Lesson 3.1 Brønsted–Lowry acids and bases
0.0(0)
Studied by 0 peopleCard Sorting
1/9
There's no tags or description
Looks like no tags are added yet.
Last updated 7:12 AM on 1/24/26
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
10 Terms
1
New cards
What is a Brønsted-Lowry acid?
A proton (H⁺) DONOR. E.g., HCl donates H⁺ to water: HCl + H₂O → H₃O⁺ + Cl⁻
2
New cards
What is a Brønsted-Lowry base?
A proton (H⁺) ACCEPTOR. E.g., NH₃ accepts H⁺ from water: NH₃ + H₂O ⇌ NH₄⁺ + OH⁻
3
New cards
Define monoprotic, diprotic, and polyprotic acids
Monoprotic: donates 1 H⁺ (e.g., HCl, HNO₃, CH₃COOH). Diprotic: donates 2 H⁺ (e.g., H₂SO₄). Triprotic/Polyprotic: donates 3+ H⁺ (e.g., H₃PO₄)
4
New cards
How does H₂SO₄ ionise in water (2 steps)?
Step 1: H₂SO₄ + H₂O → H₃O⁺ + HSO₄⁻ (strong, goes to completion). Step 2: HSO₄⁻ + H₂O ⇌ H₃O⁺ + SO₄²⁻ (weak, reversible)
5
New cards
What is an amphiprotic species?
A molecule or ion that can BOTH donate AND accept a proton — acts as acid or base depending on what it reacts with. Examples: H₂O, HCO₃⁻, HSO₄⁻, amino acids
6
New cards
Show how water acts as amphiprotic
With acid (base role): HCl + H₂O → H₃O⁺ + Cl⁻. With base (acid role): NH₃ + H₂O ⇌ NH₄⁺ + OH⁻
7
New cards
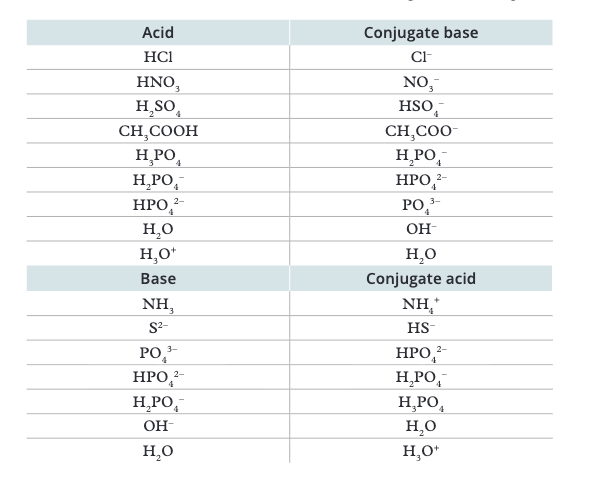
What is a conjugate acid-base pair?
Two species that differ by exactly ONE proton (H⁺). Conjugate base = acid minus H⁺. Conjugate acid = base plus H⁺
8
New cards
Identify conjugate pairs: HNO₃ + H₂O → H₃O⁺ + NO₃⁻
Pair 1: HNO₃ (acid) / NO₃⁻ (conjugate base). Pair 2: H₂O (base) / H₃O⁺ (conjugate acid)
9
New cards
How do you find the conjugate BASE of any acid?
Remove one H⁺ and decrease charge by 1. E.g., H₂SO₄ → HSO₄⁻, HSO₄⁻ → SO₄²⁻, NH₄⁺ → NH₃
10
New cards
How do you find the conjugate ACID of any base?
Add one H⁺ and increase charge by 1. E.g., NH₃ → NH₄⁺, OH⁻ → H₂O, CO₃²⁻ → HCO₃