Pharmacology Exam 2 Study Guide
1/144
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
145 Terms
What’s the difference between first and second messenger systems?
Primary messengers: the extracellular substances that can initiate intracellular activities. Located outside the cell and operate through binding with their respective receptors.
Common examples —> hormones neurotransmitters, local mediators, etc.
Secondary messengers: The actual intracellular signaling molecules that send signals from receptors to targets. Small molecules that are located inside the cell and operate through the activation of PROTEIN KINASES (often but not always)
Common examples —> cAMP system, phosphoinositol system, cGMP system, tyrosine kinase system, and arachidonic acid
Are secondary messengers synthesized on demand or present in an active state?
Second messegers can often be present in an inactive state and activated quickly, enabling rapid signal propagation
What are the different types/ classes of second messegers?
Hydrophobic - lipid, sphingolipid
Hydrophilic - cyclic nucleotides, IP3, ions
Gaseous - endogenous sources
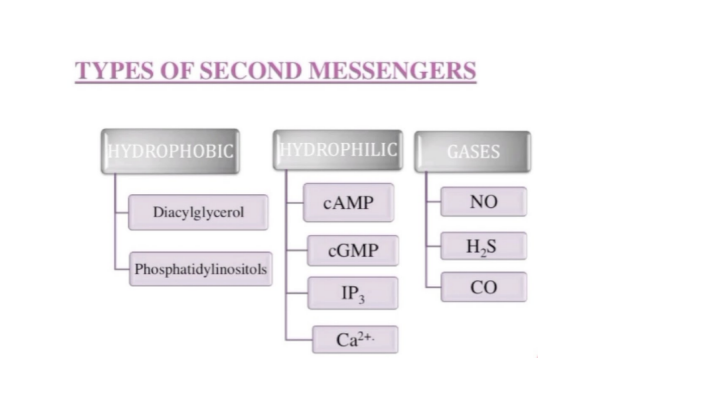
What class of second messenger is DAG?
Hydrophobic
What class of second messenger is phosphatidylinositols?
Hydrophobic
What class of second messenger is cAMP?
Hydrophilic
What class of second messenger is cGMP?
Hydrophilic
What class of second messenger is IP3?
Hydrophilic
What class of second messenger is Ca++?
Hydrophilic
What class of second messenger is NO?
Gas
What class of second messenger is H2S?
Gas
What class of second messenger is CO?
Gas
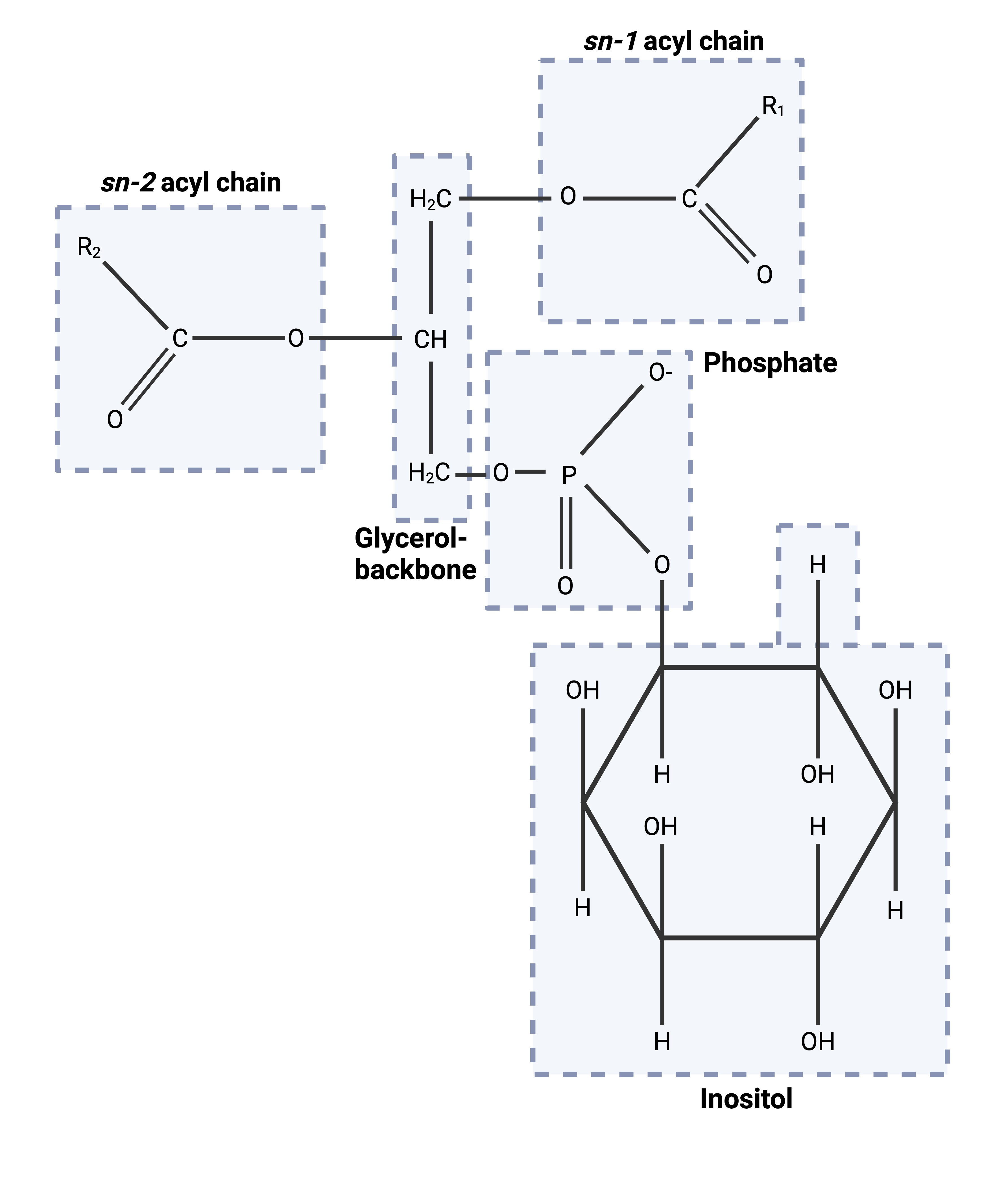
What lipid second messengers have 2 acyl chains and remain membrane associated?
phosphatidic acid, DAG and ceramide
What is a requirement for second messengers to remain membrane associated?
Must have 2 acyl chains
Second messengers that retain only 1 acyl chain will do what?
Dissociate from membranes and serve as SOLUBLE second messengers
Examples —>
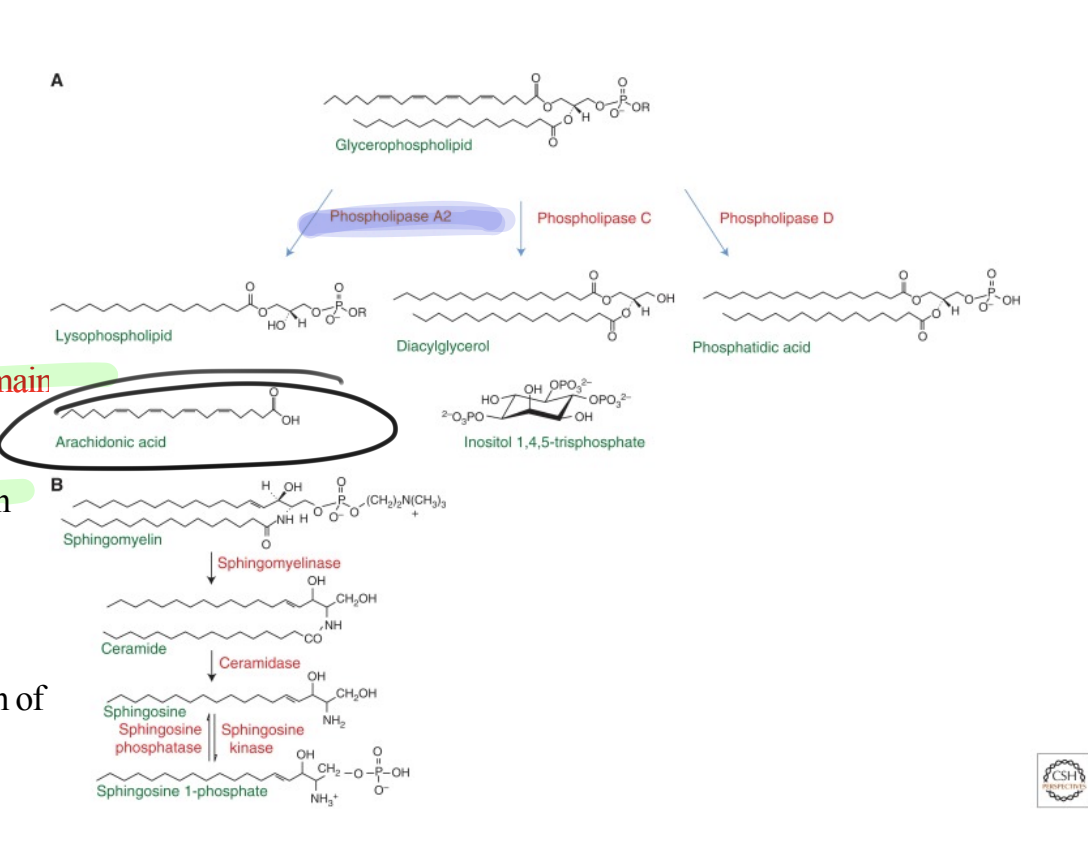
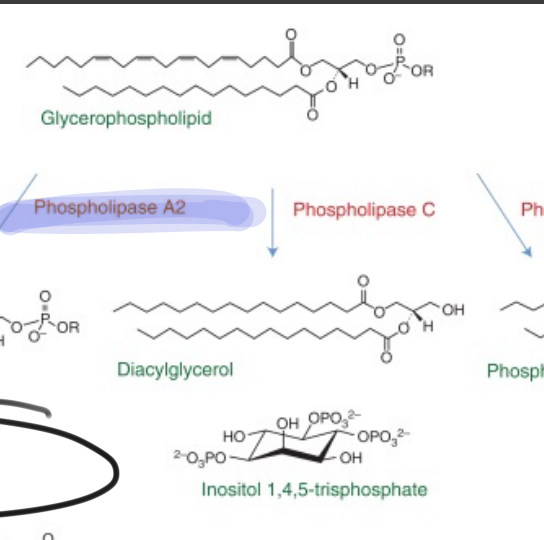
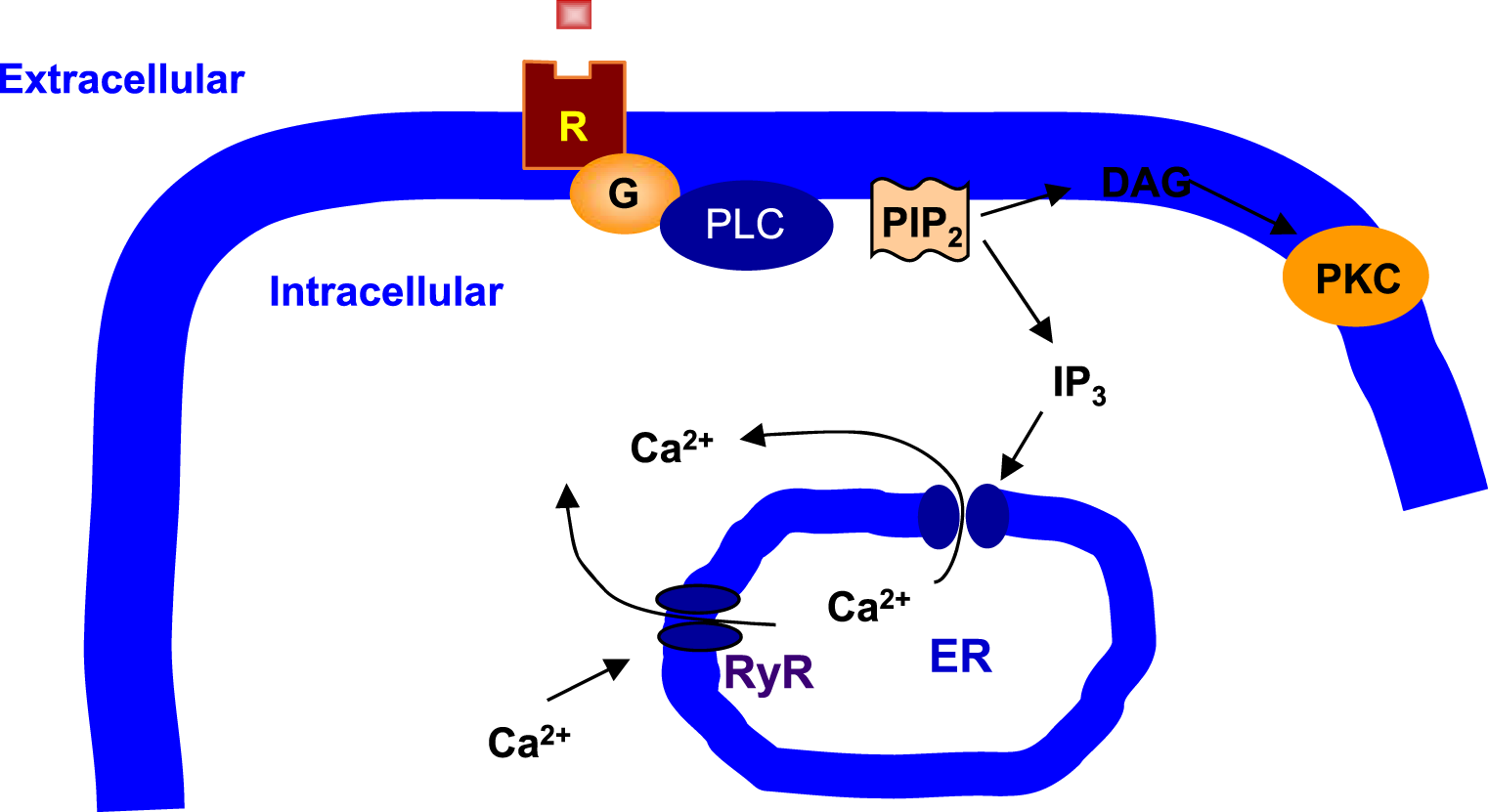
What enzyme cleaves glycerophospholipid into diaclglycerol (DAG) and IP3 (inositol)
Phospholipase C
What second messengers release calcium?
IP3 via release from ER
DAG via DAG-Ca++ binds to and activates conventional PKC isoforms, which then phosphorylate target proteins that can lead to further increases in intracellular Ca++
PLC cleaves phospholipids to create these two products
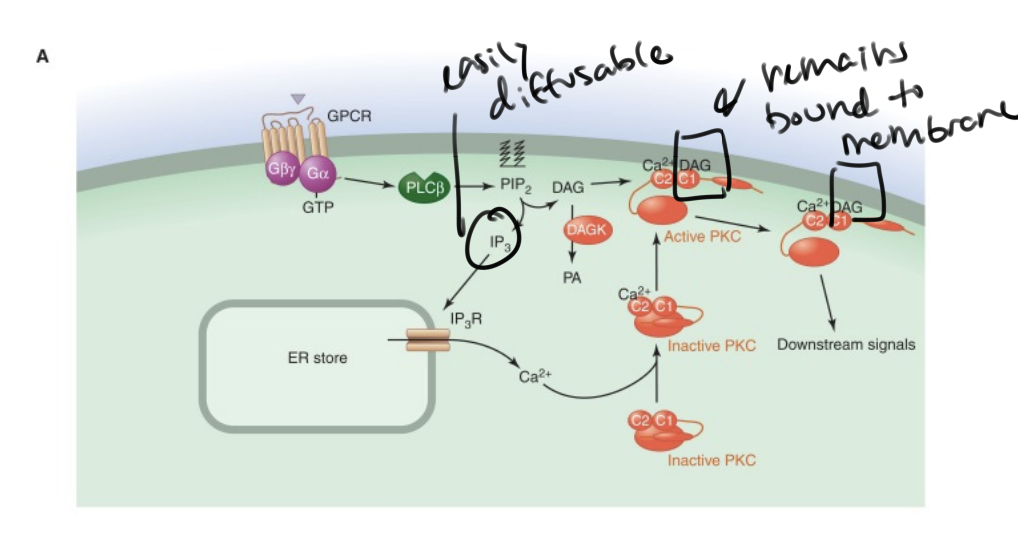
Does IP3 stay in the membrane?
No! IP3 diffuses through the cell and binds to calcium channels in the ER
Does DAG stay in the membrance?
Yes!! It remains in the membrane and activates PKC which will eventually increase intracellular calcium
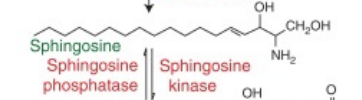
Where are sphingolipids primarily localized in the body?
Within the nervous system
What is a sphingolipid?
A specialized group of lipids that are essential to the composition of the plasma membrane of many cell types.
Ceramide is the primary active _____ that acts as a second messenger
Sphingolipid
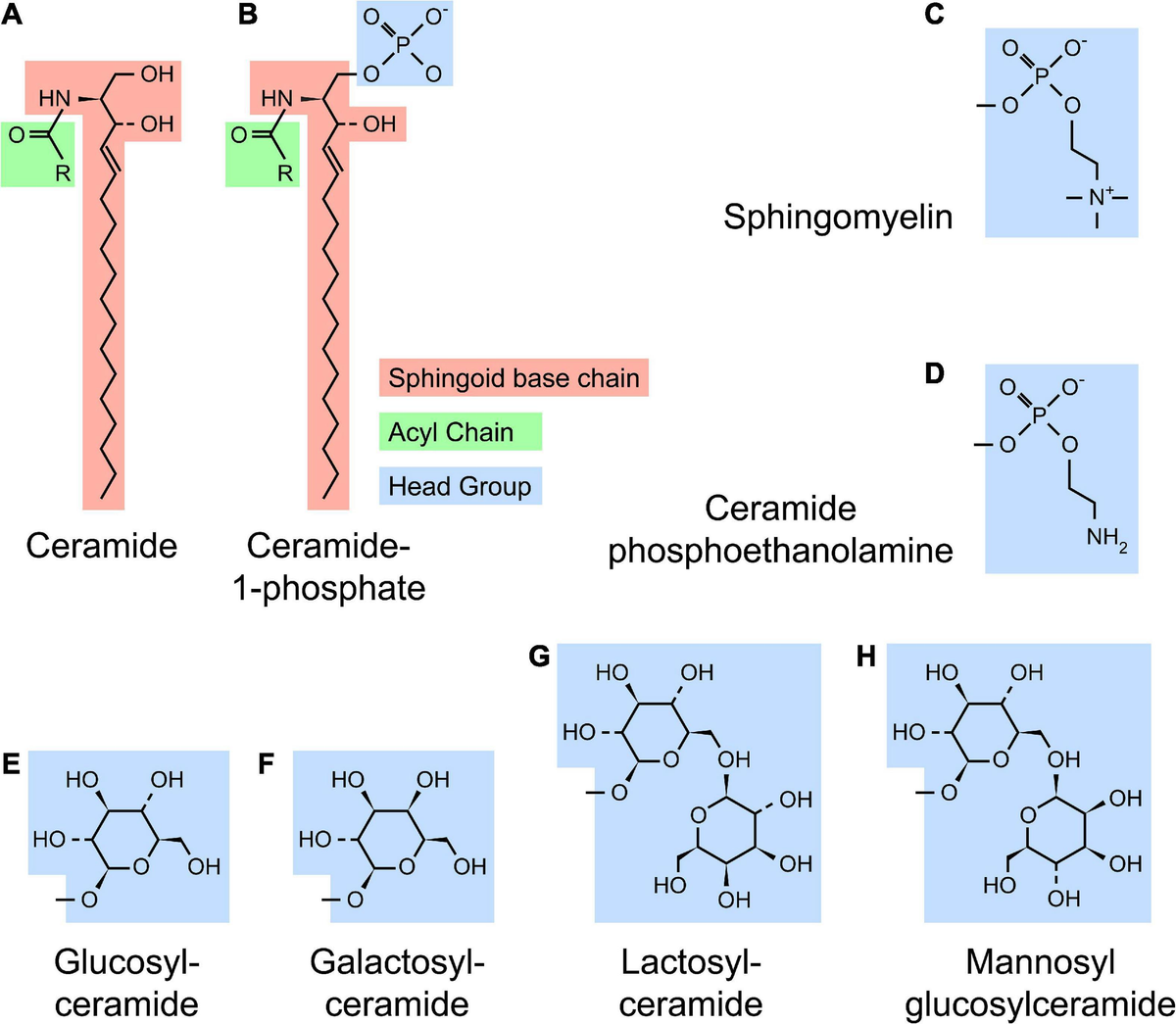
What activity does ceramide have
Bioactive liquid = Ceramide is a bioactive liquid because it is an active signal (bioactive) that resides and works within the cell membrane (liquid/lipid) to trigger or regulate major cellular actions like death or repair.
Involved in apoptosis
Major component of the skin's barrier
What impact does ceramide have on the skin?
Plays a key role in skin barrier function
Often present in skin care products. Act as the glue that holds skin cells together and strengthens the skin barrier and retaining moisture. It is not removing dead skin. Prevents dryness and irritation.
Supposed to : improve skin health by improving ceramide levels

What is sphingosine-1-phosphate derived from?
Sphingolipid
What function does sphingosine-1-phsophate have?
Acts as a counter-regulatory molecule to ceramide that promotes cell survival proliferation and migration by activating specific receptors
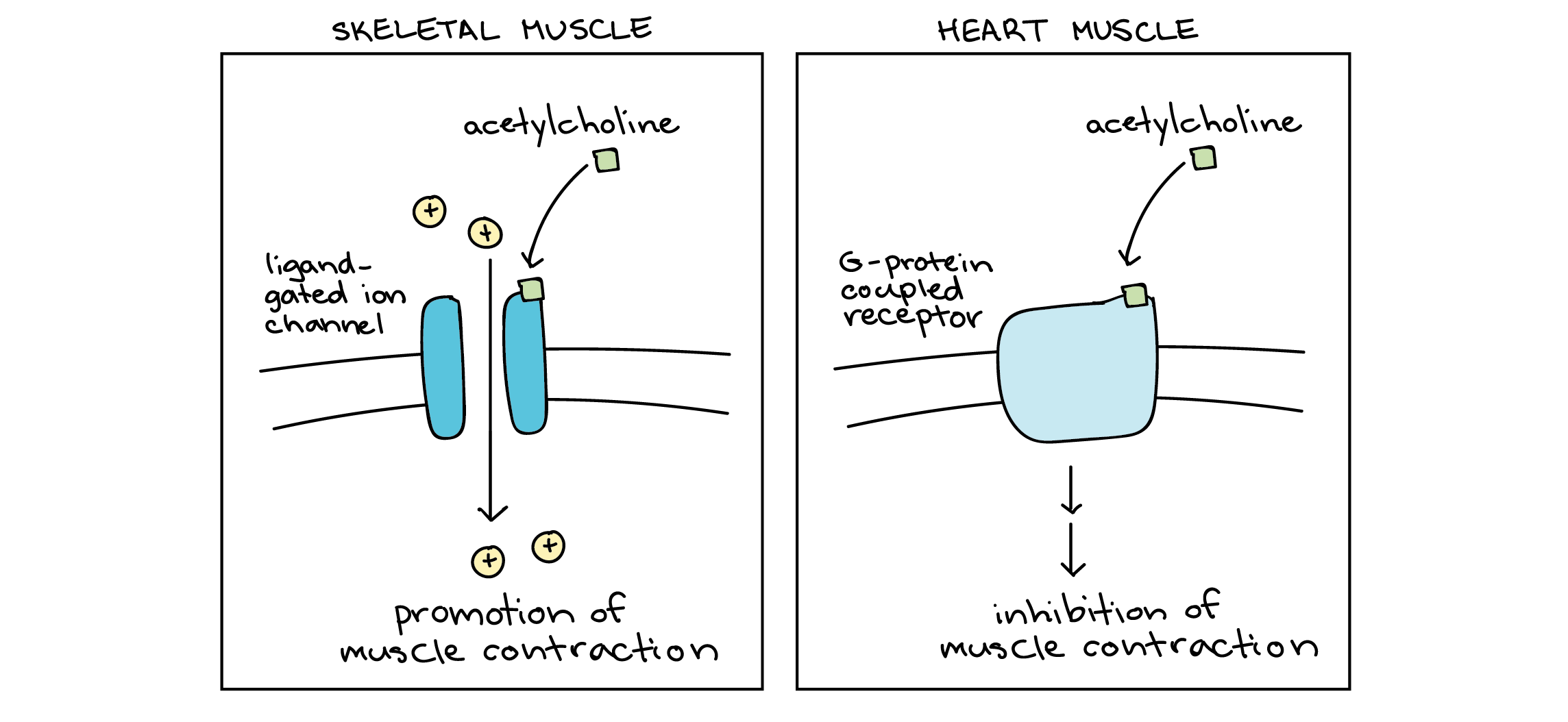
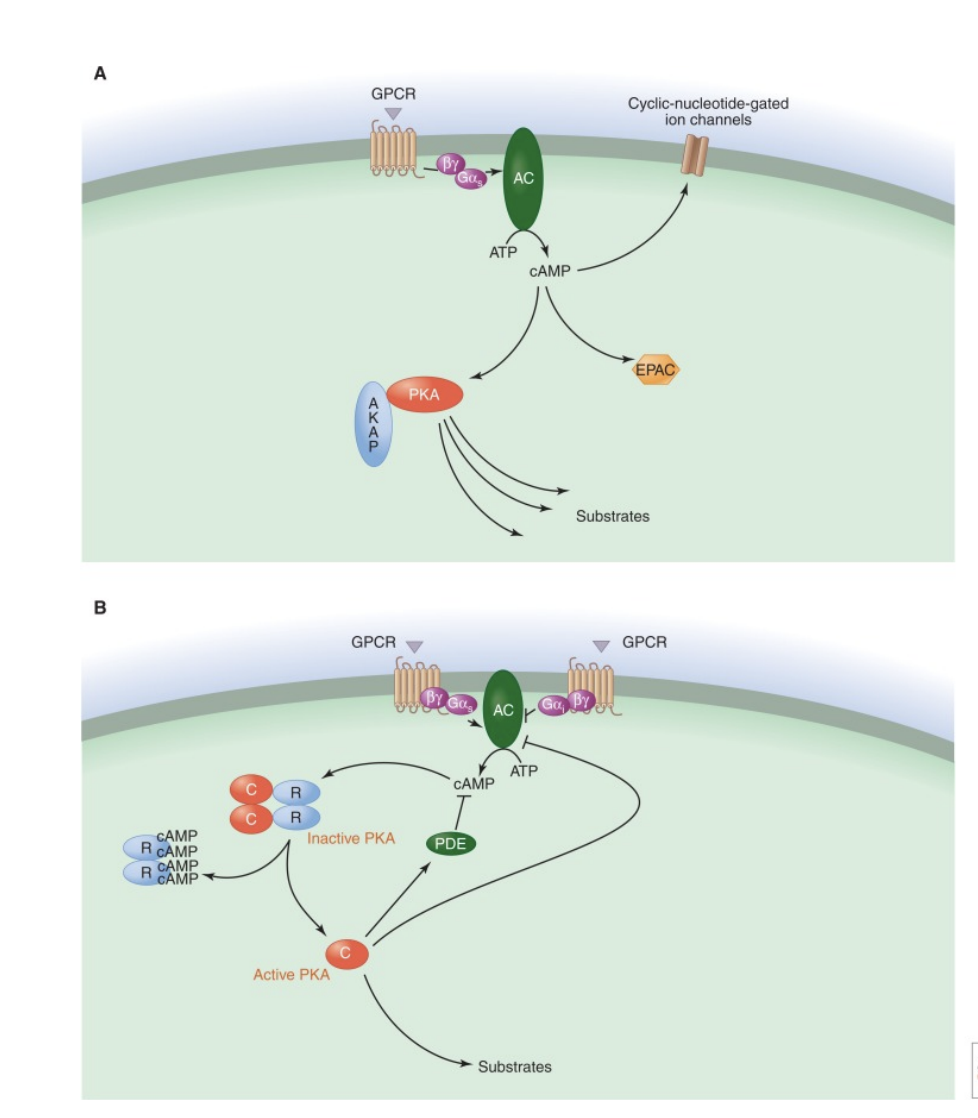
What are the actions of epinephrine released during the fight or flight response?
Triggers the RELEASE OF GLUCOSE through the use of cAMP as a second messenger
What is different about PKG?
Unlike PKA, PKG EXPRESSION IS RESTRICTED TO THE BRAIN, LUNGS, AND VASCULAR TISSUE (allows us to target
What function does PKG have?
a cyclic GMP-dependent serine/threonine kinase that plays a crucial role in cell signaling, vasodilation, and smooth muscle relaxation
What is the most prevalent ion acting as a second messenger?
Calcium!!!
How does calcium act as a second messenger?
When a signal is received, calcium channels open and that causes a RAPID influx of Ca++ ions. These ions bind to proteins to initiate cellular responses that have longer term effects.
Ca++ is important for muscle contraction, neuronal signaling, and hormone release
What’s specific about gases as second messengers?
They can cross membranes and diffuse due to their small size and non-polar nature
Functions: many!! Blood pressure regulation, neurotransmission muscle relaxation, and immune response
How are gasses used as second messengers produced?
Through dedicated enzymes within the cell and this allows for targeted signaling!!
What is the physiological source of NO?
What are the actions of NO?
Nitric oxide synthase an enzyme that produces NO from L-arginine
Can have enzyme localization!!
endothelial NOS: primarily responsible for producing vascular NO
neuronal NOS: found in neurons
inducible NOS: expressed in immune cells during inflammation
Considered the main source of NO for regulating blood flow and vascular function (potent vasodilator)
How is CO as a second messenger produced by the body?
The body produces CO through heme degradation mainly in the liver, spleen, and kidneys
What enzyme breaks down heme in the presence of oxygen and NADP?
Heme oxygenase!
The process produces CO, biliverdin IX and Fe2+
Biliverdin reductase breaks down biliverdin IX into bilirubin IX and that is excreted in the bile and the urine
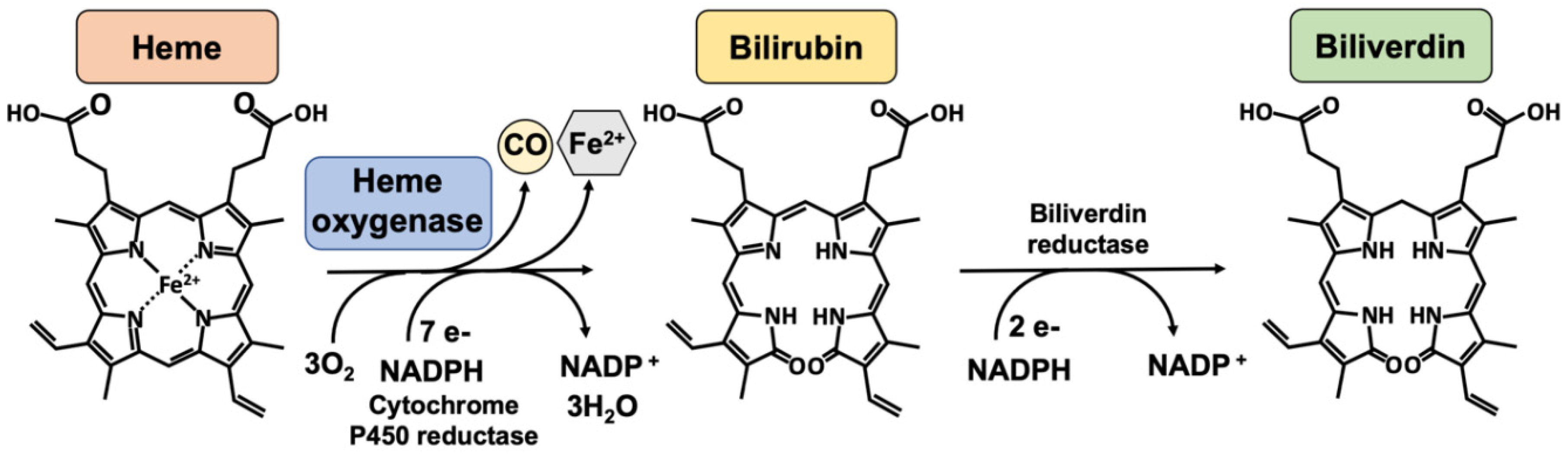
What is the function of CO as a second messenger?
CO activates guanylate cyclase and increases levels of cGMP —> affects vasodilation and neurotransmission
Vasodilation: by increasing cGMP levels, CO promotes relaxation of blood vessel smooth muscle, leading to vasoldilation
Neurotransmission: CO can modulate neurotransmitter release in certain neuronal synapses
What does CO do in the carotid body?
Main function of the carotid body?
helps regulate oxygen sensing!
senses lower oxygen levels and tells the body when it’s not getting enough oxygen
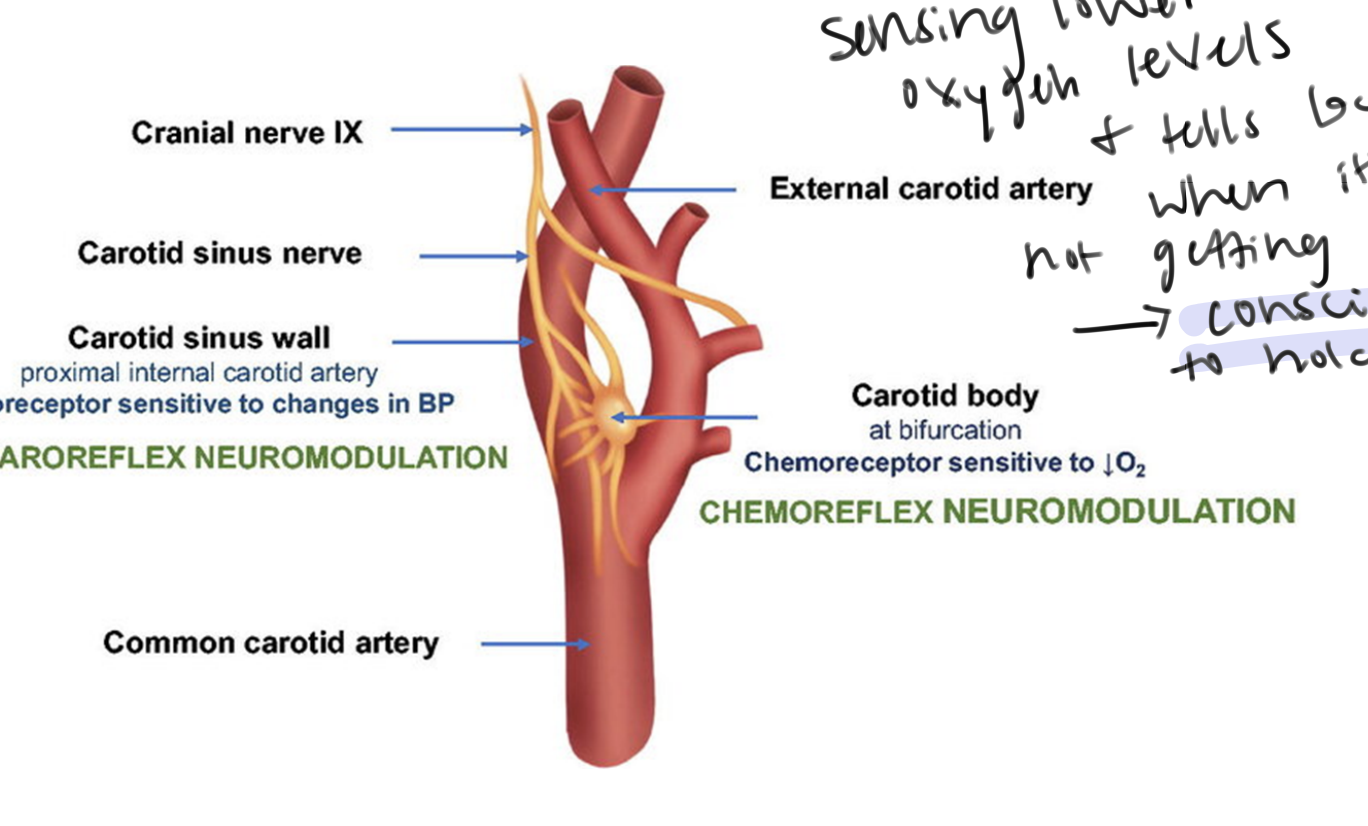
Its primary function is to monitor arterial blood, acting as the main sensor for decreases in oxygen (O2) levels (hypoxia), along with detecting changes in and , triggering rapid respiratory adjustments
How is H2S produced in the body?
Enzymatic breakdown of some amino acids
How does H2S modify protein function?
By adding a sulfur group to cysteine residues on proteins —> disulfide bonds
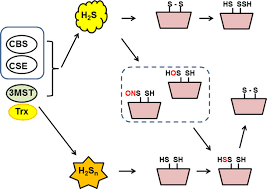
What are the important functions of H2S as a second messengers
CV system: H2S contributes to vasodilation by relaxing smooth muscle cells in blood vessels - helps regulate blood pressure
NS: Learning and memory
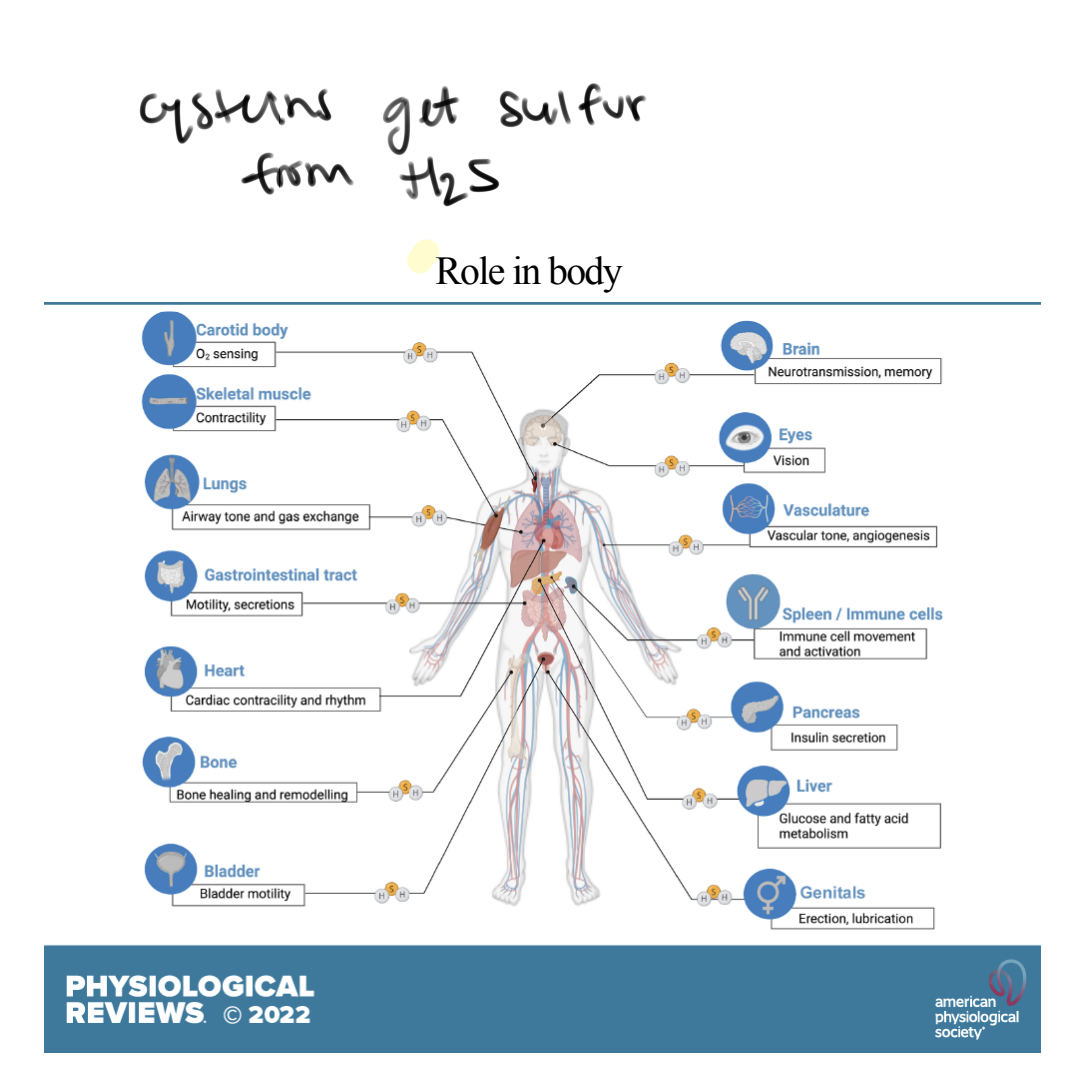
What 3 second messenger gases play a role in vasodilation?
NO
CO
H2S
Lecture 11 Summary Wyckoff Included
cAMP:
Generated from ATP
Diffuses freely in the cytoplasm (hydrophilic)
P-lates cellular proteins and regulates gene transcription factors
cGMP:
Induced by NO!
Regulates activity of some protein kinases
P-lation of cellular proteins
Regulates ion channels and inhibits intracellular calcium fluctuations
IP3 and DAG:
Created by PIP2 hydrolsis
DAG remains in the membrane where it stimulates PKC
IP3 diffuses into the cytoplasm where it releases Ca++ from the ER
Ca++:
Regulates many processes and level in body is controlled by release and removal
Direct effect on cell motility
NO:
Activates cGMP production
Regulated by Ca++
Mediator of gene regulation of during wound healing
Vasodilator
CO:
Diffuses through cytosol and across cellular membranes
Shown to increase cGMP levels by activating guanylate cyclase

What’s the difference between PKGI and PKGII?
Type I enzyme is soluble and predominately cytoplasmic
Type II enzume is attached ot biological membranes
Why are second messengers importnat?
We can use them as drug targets!!
What is important about Lithium?
(Li+) that acts as a modulator or inhibitor of second messenger systems.
Interferes with the phosphoinositide pathway
Affects second messenger signaling (IP₃/DAG pathway)
Clinically relevant in neuropsychiatric treatment
By lowering these intracellular messengers, lithium reduces excessive dopamine and glutamate activity and increases GABA signaling, helping to stabilize mood.
What are some second messenger analog drugs??
Analogs are compounds that mimic the action of a natural second messenger molecule within a cell producing therapeutic effects by triggering the production of activation of a second messenger molecule —> downstream responses
cAMP analogs: forskolin, dibutyryl cAMP
calcium channel modulators: ionomycin, thapsigargin
phosphoinositide pathways activators: lithium
What is Forskolin?
cAMP analog that activates adenylyl cyclase, increasing cAMP levels
What is Dibutyryl cAMP?
A cell-permable cAMP analog that directly mimics cAMP activity
What is Ionomycin?
A calcium channel modulator that facilitates calcium influx into the cell, mimicking the effect of calcium as a second messenger
What is Thapsigarin?
A calcium channel modulator that depletes intracellular calcium stores, leading to downstream calcium signaling
What is Lithium?
a phosphoinositide (PIP) pathway activator that impacts second messenger signaling
What drugs are cAMP analogs?
Forskolin
Dibutyrl cAMP
What drugs are calcium channel modulators?
Ionomycin
Thapsigargin
What drug is a phosphoinositide pathway inhibitor?
Lithium
What is a nucleoside analog? What are they used for?
Class of medications that mimic natural nucleosides
MOA: they are mistaken for natural nucleosides and become incorporated into the growing DNA or RNA chain —> terminates chain growth
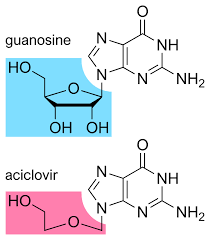
Treat viral infections such as HIV, Hep B, herpes simplex virus, and cytomegalovirus
Some can inhibit cancer cell growth and replication
Can suppress the immune system and reduce inflammation
Which of the following best describes the primary role of second messengers?
A. Binding extracellular ligands
B. Amplifying intracellular signaling cascades
C. Transporting molecules across membranes
D. Degrading signaling proteins
B
Why can second messenger systems respond rapidly to stimuli?
A. They are synthesized de novo after receptor activation
B. They are stored in vesicles
C. They are already present in inactive or controlled states
D. They require transcriptional activation
C
Which of the following second messengers remains membrane-associated?
A. IP₃
B. cAMP
C. DAG
D. Ca²⁺
C
Why: DAG retains two acyl chains, keeping it embedded in the membrane.
What is the primary function of IP₃?
A. Activates protein kinase C
B. Opens calcium channels in the ER
C. Activates adenylyl cyclase
D. Degrades phospholipids
B
—> IP₃ diffuses to the ER and releases Ca²⁺.
DAG primarily exerts its effect by:
A. Activating protein kinase A
B. Binding nuclear receptors
C. Activating protein kinase C
D. Increasing cGMP levels
C
—> DAG stays in the membrane and activates PKC.
Why is calcium an effective second messenger?
A. It is abundant in the cytoplasm at baseline
B. It is stored in the nucleus
C. It has a steep concentration gradient
D. It is lipid-soluble
C
—> Low baseline → small increases cause big effects.
The most prevalent ion acting as a second messenger is:
A. Na⁺
B. K⁺
C. Ca²⁺
D. Cl⁻
C
Epinephrine primarily exerts its intracellular effects through:
A. cGMP
B. Ca²⁺
C. cAMP
D. DAG
C
—> Classic GPCR → cAMP → PKA pathway.
—> in the fight or flight response triggers glucose release through cAMP as a second messenger.
Which statement about protein kinase G (PKG) is TRUE?
A. It is activated by cAMP
B. It is expressed in all tissues equally
C. It is activated by cGMP
D. It is only membrane-bound
C. cGMP
—> PKG is activated by cGMP and has both soluble (PKGI) and membrane forms (PKGII).
—> Unlike PKA, PKG expression is restricted to the brain, lungs, and vascular tissue
Which of the following is TRUE regarding PKG isoforms?
A. Both forms are cytoplasmic
B. PKG I is membrane-bound
C. PKG II is membrane-associated
D. PKG is only found in the nucleus
C
Sphingolipids are best described as:
A. Water-soluble signaling molecules
B. Specialized membrane lipids
C. Gaseous transmitters
D. Ion channels
B
Ceramide is most strongly associated with:
A. Cell proliferation
B. Apoptosis
C. Glucose metabolism
D. Neurotransmitter release
B
Sphingosine-1-phosphate (S1P) primarily promotes:
A. Apoptosis
B. DNA degradation
C. Cell survival and proliferation
D. Calcium sequestration
C
Which of the following is TRUE about gaseous second messengers?
A. They require membrane receptors to enter cells
B. They are stored in vesicles
C. They freely diffuse across membranes
D. They only act intracellularly
C
Nitric oxide (NO) primarily increases:
A. cAMP
B. Ca²⁺
C. cGMP
D. DAG
C. cGMP
—> cGMP regulates protein kinases and ion channels. It also inhibits Ca++ fluctuations
Carbon monoxide (CO) exerts its effects by:
A. Activating adenylyl cyclase
B. Activating guanylyl cyclase
C. Blocking calcium channels
D. Inhibiting PKC
B. Activating guanylyl cyclase
—> CO and NO are both vasodilators, they also both actibate cGMP/ activate guanylyl cyclase
Hydrogen sulfide (H₂S) signals primarily by:
A. Binding DNA directly
B. Modifying proteins (e.g., cysteine residues)
C. Activating GPCRs exclusively
D. Degrading second messengers
B
Lithium affects second messenger systems by:
A. Increasing cAMP directly
B. Blocking calcium channels
C. Interfering with the phosphoinositide pathway
D. Activating PKG
C
Which drug increases cAMP by activating adenylyl cyclase?
A. Ionomycin
B. Thapsigargin
C. Forskolin
D. Lithium
C
cAMP analogs:
-Forskolin: Activates adenylyl cyclase, increasing cAMP levels.
-Dibutyryl cAMP: A cell-permeable cAMP analog that directly mimics cAMP activity.
Calcium channel modulators:
-Ionomycin: Facilitates calcium influx into the cell, mimicking the effect of calcium as a second messenger.
-Thapsigargin: Depletes intracellular calcium stores, leading to downstream calcium signaling.
Phosphoinositide pathway activators:
-Li (Lithium): Can interfere with the phosphoinositide pathway, impacting second messenger signaling
Which drug mimics calcium signaling by increasing intracellular calcium?
A. Dibutyryl cAMP
B. Ionomycin
C. Lithium
D. Acyclovir
B
—> Calcium channel modulators:
-Ionomycin: Facilitates calcium influx into the cell, mimicking the effect of calcium as a second messenger.
-Thapsigargin: Depletes intracellular calcium stores, leading to downstream calcium signaling.
Why are A-kinase anchoring proteins (AKAPs) important?
A. They degrade cAMP
B. They transport calcium
C. They localize PKA to specific cellular regions
D. They synthesize second messengers
C
Activation of phospholipase C results in production of:
A. cAMP and cGMP
B. DAG and IP₃
C. Ca²⁺ and NO
D. PKC and PKA
B
Which statement best explains why second messengers are good drug targets?
A. They are only found extracellularly
B. They amplify and control signaling pathways
C. They are structurally complex
D. They are unique to bacteria
B
What is the point of an amplifier and what role does amplification perform in a cell?
Converts small signals into large biological responses
Enables one receptor activation → thousands to millions of downstream effects
Allows detection of very low ligand concentrations
What example did we use in class for amplification?
Phototransduction: producing a detectable electric signal that is transmitted to the brain, allowing for extreme sensitivity to dim light.
In the rod cells of the retina, the absorption of 1 photon results in the inactivation of rhodopsin. This single event causes the hydrolysis of approximately 10 ^5 cyclic GMP (cGMP) molecules, leading to the closure of cGMP-gated sodium channels. The resulting reduction in cation flow causes more than 1,000,000 sodium ions to fail to enter the rod outer segment within the single-photon response
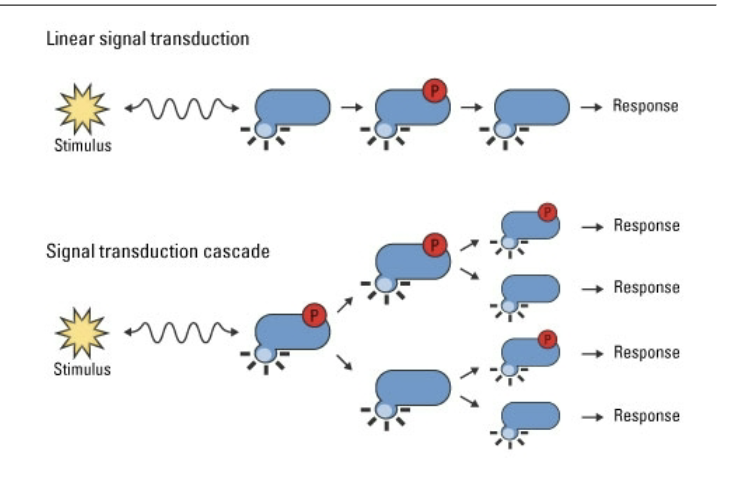
What’s the difference between a signal cascade and a linear signal?
Linear: simple 1->1->1 progression where one molecule activates exactly one downstream molecule, which activates exactly one more. There is no multiplication of the signal. If one receptor is activated, one effector responds. In general, linear signaling is fast, direct, and tightly proportional
Signal cascade amplification follows a 1→10→100→1000 pattern. Explains why a very small number of signaling molecules can generate a very large biological response. Each layer multiplies the signal, creating the cone model: narrow at the top (a single ligand-receptor interaction) and progressively wider as the signal moves downward.
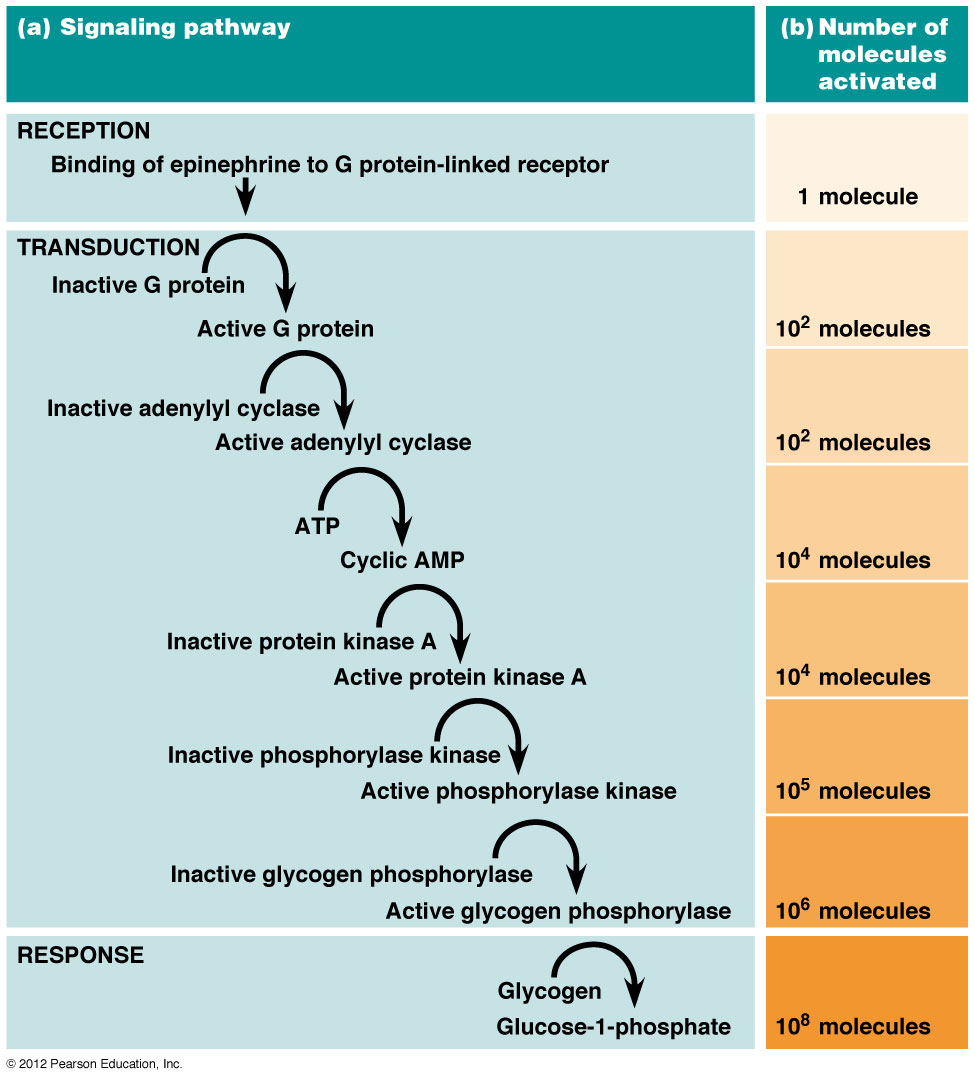
Why is this important from a therapeutic perspective?
Because where you target the pathway changes drug effects:
Top of cascade (receptor):
Blocks entire signal early
Requires less drug
BUT → less specific (more side effects)
Blocking the receptor is not always ideal. Receptors are often widely distributed throughout the body, meaning that receptor antagonism can suppress multiple physiological processes and lead to systemic side effects.
Downstream targets:
More tissue-specific (isoforms)
BUT → must overcome amplified signal
May require higher doses
Instead of shutting off the entire cone at the top (which affects every tissue expressing that receptor), targeting a tissue-specific isoform will reduce systemic side effects and preserve necessary signaling elsewhere in the body.
👉 Tradeoff: efficiency vs specificity
If you’re trying to stop (vs. modulate) a response, what’s the SIMPLEST way to do it?
If you want to STOP a response, your best bet is to have an antagonist at the receptor that would normally begin the signal cascade (at the top of the cone)
If the easiest thing to do is block the receptor that starts a signal cascade, why don’t all drugs act that way?
Specificity, selectivity, potency
Blocking receptors is non-specific because one receptor can often be found in many places throughout the body
Why should you not block epinephrine at its receptor if you wanted to drug that cascade pathway?
β-receptors are found in heart, lungs, liver, vessels
Blocking receptor → affects all tissues
👉 Problems:
Poor selectivity
More side effects
So instead:
Target downstream components (like isoforms) for specificity
One epinephrine molecule binds a β receptor, which activates a Gs protein. The Gs protein activates adenylyl cyclase, each adenylyl cyclase produces many cAMP molecules, each cAMP activates protein kinase A, and each PKA phosphorylates numerous downstream targets. The final effect is the release of thousands of glucose molecules from glycogen: all triggered by a single receptor-binding event.
What’s an effector?
A molecule that produces the functional cellular response
Ex: The kidney is the effector of ADH —> increases water reabsorption
Examples:
Enzymes (glycogen phosphorylase)
Kinases (PKA, PhK)
Ion channels
👉 It’s the “output” of the signaling pathway
What targets 5-adenyl cyclase and why does it work?
Drug: Forskolin
MOA: Activates adenyl cyclase specific to cardiac tissue (analog of CAMP) —> strongly enhances heart muscle contractility and improves cardiac output, particularly in treating congestive heart failure.
Adenylyl cyclase synthesizes cyclic AMP and has 9 different isoforms that differ depending on tissue distribution (so it should be part of the epinephrine pathway you should drug!)
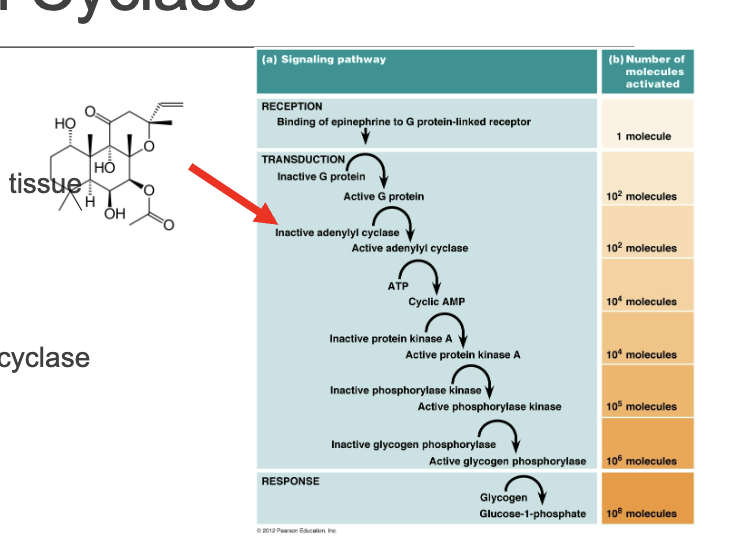
What targets PKA and why does it work?
Drug: Metformin
MOA: Activates AMPK in the liver and skeletal muscle, ultimately lowering insulin resistance = making your body’s cells more responsive to insulin allowing them to effectively take sugar from your blood for energy —> reduces high blood sugar & reduces stress put on pancreas falsely secreting more insulin when you have an effector problem —> help prevent type 2 diabetes
PKA = cAMP-dependent protein kinase
Activated by cAMP
Function: phosphorylates target proteins
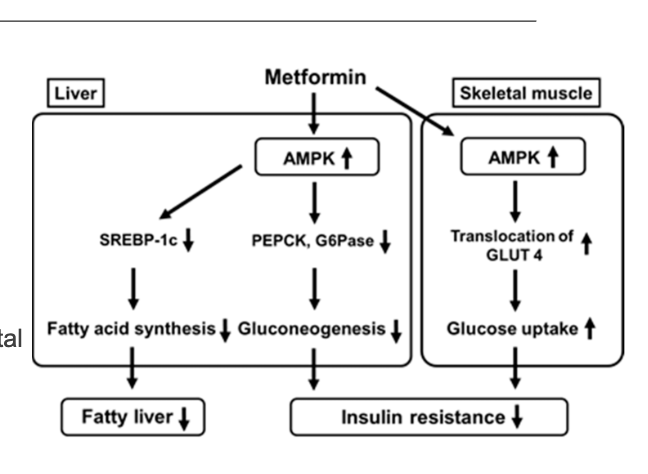
What targets PhK and why does it work?
Drug: Indirubin Analogs
PhK activates glycogen phosphorylase
Leads to glycogen → glucose breakdown
👉 Role:
Key in glycogen metabolism
Amplifies signal toward glucose release
What is one advantage and one disadvantage of targeting a point further into a signal cascade pathway?
Pro: the more specific you can get = less side effects
Con: might require higher doses because you’re often dealing with a larger signal to stop. You may also still get leakage = small activation caused by incomplete stoppage of transduction
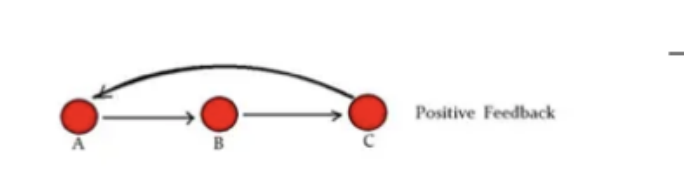
What is positive feedback?
Occur when a downstream product enhances upstream activation, often making the process difficult to stop
Considered IRREVERSIBLE because you can stop the signal, but the cascade won’t get dampened
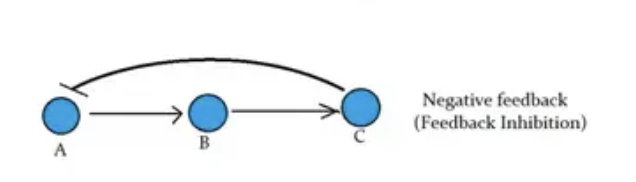
What is negative feedback?
occur when a downstream product inhibits earlier steps in the pathway, allowing regulation and reversibility. — often involved in homeostatic processes
REVERSIBLE because you can quench the signal!!
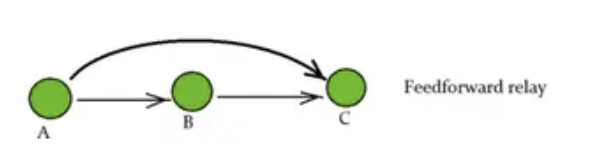
What is a feed forward relay?
where the activity of an upstream component stimulates a distant downstream component in a pathway, bypassing intermediate steps. This structure allows for rapid, far-reaching effects within a signaling network, often resulting in swift responses to external stimuli
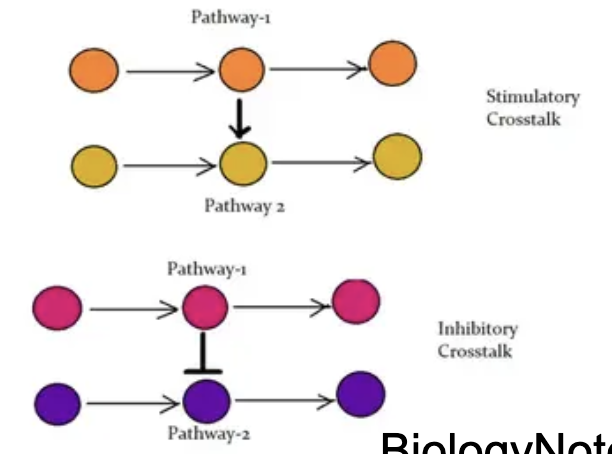
Compare stimulatory and inhibitory crosstalk.
In stimulatory crosstalk, one pathway activates another, whereas inhibitory crosstalk allows one pathway to suppress another.

Which example BEST illustrates amplification?
A. One ion channel opening per ligand
B. One ATP producing one ADP
C. One photon triggering large cGMP breakdown
D. One receptor binding one ligand
C
Amplification occurs at which levels of a signaling pathway?
A. Only at the receptor
B. Only at the effector
C. At multiple steps throughout the cascade
D. Only in the nucleus
C
Which of the following is characteristic of linear signaling?
A. Exponential amplification
B. Multiple enzyme activation steps
C. 1:1 proportional response
D. Signal multiplication
C
Which pathway is MOST likely linear?
A. GPCR → cAMP pathway
B. MAPK cascade
C. Nicotinic acetylcholine receptor
D. β-adrenergic signaling
C