Learning and Memory 2
1/28
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
29 Terms
Implicit memory
Patient HM who lacked the medial temporal lobe was able to do tasks with improvement → tracing a star while looking in a mirror
Some implicit learning tasks which are hippocampal-independent: motor skills, habituation, sensitisation
Types of learning:
Non-associative:
Habituation – if a stimulus persists, then our brain can get used to it, ignore it, tone down the response
Sensitisation – the opposite of habituation and a modest stimulus can be highlighted by context
Associative:
Classical/Pavlovian – associate an unconditioned stimulus to a conditioned stimulus (passive)
Operant conditioning – where the conditioning is less passive and when an animal is prompted to do so it can be trained to press a lever in order to obtain a reward (abstract)
Cognitive learning – applying abstract concepts
Aplysia californica gill withdrawal reflex – Non-associative learning
Habituation
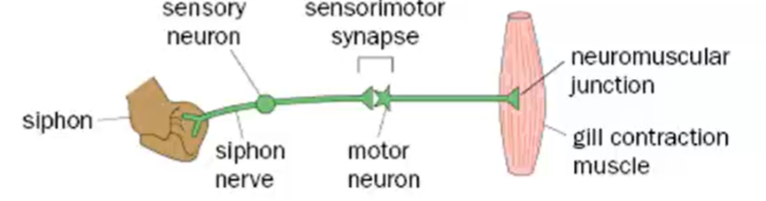
Paradigm that if you touch the siphon of the Aplysia it leads to the gill withdrawal reflex
Naive Aplysia will withdraw the gill but over time you may see habituation → the gill withdrawal response will diminish
This is due to the sensory input of touching the gill, causing a motor response (contraction of the gill)
Aplysia has large neurons which form simple circuits, making them the ideal organism to work with
Sensitisation
Can then sensitise the animal to the tactile stimulus by applying an electrical shock to the tail
A shock to the tail followed by the tactile touch of the siphon sensitises the animal causing an exaggerated gill withdrawal which negates the habituation
We know that the tail shock (sensed by sensory neuron) acts an a serotonergic interneuron which synapses onto both the cell body of the sensory neuron and the synaptic connection of the sensory neuron to the motor neuron
Serotonin from that interneuron results in the sensitisation of the circuit
Molecular processes underlying habituation
Experiment 1
Just stimulate the motor neuron and see if the repeated stimulation of the motor neuron results in the reduced gill withdrawal over time → it doesn’t
We now know that habituation needs to act upstream of the motor neuron
Experiment 2
We can now ask if input from the sensory neuron is required? We can block the sensory input and see that if the sensory input there’s no gill withdrawal, but if we unblock this then there's not much habituation
So the input of the sensory neurons is required for habituation
Experiment 3
Can overcome habituation artificially by direct stimulation of the serotonergic interneuron by their axon bundle
Same sensory neuron stimulation whose output is habituated, when neurological equivalent of a tail shock is applied (stimulation of the interneurons) then the response is sensitised
This shows us that it’s the synpase between the sensory and motor neuron that habituates and the serotonergic neuron can reverse the effects of habituation
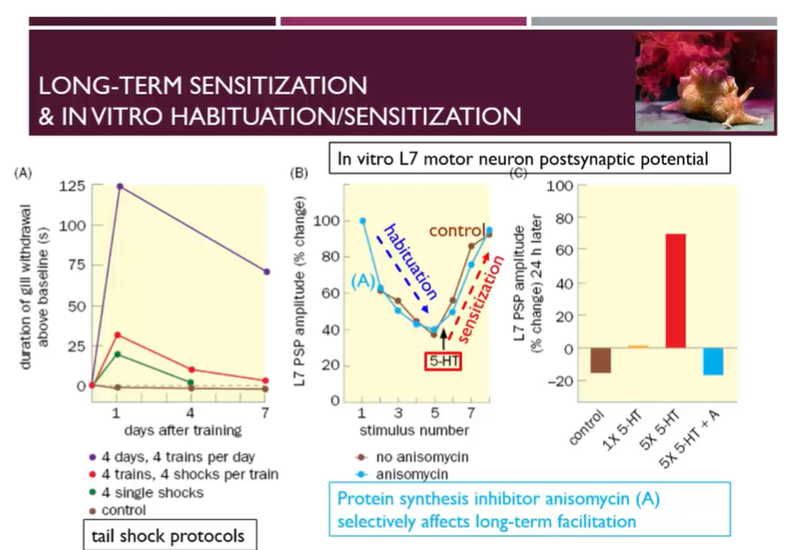
Long term sensitisation effects
Under control conditions, there’s a certain level of gill withdrawal
If we apply 4 single shocks to the tail per application, then check the gill withdrawal om day 1 and day 4
On day 1 there’s an increased gill withdrawal, which has been habituated by day 4
If instead of 4 single shocks, we apply shocks in trains and do 4 trains, we get an exaggerated sensitisation
With 4 days of training with 4 trains of 4 shocks per day then we have an even larger sensitisation which stays well above baseline at day 7
Just like short- and long-term memory, we have long and short-term sensitisation which is dependent on the amount of training
Evidencing the difference in quality between the long- and short-term increases
Different quality of the long and short-term increases
Anisomycin is a protein synthesis inhibitor which does not affect the process of sensitisation or habituation in the early stages
When the stimulus is applied, first we are able to habituate, brining the L7 motor neuron’s PSP down
then when serotonin is applied, then we are able to sensitise, brining the L7 motor neuron PSP amplitude back up
However, after 24 hours, the anisomycin decreases the amplitude of the L7 motor neuron back to the level of the control → showing that long term sensitisation is dependent on protein synthesis
CREB – the molecular basis of short and long-term facilitation
Molecular basis of habituation
Synapse of the sensory neuron to the motor neuron (one which is responsible for the habituation) is a glutamatergic synapse
When the sensory neuron is activated calcium influxes but over time the availability of the glutamate to release and the availability of calcium to influx is depleted (responsible for habituation)
Short term molecular basis of sensitisation
Serotonin binding to a GPCR causing the activation of adenylyl cyclase, producing cAMP
cAMP activates PKA which causes the phosphorylation of a potassium leak channel, raising the resting membrane potential in the neuron and making it more available for depolarisation → offsetting the habituation in the short term
useful for before proteins are able to be synthesised
Long term molecular basis of sensitisation
PKA phosphorylates CREB which drives the expression of proteins which stabilise the synapse and support increased number of outgrowths
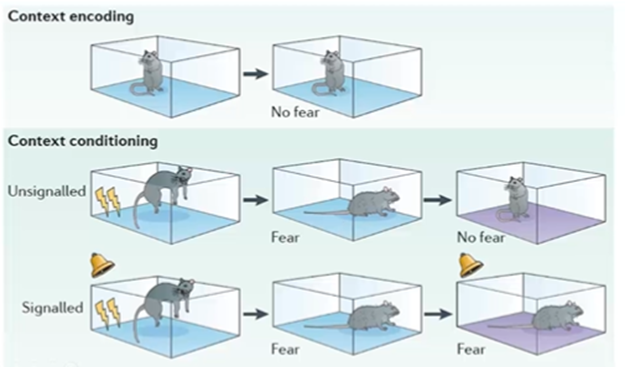
Associatiive learning - Classical conditioning and fear conditioning
Unsignalled conditioning
couple the electrical shock with the memory of a cage and there’s an association of the shock with the spatial context
Signalled conditioning
just like in Pavlovian conditioning, then we can couple the sound of a bell ringing with a shock so that when the animal is in a new cage, makes a non-associative spatial context fear inducing when the sound is heard
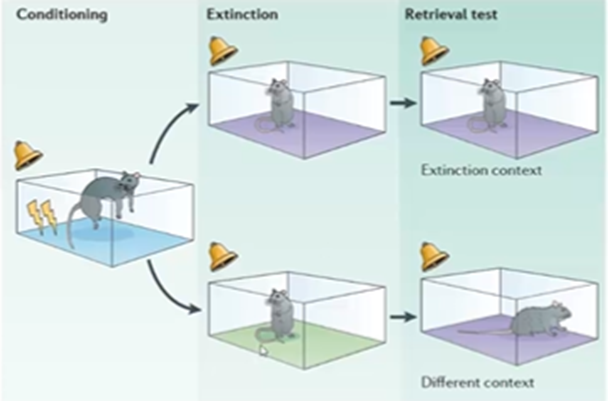
Dishabituation
separate this association by giving the rat a tone repeatedly in a cage where there was no shock and over time the tone becomes less of a signal for fear. If more than one type of stimuli are conditioned (E.g: a cage and a sound) then you would have to decondition these stimuli separately
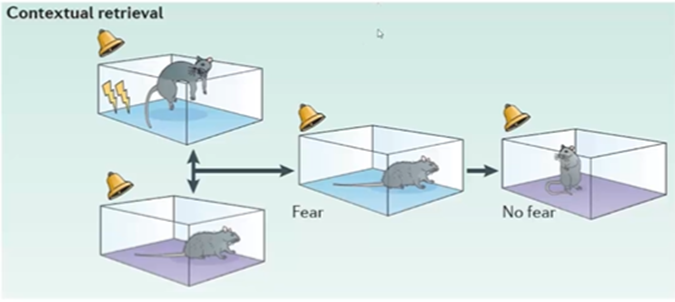
Context dependent extinction of fear conditioning:
when we extinguish a conditioned response in a particular environment, but it is only extinguished in that particular environment and if the rodent were to be placed in a different environment (not the original one) and hear the noise, then it would still exhibit freezing behaviour
Three parameters feeding into the conditioning: unconditioned foot shock, conditioned tone and also the conditioned room, and extinction can be room specific
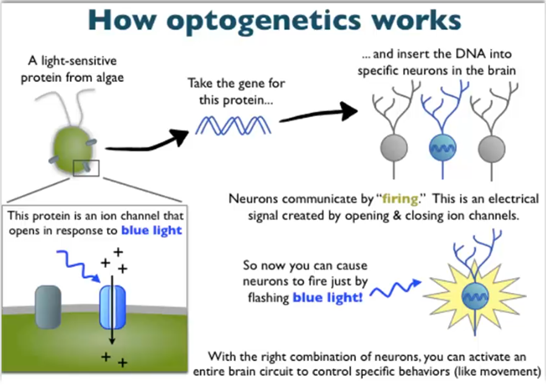
Optogenetics
Using light to manipulate neuronal activity to activate or silence neurons
Can be done easily in living animals and is easier than electrophysiology
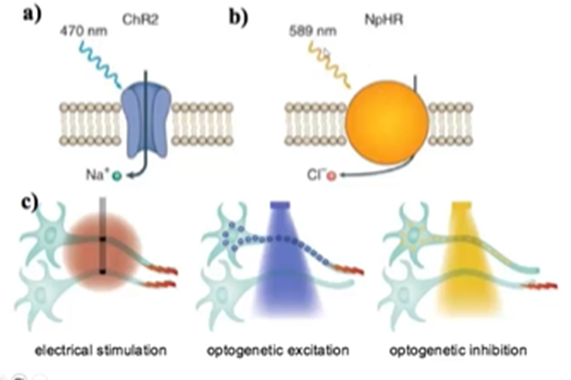
Channel rhodopsin is a blue light-activated sodium channel, halorhodopsin is a chloride channel activated in response to light
Can be quite selective in our gene expression and only those neurons which express the channes will respond as opposed to electrophysiology, where all of the neurons which touch the electrode will respond
Combining fear conditioning with optogenetics:
Take a mouse and in the hippocampus inject the virus which infects the neurons and allows channel rhodopsin expression in a controlled manner
Can express the channel rhodopsin in the hippocampus which can take the place of the tone → stimulate the subset of hippocampal neurons with blue light to a foot shock and so if the blue light signal is present on its own it results in freezing
Now we can know which neurons are responsible, as they will be marked by channel rhodopsin expression
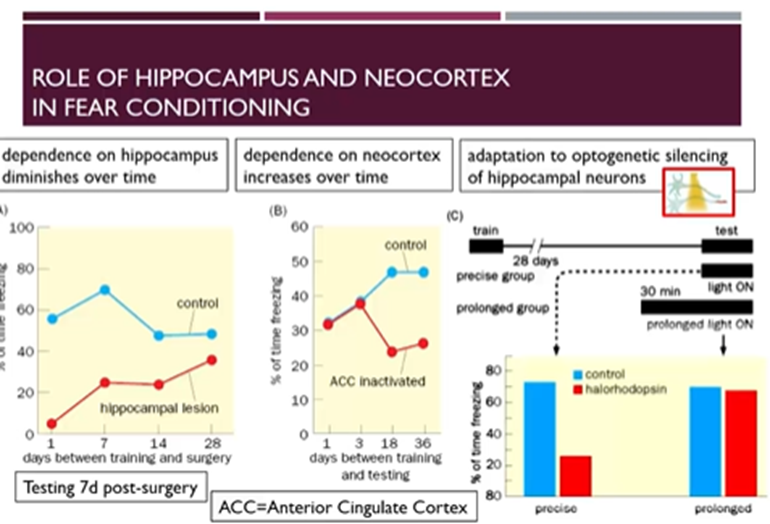
Over time memories are more dependent on the cortex and less dependent on the hippocampus
Hippocampus lesioned and 7 days after the surgery fear conditioned tests were conducted
if the previous training where the animal was conditioned to the foot shock happened a long time prior to the surgery, then fear conditioning was restored to a higher level (not quite to the control)
Learning was less hippocampus dependent over time
After they then inactivated the anterior cingulate cortex (ACC) which connects to the hippocampus then we see the inverse: recall of memories becomes more dependent on the ACC with time
Channel rhodopsin can also be used to silence the hippocampus at various timepoints
if this is done during the testing, then there’s a reduced response
but ahead of the testing, then there’s some adaptation so that other neurons are recruited to make up for the lack of association
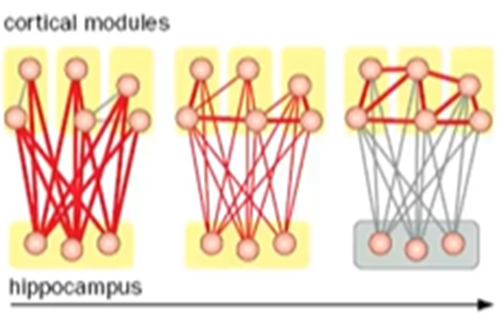
Cortical long term memory
Initially, connections are heavily reliant on the hippocampus which strengthen the connections between hippocampus and the cortex
Over time, connections between the cortical areas and can to some extent exist in the absence of the hippocampus
This is supported by fMRI which shows that memories using visual cues locate to the visual centres of the brain and memories using auditory cues locate to the auditory centres of the brain
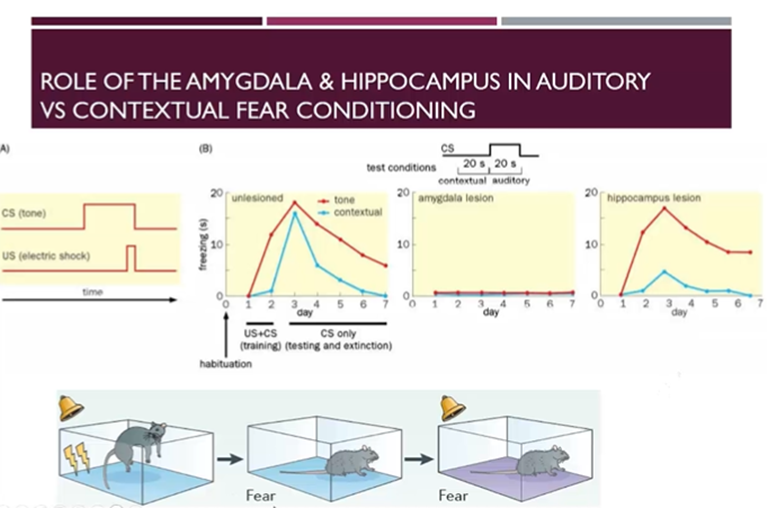
Role of the amygdala and hippocampus in auditory vs contextual fear conditioning
Can time the amount of time sent freezing driven the contextual conditioned stimulus (the cage) vs the tone conditioned stimulus alone
After training, there’s a dramatic amount of freezing, to which the tone conditioned stimulus has a greater contribution compared to the contextual conditioned stimulus
If we lesion the amygdala, then there’s no freezing behaviour as the amygdala is important for the expression of fear
If we lesion the hippocampus, then it is not as important for the association of the tone with the fear, but more important for the association of the cage with fear. Freezing response driven by the contextual stimulus (compared to tone conditioned) reduced much more in hippocampus-lesioned mice
Human fMRI signals in fear conditioning
If we pair an unpleasant unconditioned stimulus with neutral conditioned stimulus, we can see in fMRI imaging, there’s activity in the amygdala and we can also see activity in the amygdala during extinction when the conditioned stimulus only is applied
Amygdala in humans is also important in fear conditioning
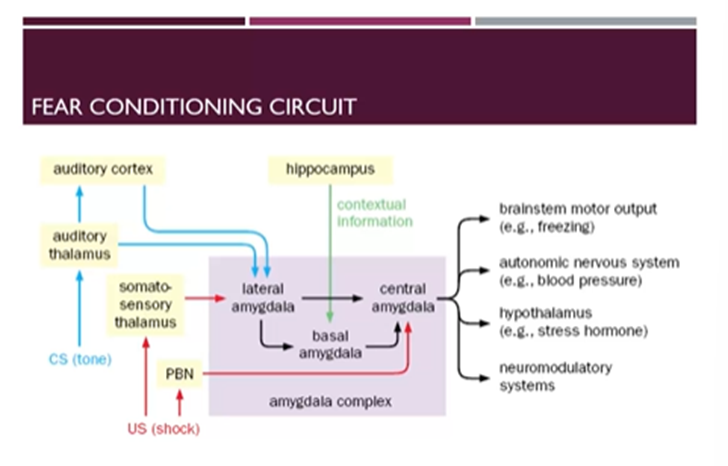
How do these circuits work?
Auditory stimulus comes in through the auditory system → auditory thalamus → auditory cortex → inputs into the lateral amygdala
The foot shock comes in through the somatosensory thalamus and the parabrachial nucleus
Somatosensory thalamus → lateral amygdala
PBN → the central amygdala
Spatial context comes in through the hippocampus → feeds into the basal amygdala
Within the amygdala complex there are relays from the lateral and basal amygdala to the central amygdala which co-ordinates
Brainstem motor output
Autonomic NS (blood pressure)
Hypothalamus hormone release
Neuromodulatory systems
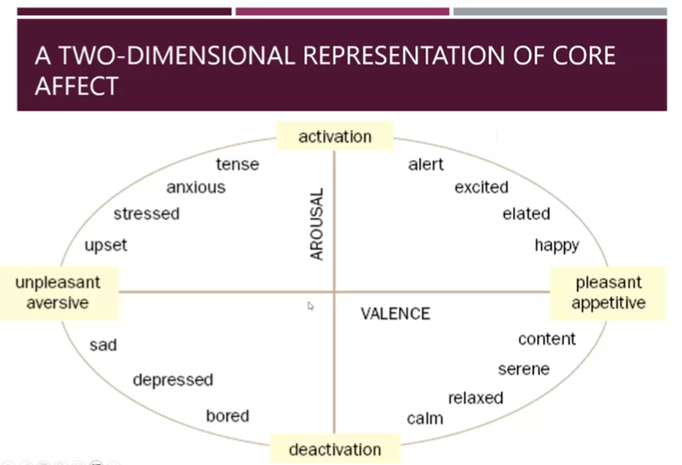
Representation of core affect
Can encode a range of valence and arousal states with this activation
These act as important motivational drivers for learning
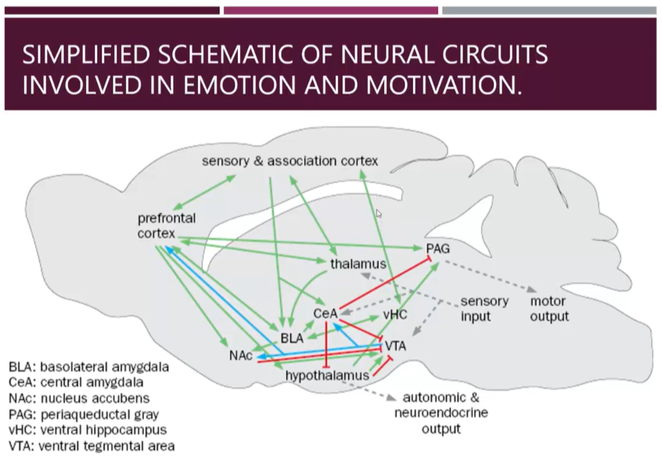
Nerual circuit connections of the central amygdala
This schematic shows all the inputs feeding into the amygdala with excitatory input in green, inhibitory inputs in red and dopaminergic, modulatory input in blue
Central amygdala inhibits the periaqueductal grey which inhibits motor output to regulate freezing
Regulates stress hormone release from the hypothalamus
Has mainly inhibitory outputs
Dopaminergic modulation from the VTA is an important modulator of the reward of learning
Olfactory conditioning in drosophila
T maze experiment where fruit flies placed in a training chamber where they were trained to associate an odour (conditioned stimulus) with a foot shock (unconditioned stimulus)
Then bought to a T maze where they are given the choice between 2 odours: conditioned or unconditioned
Have to tightly associate the conditioned stimulus to the foot shock and avoidance of the associated odour is only seen when you have this paired protocol
Wildtype flies learn the association relatively well but will forget about it, this can be improved with more training
Mutant flies were taken and training undertaken to identify mutations which result in decreased associative learning → two mutants identified: dunce and rutabaga which encode parts of the cAMP pathway
Rutabaga: encodes the adenylate cyclase
Dunce: encodes the phosphodiesterase
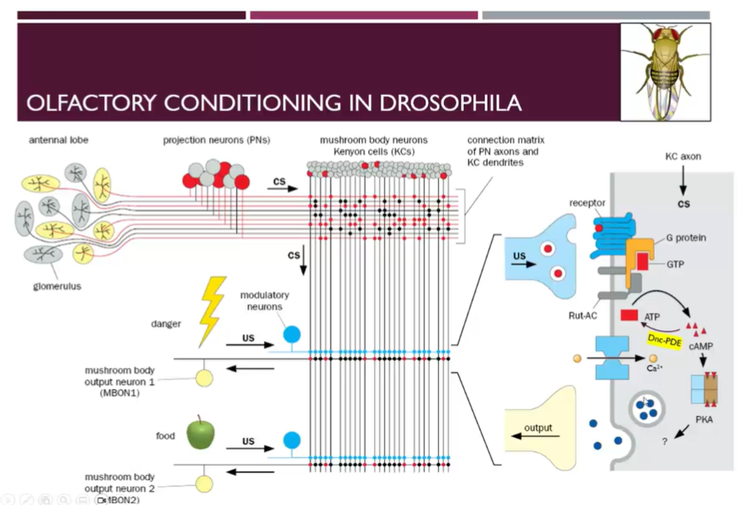
Circuit for olfactory learning in fruit flies
Flies have antennae which are multifunctional sensory organs responsible for sensing odours, temperature, hearing, humidity etc.
Cell bodies of the ORNs are in the antennae and they make contacts with the projection neurons in the antennae lobes → kenyon cells in the mushroom bodies (equivalent of the hippocampus in the fly)
Kenyon cells found in different anatomical structures or lobes
Different Kenyon cells make different projections to different projection neurons and each Kenyon cell will get a different level of input from each odorant
Kenyon cells will connect further making synapses with:
different modulatory dopaminergic neurons which can carry information from the unconditioned footstock
Also make connections with mushroom body output neurons (Mbods) which lead to motor responses
Dopamine released from the MBOD1 neuron to the receptor on the KC axon causes the activation of G protein, which causes the activation of adenylyl cyclase converting ATP→cAMP which activates PKA etc → this can also result in activation of calcium channels and cause the influx of calcium
Also have the activation of CREB and protein synthesis with long term stimulation
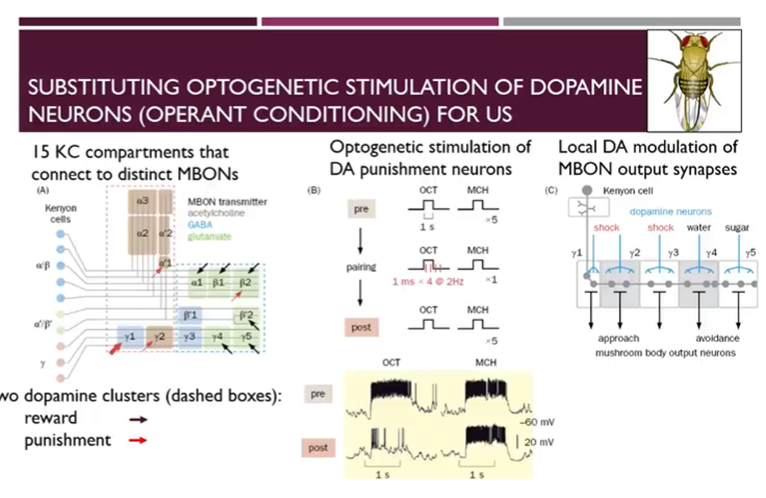
Substituting optogenetic stimulation of dopamine neurons
Use optogenetic to stimulate the right DA neurons and so we can substitute footshock with blue light exposure (which can be coupled with an odourant)
Creates a reasonable change in avoidance based on this training
If you apply both odours simultaneously or use a mutant, this is diminished
15 different compartments of the kenyon cells which cluster based on anatomy and the neurotransmitters of the output neurons
Cell bodies of the Kenyon cells listed on the left hand side and they then make connections with different mushroom body output neurons to the 15 different areas
Dashed outlines of the two rectangles represent 2 different clusters of DA cells which modulate the connections between mushroom body cells and output neurons
For one of the clusters, the dominant valence of the dopaminergic input is reward (E.g: water when its thirsty) → indicated by black arrows
However, there are also dopamine clusters which are responsible for punishment input (indicated in red)
Use optogenetics to simulate punishment with a particular odour and record from neurons post training and shows that octanol now leads to reduced activity at the relevant synapses
Single kenyon cell makes many local connections where it gets input from one class of DA neurons which may encode punishing stimuli, but other DA inputs which are associated with rewarding unconditioned stimuli
Unconditional stimuli have local effects such that they affect local signals which are then transmitted to mushroom body output neurons → single mushroom body neurons can be a relay for DA input and mushroom body outputs at different positions along the projections
Re-Explained Pathway of drosophila learning
Basic circuitry: ORNs (in antennae) → Projection neurons (antennae lobes) → Kenyon cells (Mushroom bodies) → MBONs → behaviour
Kenyon cells are important for encoding the odour: different doodurs will preferentially activate a different subset of Kenyon cells
Their synapse onto MBON neurons generate the behavioural output, but this can be modified by dopaminergic neurons which synapse onto the KCs which encode value (E.g: reward or punishment)
Dopaminergic neurons synapse onto the Kenyon cell axon terminals to preferentially release DA onto specific regions of the KC axon and provide the teaching signal by modifying the strength of the synpase between the KC and the MBON
Dopamine release onto the KC axon terminal will result in incraesed cAMP, increased PKA etc. leading to increased synaptic plasticity. Therefore, the KC → MBON synapse will become preferentially strengthened depending on the circuit
Even though the odour will activate the same Kenyon cells, the KC → MBON synapse has been altered so that the behaviour produced in response to KC activation will change.
Relevance for mammalian learning
In drosophila we see that if we knock out adenylate cyclase in rodents then we see that LTP is reduced
If we do a behavioural assay where we associate a darkened part of the environment with a footshock → the more hesitant they are the better trained they are
Mice naturally want to seek out dark environments so should theoretically prefer this part of the arena
Mice that lack adenylate cyclase have less hesitancy going into the dark environment
Operant conditioning of reward
Train mice to press a lever to obtain a reward
However, this can be stimulated with an electrode inserted into the brain which can electrically stimulate the reward centre
Can set up a cage where on one side of the cage receive a reward and must walk across a cage which gives them foot shocks in order to obtain the next reward
Found that if placed in the right position, mice are more willing to tolerate the foot shock for neural reward than a food reward after a 24h fast
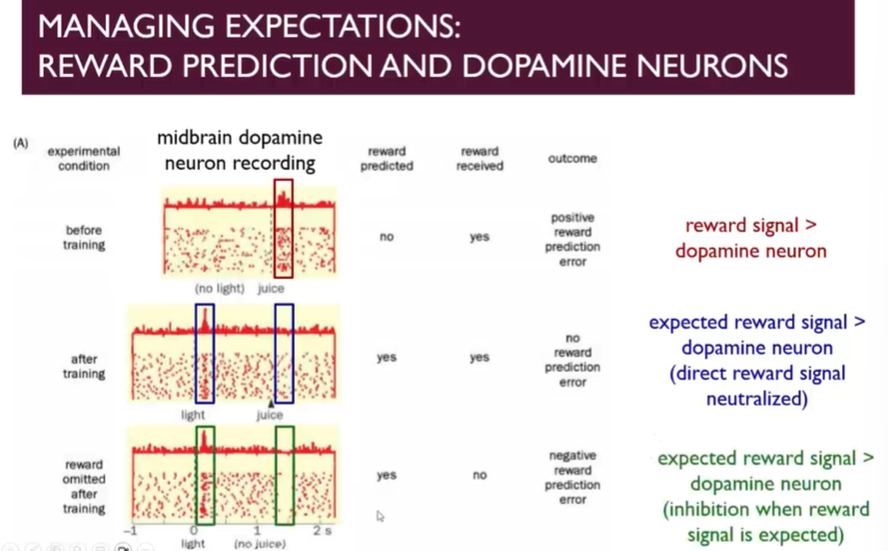
reward prediction and dopamine neurons
Record from a midbrain DA neuron in primates and associate a light stimulus with a drop of juice for an immobilised monkey
In the absence of light giving the monkey a drop of juice leads to a rewarding signal → if you don’t expect it, it feels rewarding
However, if trained and receiving the reward when its expected, then the reward is associated with the prediction of the reward (conditioned stimulus), no longer the unconditioned stimulus (the drop of juice)
Once the monkeys are conditioned to expect the juice and the conditioned stimulus is signalled (indicating that they will receive a reward soon), the basal level of activity is reduced
Theres inhibition when the reward signal is expected, known as negative reward prediction error
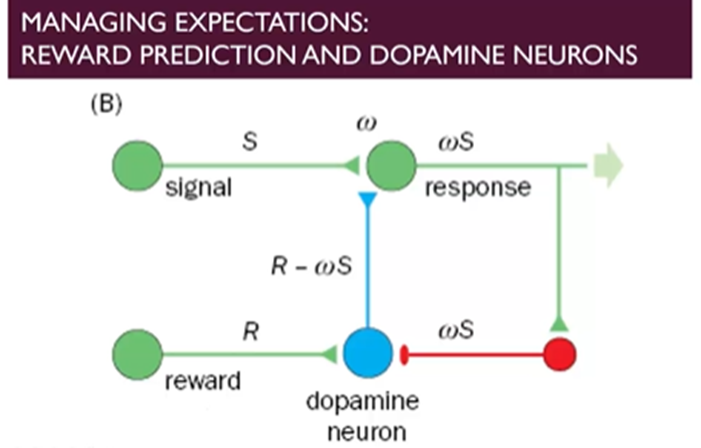
How does this work at the level of the neural circuit?
Light stimulates a response and the reward will stimulate the activity of the DA neuron
This synapse is tuneable and if associated with reward then it can give a stronger signl
As soon as the expectation is raised then there’s negative feedback which brings down the activity of the DA neurons → the level of activity is reduced relative to the reward and the main signal comes from the signal itself
If you have the signal itself and not the reward, then you have the negative feedback but without the dopamine from the signal itself
What happens at the molecular level
Strengthening of the circuit
juice activates sensory and reward pathways releasing glutamate and exciting DA neurons
Initially the light stimulus provides weak activation, but then when light occurs and reward follows, DA neurons fire strongly and the synpases carrying info about the light become strengthened
Due to LTP like processes strengthening this synapse, the light alone is enough to stimulate DA neurons
DA itself is also important as a neuromodulator, preferentially tagging and strengthening important synapses
Negative reward prediction error
because this circuit is reliably being frequently stimulated, negative feedback mechanisms occur and GABAergic neurons are recruited to tune down this synapse
Additionally, DA autoreceptors (D2) inhibits AC, decreases cAMP, reduces Ca2+ entry and supresses further firing
When the reward is not given, the inhibitory pathways overcompensate for the DA release when there is none and therefore DA firing falls below baseline
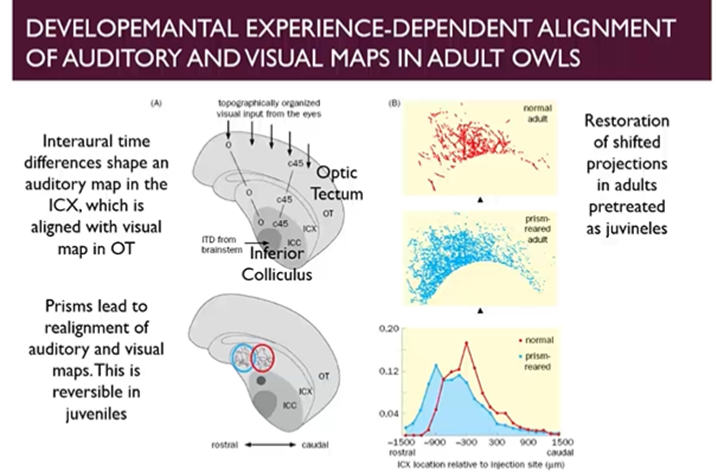
Developmental experience dependent alignment of auditory and visual maps in adult owls
Owls mostly nighttime hunters which pairs audity and visual stimuli to pinpoint the location of its prey
Does this by linking a spatial map of its visual environment in the optic tectum to a spatial map of its auditory environment in the external nucleus of the inferior colliculus
If you distort the visual field of the owls by placing prisms on their eyes, initially, they will have a hard time hunting, however it can adapt to this by realigning the association of auditory/visual maps once it has a distortion of vision
We can make adjustments during development in juvenile owls to shift the alignment of visual/auditory maps
After the removal of the prisms which caused a shift in alignment during the development, the owl can readjust and shift the alignment of the maps back to the original configuration
If you reapply the prisms in adulthood to either those that had been prism-reared and those that had not, theres a dramatic advantage to the prism-reared owls
Shows that its much harder to achieve the same level of learning in adulthood
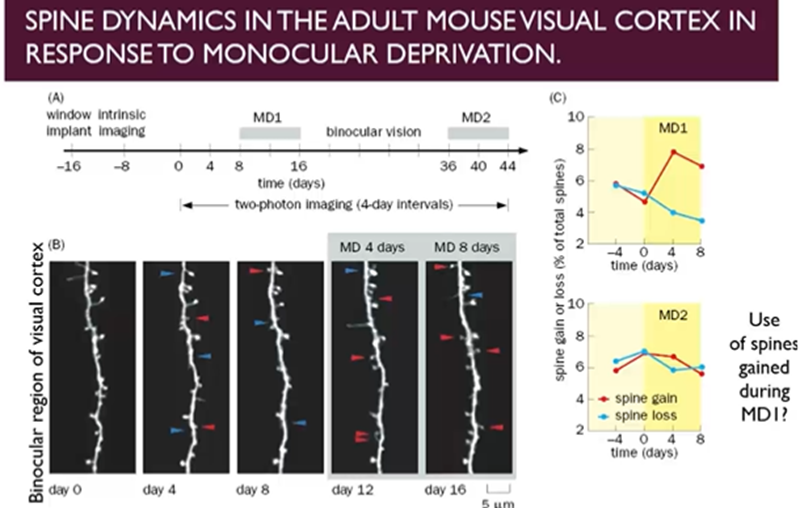
Spine dynamics in the adult mouse visual cortex in response to monocular deprivation
Close light input to one of the eyes of the mouse and look at the dendritic spines in the visual cortex in the binocular region
Dendritic spines constantly forming and disappearing, but only when they form meaningful connections will they stabilise
After 8 days of monocular deprivation, dendritic spines preferentially gained and not lost → pressure to change the connectivity of these neurons to restore the level of input and processing, done at the subcellular level by creating more dendritic spines
If we now allow a period of binocular vision for 20 days, then repeat this process, then we no longer see a repeat of this pattern
suggesting that the extra spines which were created during the initial period of monocular deprivation may still be available during the second period of monocular deprivation
Connections made during initial period of monocular deprivation, even though the neurons may have restored levels of functioning when both eyes available, they are still untapped resources
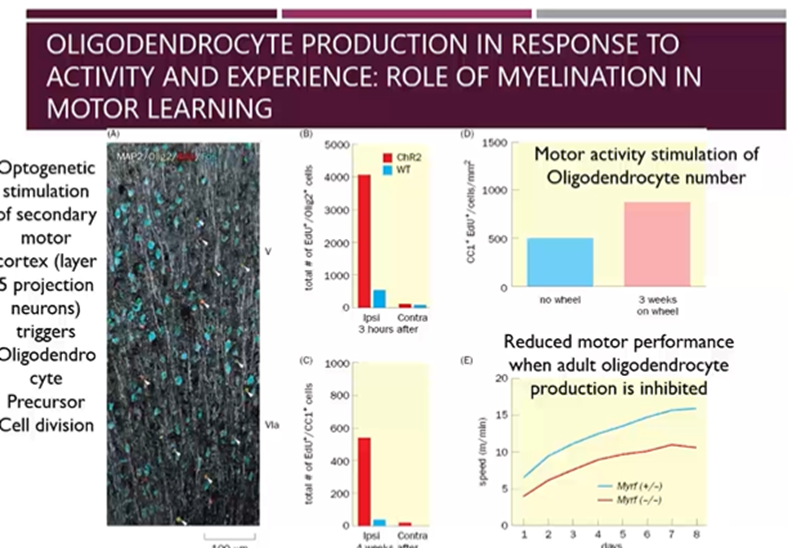
Oligodendrocyte production in response to activity and experience: role of myelination in motor learning
Myelination results in more effective learning due to increased myelination of neurons
Activity and experience can stimulate the production of oligodendrocytes
Optogenetic stimulation of the layer 5 projection neurons of the motor cortex triggered OPC cell division, resulting in more mature oligodendrocytes
Also allowed mice to train on a running wheel and those that were not and found increased number of oligodendrocytes in the mice who ran on the wheel
Finally, tested motor performance when adult oligodendrocyte production is inhibited
Motor activity stimulates oligodendrocyte division maturation, myelination of motor neurons and if we inhibit this motor performance is affected
Maybe this is how motor learning is improved too, with increase stimulation, you get increased myelination and therefore better insulation results in better performance
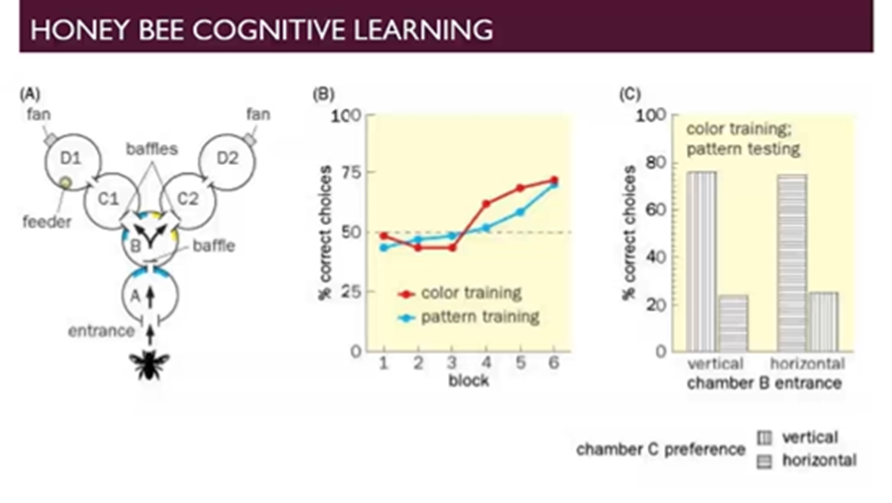
Honeybee cognitive learning
Train honeybees to travel through a system of compartments to find a feeder based off colours or patterns
Each compartment that the bee travels through has a different colour in one experiment and based of pattern in a separate experiment
The honeybees trained on colours but tested on patterns the honeybee must use the context of sameness rather than just the direct visual stimulation and can do this to a reasonable extent
Shows that cognitive learning can happen in honeybees and certainly happens to a higher extent in higher order organisms