PATHO2) W6- Alterations of Hormones, Part 2
1/47
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
48 Terms
Endocrine Pancreas
•The pancreas is both an endocrine and an exocrine gland
•Houses the islets of Langerhans
•Hormone-secreting cells:
•Alpha—glucagon
•Beta—insulin and amylin
•Delta—somatostatin and gastrin
•F (PP) cells—pancreatic polypeptide
Hormones of Endocrine Pancreas
Insulin
Amylin
Glucagon
Pancreatic Somatostatin
Gastrin
Pancreatic Polypeptide
Ghrelin
Insulin
•Synthesized from proinsulin
•Secretion is promoted by increased blood levels of glucose, amino acids, GI hormones
•Facilitates the rate of glucose uptake into the cells
•Anabolic hormone
•Synthesis of proteins, lipids, and nucleic acids
Amylin
•Peptide hormone co-secreted with insulin
•Delays gastric emptying
•Suppresses glucagon secretion
Glucagon
•Secretion is promoted by decreased blood glucose levels
•Stimulates glycogenolysis, gluconeogenesis, and lipolysis
Pancreatic Somatostatin
•Involved in regulating alpha- and beta-cell function
•inhibits secretion of insulin, glucagon, & pancreatic polypeptide,
Gastrin
•Stimulates secretion of Gastric Acid
Pancreatic Polypeptide
•Inhibits gallbladder contraction & exocrine pancreas secretion.
Ghrelin
Controls appetite and insulin sensitivity
Diabetes Mellitus
•Metabolic disease characterized by Hyperglycemia.
•Results from defects in Insulin secretion, Insulin action, or both
•Categories:
•Type 1
•Type 2
•Gestational
Type I Diabetes Mellitus
-most common pediatric chronic disease
-genetic and environ. factors
Types:
•Idiopathic type 1- strong genetic component, no evidence of autoimmunity
•Autoimmune type 1- slow progressive destruction of own Beta cells
Pathophysiology of Type 1 Diabetes Mellitus
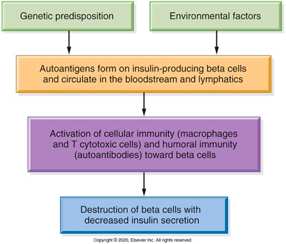
Symptoms of Type I DM
• follows long Preclinical course
•Insulin deficiency and hyperglycemia then develop (destruction of beta cells)
•Symptoms result from insulin deficiency:
•Hyperglycemia
•High glucose in urine - exceeded renal threshold —> diuresis =
Polydipsia & Polyuria
•Polyphagia
•Weight loss -Insulin deficiency causes protein & fat breakdown, contributing to weight loss and Ketoacidosis
•Fatigue
•Recurrent infections
•Prolonged wound healing
Diabetic Ketoacidosis (DKA)
•Serious complication of insulin deficiency
•More common in Type 1 (higher insulin deficient)
•Insulin normally inhibits fat catabolism: Deficiency leads to increased Ketone bodies leading to metabolic acidosis and hyperkalemia
• Symptoms: Kussmaul respirations, ketonuria, nausea, abdominal pain, & polyuria
•Managed with fluids, insulin, & electrolyte management
Type 2 DM
•Caused by genetic-environmental-lifestyle interaction
•Risk factors: family history, age, obesity, hypertension, poor diet, & physical inactivity
•Diet during pregnancy may increase long term risk for child
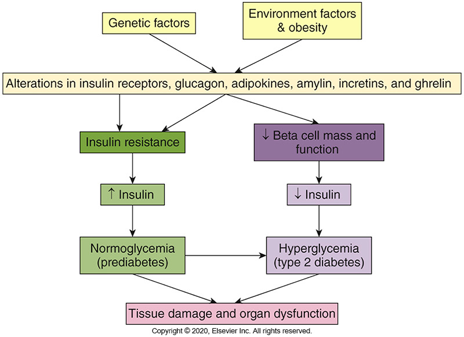
Type 2 DM (patho level)
•Initial insulin resistance = lot of glucose in blood : Compensatory hyperinsulinemia prevents clinical appearance
•Later loss of beta cells causing deficiency of insulin activity
•GI hormones play role in insulin resistance
•Incretin & Ghrelin both alter insulin resistance and secretion
•Manifestations (nonspecific): fatigue, pruritus, recurrent infections, visual changes, or symptoms of neuropathy; often overweight, dyslipidemic, and hypertensive
2 Other types of DM
•Gestational diabetes mellitus (GDM): Any degree of glucose intolerance with onset or first recognition during pregnancy
•Maturity onset diabetes of youth (MODY): Beta-cell function or insulin action affected by autosomal dominant mutations
Hypoglycemia
(lab value under 70)
•Causes: Significant change in caloric intake, exercise without insulin dosage modification, incorrect medication dosing
•Signs and symptoms: Pallor, tremor, anxiety, tachycardia, Headache, dizziness, visual disturbances, Seizures
•Treatment: Immediate glucose replacement (Oral or IV)
“cold and clammy, eat a Sammy”
15:15 rule

Hyperosmolar hyperglycemic non ketotic syndrome (HHNKS)
•Uncommon, but serious complication of T2DM
•Predisposing factors: infections, Cardiac & renal disease.
•Lesser insulin deficiency which prevents lipolysis & ketone production (low), BUT usually has higher degree of hyperglycemia
•Symptoms: Severe dehydration, Neurologic changes, Change in Level of consciousness, loss of electrolytes (Especially K+)
Chronic Complications of DM
•Microvascular disease- characterized by occlusion of capillaries with ischemia of tissues
•Diabetic retinopathy
•Diabetic nephropathy (kidney disease)
•Diabetic neuropathies
•Macrovascular disease:
•Cardiovascular disease, Stroke, Peripheral vascular disease
Individuals with diabetes are at increased risk of infection because…
1. Senses
•Loss of sensation
2. Hypoxia
•Glycosylated HGB reduces blood flow to tissues
3. Pathogens
•Hyperglycemia causes certain pathogens to proliferate
4. Blood Supply
•Vascular changes affect blood flow
5. Suppressed immune response
•Hyperglycemia affects innate and adaptive immune responses.
Adrenal Glands
•Located close to upper part of each kidney
•Consist of two portions
•Adrenal Cortex
•Adrenal Medulla
= separate, but interrelated glands
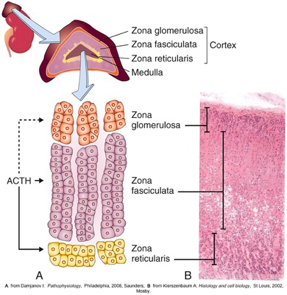
Adrenal Cortex
-80% of adrenal gland’s total weight
-3 zones
-stimulated by ACTH
-hormones are synthesized from cholesterol
Hormones of Adrenal Cortex
Glucocorticoids
Mineralcorticoids
Adrenal estrogens nd androgens
Glucocorticoids
•Direct effects on carbohydrate metabolism
•Suppress immune and inflammatory reactions
•Most potent naturally occurring glucocorticoid is cortisol
Mineralcorticoids
•Affect ion transport by epithelial cells
•Most potent naturally occurring mineralocorticoid is aldosterone (Regulated by the renin-angiotensin system)
Adrenal Estrogens and androgens
•Minimal amounts secreted
•Androgens are weak
•Converted by peripheral tissues to stronger androgens such as testosterone
•Peripheral conversion is enhanced by aging or obesity
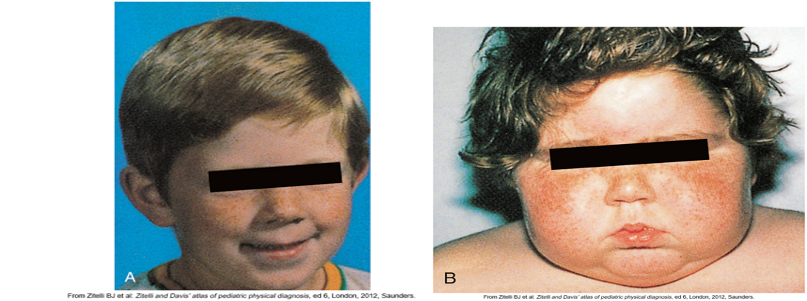
Disorders of Adrenal Cortex - Cushing’s
•Hypercortical function:
•Cushing syndrome- manifestations resulting from chronic excess cortisol, regardless of cause
•Cushing disease- excessive anterior pituitary secretion of ACTH
•Manifestations:
-Weight gain in trunk (“truncal obesity”)
-facial (“moon face”)
-cervical areas (“buffalo hump”)
•Glucose intolerance & protein wasting
Disorders of Adrenal Cortex: CAH & Hyperaldosteronism
•Congenital adrenal hyperplasia
•Deficiency in cortisol production causing increase in ACTH concentration
•Hyperaldosteronism:
•Primary hyperaldosteronism (Conn syndrome)
•Increased aldosterone secretion from abnormality in adrenal cortex
•Secondary hyperaldosteronism:
•Extra-adrenal stimulus of aldosterone secretion
•Manifestations:
•Hypertension, hypokalemia, and hypervolemia
Hypersecretion of Adrenal
•Hypersecretion of adrenal androgens
•Feminization (female secondary sex characteristics)
•Hypersecretion of adrenal estrogens
•Virilization (male secondary sex characteristics)
Addison’s Disease
•Primary adrenal insufficiency:
•Autoimmune - self destruction of adrenal cortical cells —> low cortisol and aldosterone
•High levels of ACTH- loss of normal feedback loop
•Signs: fatigue, hyperpigmentation (bronze), n/v, diarrhea
•Addisonian crisis—hypotension leading to vascular collapse & shock
•Diet should contain 150mEq of sodium per day, but low potassium
carry sodium tablet + medical bracelet
•Secondary hypocortisolism:
•Decreased ACTH (stimulation) —> adrenal atrophy
•Often results from prolonged exposure to exogenous glucocorticoids
Adrenal Medulla
•Adrenal medulla = space in middle
•Chromaffin cells (pheochromocytes) secrete and store catecholamines which turn into epinephrine and norepinephrine
•Release of catecholamines has been characterized as a fight or flight response
•Catecholamines promote hyperglycemia (innervated by the sympathetic nervous system)
Tumors of the Adrenal Medulla
•Adrenal medulla hyperfunction caused by:
1) Pheochromocytomas (tumors derived from chromaffin cells)
2) Sympathetic paragangliomas
•Tumors cause excessive production of catecholamines:
•Side effects = stimulation of fight or flight response
HTN, Headache, tachycardia, Sweating, & glucose intolerance.
Adipose Tissue
•Functions: Insulation, Mechanical support, Secretes adipokines, Immune cell function, Energy reserve
•Adipocytes = Fat storing cells
Adipose Tissue is a part of you
Endocrine Organ
•Adipocytes secrete adipokines
•Adipokines: Cell signaling proteins, Function like hormones, Include all biologically active substances synthesizes by White Adipose Tissue (WAT)
•Excess white adipose tissue (WAT) —> dysregulation of secretion + function of adipokines
Regulation of Food Intake and Energy Balance
•Controlled by central and peripheral physiological signals
•Arcuate nucleus in the hypothalamus balances opposing effects of neurons
•Orexigenic neurons: promote appetite, stimulate eating, decrease metabolism
•Anorexigenic neurons: suppress appetite, inhibit eating, increase metabolism
•GI tract secretes hormones that control hunger and satiety
Obesity (what is it & risk factors)
•An increase in body adipose tissue
•Body mass index greater than 30 kg/m2 for adults
•Greater than the 95th percentile on growth charts for children
•Develops when caloric intake exceeds caloric expenditure in genetically susceptible people
•Risk factors:
Polygenic defects
Metabolic abnormalities
Environmental factors
Depression and mood disorders
Obesity pt. 2
•Pathophysiology:
•Interaction of peripheral and central pathways and numerous adipokines, hormones, and neurotransmitters
•Signaling mediators act on hypothalamus and brainstem to regulate hunger and satiety
•Produces state of chronic, low-grade inflammation in White Adipose Tissue (WAT):
•Insulin resistance
•Metabolic syndrome
•Other complications (increased risk for MI + stroke)
•Alterations in intestinal microbiome:
•May have a causal role in obesity
•Weight loss (bariatric) surgery = most effective treatment for decreasing obesity-related morbidity
Phenotypes of Obesity
Visceral Obesity:
•Distribution of body fat is localized around the abdomen and upper body
•“Apple shape” 🍎
•Associated with more obesity complications than peripheral
Peripheral Obesity:
•Distribution of body fat is extraperitoneal and distributed around the thighs and buttocks
•“Pear shape” 🍐
Normal Weight Obesity:
•Normal body weight and BMI with percent of body fat greater than 30%
Metabolically Healthy Obesity:
•Obese but have no metabolic-obesity associated complications and decreased risk for morbidity and mortality
Starvation
•Decreased energy intake leading to weight loss —>
•Malnutrition
•inadequate amounts of calories, protein, vitamins, or minerals
•Caused by diet, alterations in digestion, or disease
Types of Starvation : Short Term
•Short-term
•Extended fasting, several days of dietary abstinence or deprivation
•Therapeutic: initial rapid weight loss
•Body responds to protect protein mass
Types of Starvation: Long Term
Long-term…
Begins after several days of dietary abstinence
Therapeutic: weight loss in morbidly obese people
Pathologic: poverty, disease, and anorexia nervosa
Causes death from proteolysis
Types of Starvation Other
•Marasmus—protein energy malnutrition
•Kwashiorkor—protein deprivation with carbohydrate intake
•Cachexia—muscle wasting from cancer
•Refeeding syndrome (Box 21.4)
Can lead to life threatening dysrhythmias due to shifting of electrolytes
Reinstitute feedings slowly (20kcal/kg/day)
Sarcopenia
•Age-related loss of skeletal muscle & muscle strength
•Thought to be secondary to progressive muscular changes and decreased anabolic hormones, more fat, risk of falls
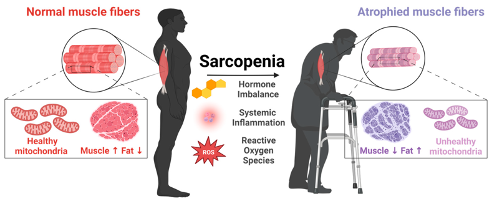
Anorexia of Aging
•Decrease in appetite or food intake
•decreased orexigenic signals and increased anorexigenic signals
•Risk factors:
•Functional impairments
•Medical and psychiatric conditions
•Loneliness and grief; social isolation
•Abuse or neglect
Anorexia of Aging Pt. 2
•Undernutrition leads to adverse outcomes:
•Malnutrition
•Frailty
•Mitochondrial dysfunction
•Reduced regenerative capacity
•Increased oxidative stress
•Imbalanced hormones
•Treatment is supportive