaccharides
Cellulose: found in plant cell wall for support
Chitin: found in fungi cell walls and exoskeletons of arthropods
What are storage Carbohydrates and where are they found?
Polysaccharides
Starch: stored in plants for energy reserve.
Glycogen: stored in animals in liver and muscles.
Difference between starch and cellulose? diffrent linkage?
Starch, storage : Alpha linkage bond → can break down, digestible
Cellulose, structural : Beta linkage → Can not break down, not digestible
What are proteins composed of?
Carbon, Hydrogen, oxygen, nitrogen, and sulfer
What is the monomer of proteins? What are they made up of what and are they acid/basic and are they polar/non polar?
Amino Acid → made of carboxyl group (acidic, polar), amino group (basic, polar), and a side chain (R group), can differe
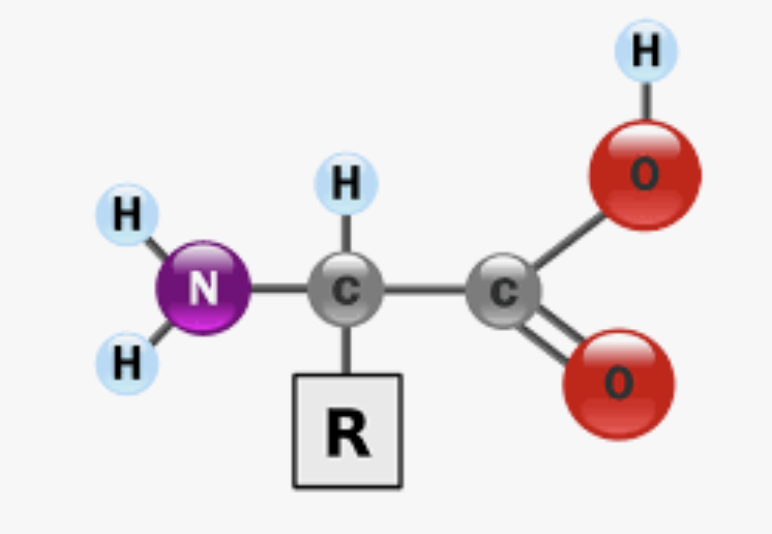
What is the bond between the carboxyl of ONE amino and amino group of ANOTHER amino acid (in a protein)? What type of bond?
Peptide bond, it is covalent so it shares valence electrons
How many different amino acids are there, and what differs on them?
20, the R group
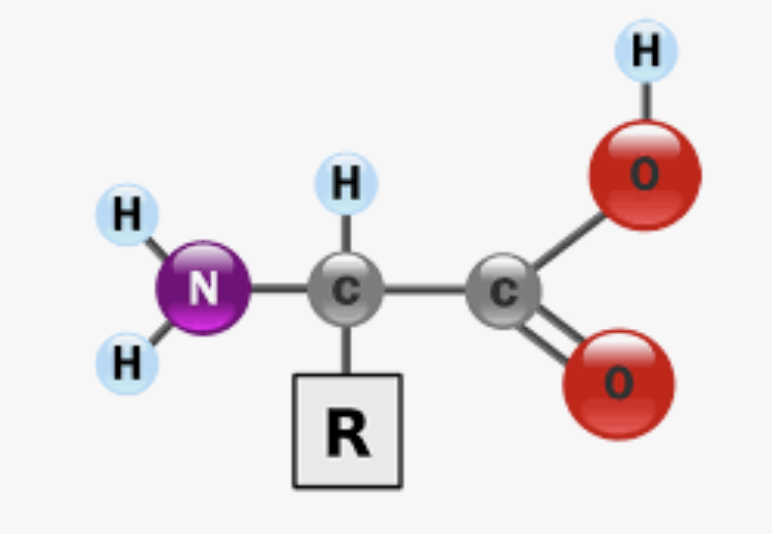
What is the directionality of a protein?
the amino N-terminal to the carboxyl C-terminal end
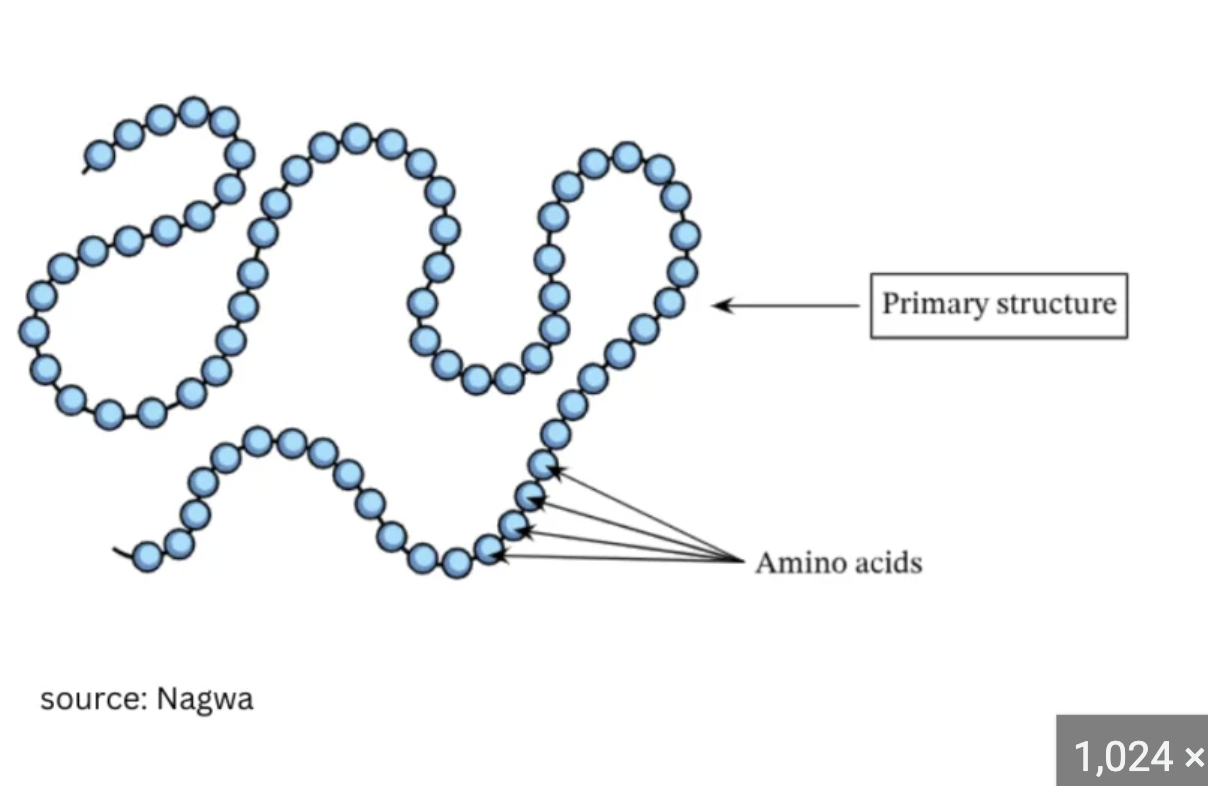
First level of Protein structor? Bond? Structure?
Primary structure
Bond: peptide bonds between amino acids
Structure: string of amino acids
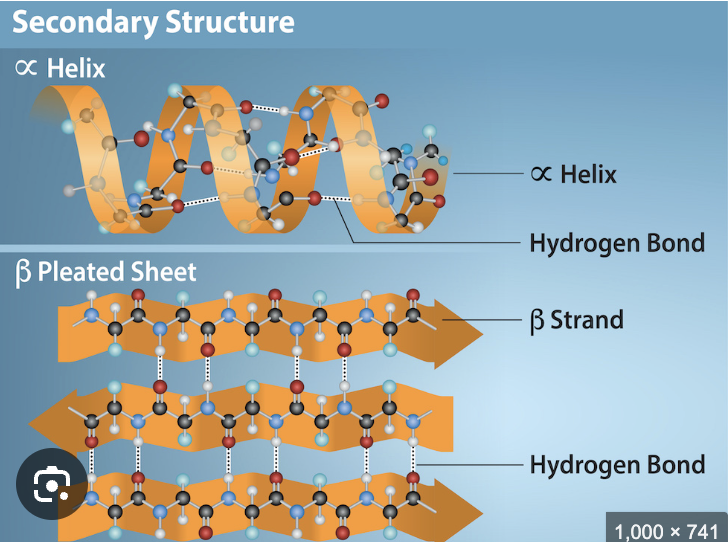
Second level of Protein structor? Bond? Structure?
Term
Secondary structure
Bond: hydrogen bonds between backbone atoms
Structure: alpha helices and beta sheets
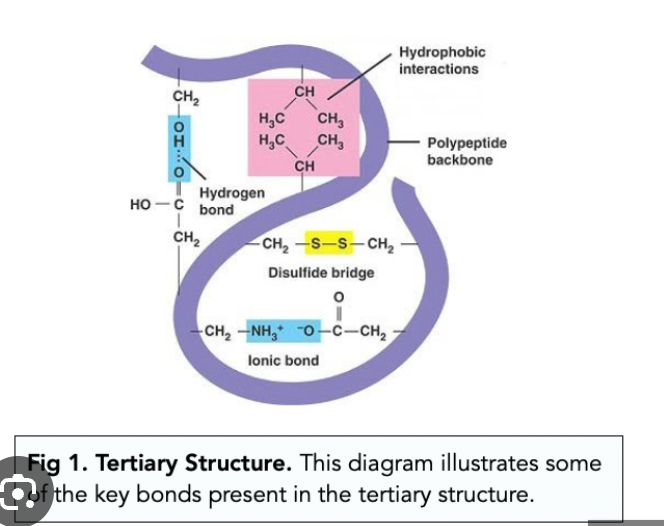
Third level of Protein structor? Bond? Structure?
Term
Tertiary structure
Bond: various interactions including hydrogen bonds, ionic bonds, hydrophobic interactions, and disulfide bonds
Structure: three-dimensional folding of a protein
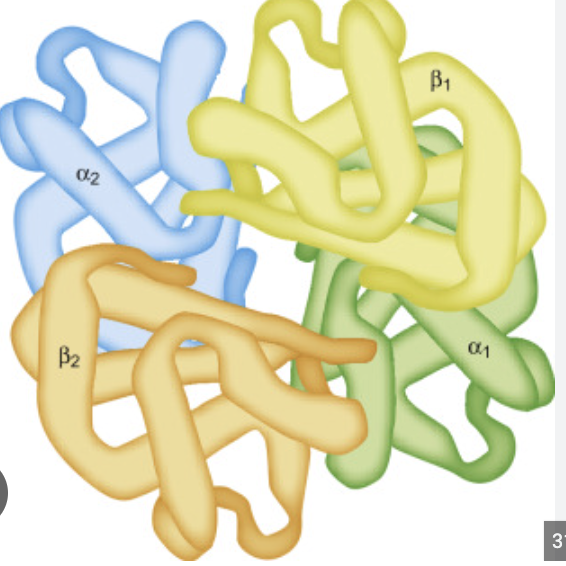
Forth level of Protein structor? Bond? Structure?
Quaternary structure
Bond: interactions between multiple polypeptide chains
Structure: arrangement of multiple protein subunits
What are the 3 R groups? Polarity?
Hydrophobic (nonpolar), hydrophilic (polar) , and Charged (pos or neg)
How do hydrophilic R groups make the protein fold?
into the aqueous environment of a cell, EXTERIOR
→ interacting with water molecules and forming hydrogen bonds.
How do Hydrophobic R groups make the protein fold?
into the INTERIOR of the protein, away from aqueous environments, stabilizing the structure.
How do charged R groups make the protein fold?
exterior of the protein, interacting with water or other charged molecules, contributing to overall stability
What is a nucleic Acid composed of?
Carbon, hydrogen, oxygen, nitrogen, and phosphorus atoms.
What is the monomer of Nucleic Acid? What does it consiste of?
Nucleotide: that consists of a sugar, phosphate group, and nitrogenous base.
What is the bond a Nucleotide? What does it link and what does that form?
Phosphodiester linkage:
linking the phosphate group of one nucleotide to the sugar of another nucleotide
forming the backbone of the nucleic acid chain.
What makes something a 3 prime end?
The presence of a free hydroxyl (-OH) group attached to the 3' carbon of the sugar molecule in a nucleotide.
Where are hydrogen bonds in DNA acid?
Between complementary nitrogenous bases, stabilizing the double helix structure
What makes something a 5 prime end?
The presence of a free phosphate group attached to the 5' carbon of the sugar molecule in a nucleotide
What are you purine bases in DNA?
Nitrogenous bases that have a double-ring structure, specifically adenine and guanine, which pair with pyrimidines in DNA and RNA.
What are the pyrimidine bases in DNA?
Nitrogenous bases with a single-ring structure, specifically cytosine, thymine, and uracil. They pair with purines in DNA and RNA.
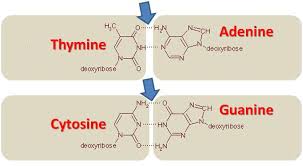
Cytosine goes with…
guanine in DNA
Thymine goes with…
adenine in DNA
Adenine goes with…
uracil in RNA and thymine in DNA.
adenine and thymine have ——— bonds
two hydrogen
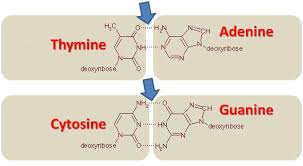
Cytosine and guanine have ——— bonds
three hydrogen
Deoxyribose + double stranded VS Ribose single stranded
Deoxyribose is the sugar found in DNA, which is double-stranded,
Ribose is the sugar in RNA, which is single-stranded
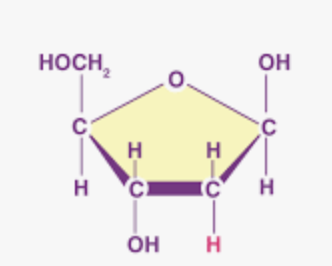
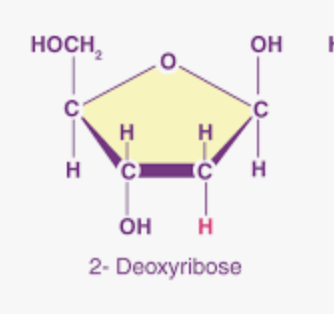
What is this?
Deoxyribose: pentose sugar that is a component of DNA.
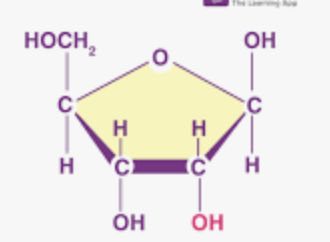
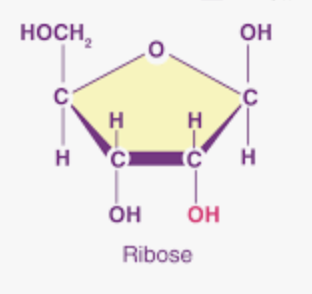
What is this?
Ribose: pentose sugar that is a component of RNA. Difference is oxygen at the 2' carbon
What are lipids are composed of?
Carbon, hydrogen, and oxygen molecules, with Phosphate in phospholipids.
So many carbon and hydrogen in lipids that they are
non-polar
What is a monomer? what is a polymer?
A monomer is a small, basic molecular unit that can join together to form a larger structure called a polymer.
Polymers are long chains of repeating monomer units, such as proteins, nucleic acids, and carbohydrates.
Lipids do no have ———— because they don’t have ————
true polymers, repeating monomer units
Carbohydrate’s monosaccharides make up (their polymer)→
polysaccharides like starch, glycogen, and cellulose.
Proteins monomer Amino acids make up (their polymer) →
polypeptides or proteins (trick question lowkey) that perform various functions in the body
Amino acids polypeptide in nucleotides and they come together to form →
nucleic acids like DNA and RNA.