BIOC 406 Lecture 4
1/101
Earn XP
Description and Tags
Regulation of Fatty Acid Metabolism
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
102 Terms
Acetyl-CoA can be used for…
energy
TCA-cycle to make ATP (most tissues)
ketone bodies (liver)
storage/synthesis
fatty acids (liver, adipose tissues)
cholesterol (liver)
What determines what acetyl-CoA will be used for?
Acetyl-CoA carboxylase (ACC): catalyzes committed step for fatty acid synthesis
What stabilizes the inactive conformation of the biotin carboxylase (BC) domain of the ACC?
reversible phosphorylation
What does increasing citrate do to ACC?
Increasing citrate activates ACC and drives ACC polymerization (making enzyme activity even better)
What does palmitoyl-CoA do to ACC?
Palmitoyl-CoA inhibits the ACC
What is the hormonal control of ACC?
PKA and AMPK can phosphorylate ACC, which will inhibit it
fatty acid mobilization [low energy conditions]
glucagon & epinephrin –> ACC phosphorylation
low cellular energy state –> activate AMPK –> ACC phosphorylation
How do we dephosphorylate the ACC?
insulin –> signaling activates PP2A (phosphatase) –> ACC dephosphorylation –> activates ACC
What are three different ways palmitoyl-CoA negatively regulates cellular fatty acid synthesis?
promotes ACC filament disassembly
inhibits the citrate transporter (citrate can no longe be transported OUT of the mitochondrial matrix)
inhibits glucose-6-phosphate dehydrogenase (reduces flux through pentose phosphate pathway – a source of NADPH for fatty acid synthesis)
What is the result of enzymes forming different filaments?
It can change the activity of the enzyme (can stabilize active/inactive enzyme state)
long platform for signaling molecules & regulation
What does dynamic assembly and disassembly of filaments enable in response to changing cellular or environmental cues?
rapid response
coordination of cellular activity over large distances
How does insulin regulate fatty acid metabolism?
After a meal: increase in blood glucose –> insulin release; signal to synthesize and store fat
What is the first effect of insulin on glucose transport shown in the diagram?
increase in glucose uptake by GLUT4
How does insulin affect the Pyruvate Dehydrogenase (PDH) complex?
Insulin activates PDH Phosphatase, which removes a phosphate group from PDH to activate it. This increases the flux of pyruvate into acetyl-CoA.
What is the "gatekeeper" protein that insulin indirectly inhibits to stop beta-oxidation?
CPT1 (Carnitine Palmitoyltransferase 1)
Explain the mechanism by which insulin blocks fatty acids from entering the mitochondria for beta-oxidation
Insulin increases the production of Malonyl-CoA. Malonyl-CoA acts as an inhibitor of CPT1, preventing fatty acids from being transported into the mitochondria for beta-oxidation.
Why is Malonyl-CoA's inhibition of CPT1 biologically necessary after a meal?
It prevents a "futile cycle" where the cell would be trying to synthesize fat (palmitate) and burn it (beta-oxidation) at the same time.
How does insulin activate Acetyl-CoA Carboxylase (ACC)?
Insulin activates PP2A (Protein Phosphatase 2A), which dephosphorylates ACC, shifting it from its inactive form to its active (often filamented) form.
What is the role of Citrate Lyase in this pathway, and how does insulin affect it?
Citrate Lyase breaks down citrate (which has exited the mitochondria) into acetyl-CoA in the cytosol. Insulin increases its activity.
What effect does insulin have on cAMP levels?
Insulin activates cAMP phosphodiesterase, which breaks down cAMP into AMP. This generally lowers the signaling for catabolic (breakdown) processes.
What two factors determine the rate of beta-oxidation?
availability of fatty acids
transport into mitochondria (controlled by CPT1)
What are mobilized fatty acids going to be used for in most tissues?
to generate ATP
What are mobilized fatty acids going to be used for in the liver?
generate ATP and ketone bodies
name 3 different ketone bodies
acetone, acetoacetate, beta-hydroxybutyrate
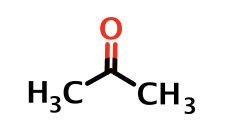
acetone
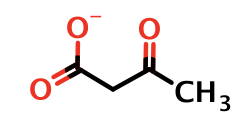
acetoacetate
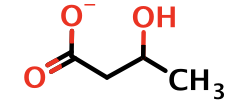
beta-hydroxybutyrate
What are the three primary ways the liver maintains blood glucose and energy levels during a fast?
mobilize glycogen stores (breakdown glycogen & release glucose)
synthesize glucose (gluconeogenesis)
Ketogenesis (generates and distributes ketone bodies)
Why does the liver produce ketone bodies specifically for "other" tissues?
To spare glucose for the brain, which has "first call" on circulating glucose.
What property of ketone bodies allows them to be easily distributed throughout the body?
They are water-soluble. Unlike fatty acids, they don't require transport proteins (like albumin) to travel in the blood.
Beyond sparing glucose, what is a major survival advantage of using ketone bodies during starvation?
They reduce the reliance on protein (muscle) as a carbon source for gluconeogenesis, preventing excessive muscle wasting.
How much energy do ketone bodies provide during an overnight fast versus a 3-day fast?
Overnight fast: 2-6% of energy requirements.
3-day fast: 30-40% of energy requirements.
Approximately how many grams of ketone bodies can a healthy adult produce per day during fasting?
185 g/day
How does ketone body dependency vary with age?
KBs accumulate rapidly in newborns & infants, but as we age its takes longer to produce circulating KBS
Why does a fast promote ketone body production in the liver regarding Oxaloacetate (OAA)?
During a fast, OAA is diverted away from the Citric Acid Cycle to be used for gluconeogenesis. Without enough OAA to pair with Acetyl-CoA, the Citric Acid Cycle slows down.
What causes Acetyl-CoA to accumulate in the mitochondria during a fast or low-carb diet?
High rates of beta-oxidation (fat burning) produce massive amounts of Acetyl-CoA, but it cannot enter the Citric Acid Cycle because OAA levels are depleted.
List three physiological states that trigger the production of ketone bodies.
1. Fasting
2. Diabetes (uncontrolled)
3. Low-carb diets
What is the first intermediate formed when two molecules of Acetyl-CoA condense at the start of ketogenesis?
Acetoacetyl-CoA
Between Acetoacetate and beta-hydroxybutyrate, which is described as "more stable"?
beta-hydroxybutyrate.
What happens to Acetoacetate if it is not converted to beta-hydroxybutyrate?
It is chemically unstable and can spontaneously break down into CO2 and Acetone.
What cofactor is required to convert Acetoacetate into beta-hydroxybutyrate?
NADH (it is a reduction reaction, converting a ketone to an alcohol).
When does ketogenesis in the liver occur?
beta-oxidation exceeds energy requirements of liver and/or
not enough carbohydrates to maintain oxaloacetate levels
What organs use ketone bodies?
muscles & kidney; brain during starvation; NOT LIVER
What is the first step in utilizing $\beta$-hydroxybutyrate for energy in extra-hepatic tissues?
t is oxidized back into acetoacetate, a reaction that produces one molecule of NADH + H+
Which specific enzyme is responsible for activating acetoacetate into acetoacetyl-CoA?
beta-ketoacyl CoA transferase
What molecule acts as the CoA donor for the activation of acetoacetate?
Succinyl-CoA (from the TCA cycle), which is converted to succinate in the process.
What is the final enzyme in the pathway, and what are the final products?
Thiolase. It uses a free HS-CoA to cleave acetoacetyl-CoA into two molecules of acetyl-CoA, which then enter the TCA cycle.
True or False: The liver can use the ketone bodies it produces for its own energy needs.
False. The liver lacks the enzyme beta-ketoacyl CoA transferase. This ensures that the liver exports ketone bodies to other tissues rather than consuming them itself.
How does the export of ketone bodies benefit the liver's own internal metabolism?
allows liver to continue beta-oxidation
no build up of acetyl-CoA
support energy needs of liver
How many molecules of Acetyl-CoA are produced from one molecule of acetoacetate?
Two.
What is cholesterol an essential component of?
eukaryotic cell membranes
cholesterol esters
transported to other tissues (via lipoproteins)
storage in lipid droplets
What 3 molecules is cholesterol a precursor for?
in liver, makes bile acids (cholate), aids in digestion soft dietary lipids
steroid hormones (signaling molecules)
Vitamin D
What are the four primary ways the liver obtains cholesterol?
1. Diet: Delivered via chylomicron remnants.
2. Import/Recycle: Taking up IDLs and LDLs from the blood.
3. Reverse Transport: Collected from extra-hepatic tissues via HDL.
4. De Novo Biosynthesis: Synthesizing it itself.
Which lipoprotein specifically delivers dietary cholesterol to the liver?
Chylomicron remnants. (Dietary fats are first packaged into chylomicrons in the intestine, then processed in capillaries before the remnants reach the liver).
Once the liver has processed cholesterol, in what form does it export it to the rest of the body?
As VLDL (Very Low-Density Lipoprotein).
What is the primary function of the Reverse Transport pathway?
To pick up excess cholesterol from extra-hepatic tissues and return it to the liver using HDL.
primary site of cholesterol biosynthesis
liver
secondary cholesterol biosynthesis sites
intestinal epithelial
tissues that synthesize steroid hormones (adrenal cortex, ovaries, testes)
Where do cholesterol reactions take place?
cytosol & ER
soluble compounds early on (cytosol) to large hydrophobic compounds (ER)
Biosynthesis of cholesterol (TLDR)
3 acetyl-CoA –> mevalonate –> isopentyl-pyrophosphate –> squalene –> cholesterol
In which cellular compartment does the synthesis of mevalonate occur?
cytosol
What are the two primary requirements for a cell to begin synthesizing cholesterol?
1. Availability of metabolic precursors (acetyl-CoA).
2. A high energy state (a lot of energy available).
Sequence the intermediates in the pathway from Acetyl-CoA to Mevalonate.
1. Acetyl-CoA (2 molecules)
2. Acetoacetyl-CoA
3. HMG-CoA (beta-hydroxy-beta-methylglutaryl-CoA)
4. Mevalonate
Which enzyme catalyzes the committed step and serves as the primary regulatory target in cholesterol biosynthesis?
HMG-CoA reductase.
What reaction does HMG-CoA reductase catalyze, and what cofactors are required?
It reduces HMG-CoA to Mevalonate, requiring 2 NADPH as the reducing agents.
Name the enzymes responsible for the first two steps of this pathway.
1. Acetyl-CoA Acetyl Transferase (condenses 2 acetyl-CoA).
2. HMG-CoA synthase (adds a third acetyl-CoA unit).
How many carbons are in one molecule of Mevalonate?
6 carbons. (It is formed from three 2-carbon acetyl-CoA units).
What is the basic 5-carbon structural unit used to build cholesterol?
The isoprenoid unit (isoprene).
What are the two "activated" 5-carbon isoprenoids formed in this stage?
1. Isopentyl pyrophosphate (IPP)
2. Dimethylallyl pyrophosphate (DMAPP)
Describe the energetic cost of converting one mevalonate into 5-pyrophosphomevalonate.
It requires the consumption of 2 ATP molecules
What major chemical change occurs to turn a 6-carbon mevalonate derivative into a 5-carbon isoprenoid?
Decarboxylation. One carbon is lost as CO2 (along with the release of Pi).
What is the relationship between Isopentyl pyrophosphate and Dimethylallyl pyrophosphate?
They are isomers of each other. They can be interconverted to provide the specific building blocks needed for cholesterol synthesis.
Name two molecules, other than cholesterol, that are synthesized using isoprenoid units.
1. Ubiquinone (CoQ): Essential for the electron transport chain.
2. Farnesyl pyrophosphate: Used to anchor proteins to biological membranes.
What is the structural significance of Farnesyl pyrophosphate?
It is made of 3 isoprenoid units (15 carbons total) and is used as a membrane anchor for certain proteins.
Where in the cell does 5-carbon isoprenoid synthesis take place?
The cytosol.
True or False: The synthesis of activated isoprenoids from mevalonate is an ATP-driven process.
True. It is a highly "expensive" pathway energetically, which is why it only occurs when the cell has a high energy charge.
How many carbons are in one molecule of Squalene, and how many isoprenoid units are required to build it?
30 carbons, built from six 5-carbon isoprenoid units.
Describe the two types of condensation reactions
1. Head-to-tail reaction: Condenses DMAPP and IPP units to form longer chains (like FPP).
2. Head-to-head reaction: Condenses two 15-carbon FPP molecules to form the 30-carbon squalene.
What is the 15-carbon intermediate formed before squalene?
Farnesyl-pyrophosphate (FPP).
Which enzyme catalyzes the final step of this stage, and where is it located in the cell?
Squalene synthase, which is bound to the endoplasmic reticulum (ER) membrane.
What reducing cofactor is required for the synthesis of squalene from FPP?
NADPH (it also releases inorganic pyrophosphate, PPi)
What is the significance of the "head-to-head" reaction in squalene synthesis?
It joins two identical 15-carbon chains (FPP) at their "heads" to create a symmetrical 30-carbon molecule, which is necessary for the eventual cyclization into cholesterol.
True or False: Squalene synthesis involves the release of pyrophosphate (PPi)
True. The release of PPi and its subsequent hydrolysis helps drive these condensation reactions forward.
In which specific cellular location does the final stage of cholesterol synthesis occur?
In the Endoplasmic Reticulum (ER).
What is the first "cyclized" intermediate formed in this pathway?
Lanosterol.
Sequence the structural transformation from squalene to cholesterol.
1. Squalene (linear 30-carbon)
2. 2,3-oxidosqualene (epoxide intermediate)
3. Lanosterol (first 4-ring structure)
4. Cholesterol (final product)
What enzyme converts squalene into the reactive epoxide intermediate?
Squalene monooxygenase.
Which enzyme is responsible for the massive structural shift from a linear chain to a ring system?
Cyclase (also known as lanosterol synthase).
Approximately how many additional enzymes are required to convert lanosterol into cholesterol?
19 more enzymes, all located within the ER membrane.
Name two important biological molecules that Konrad Bloch discovered are also derived from cholesterol.
1. Bile acids/salts 2. Sex hormones (e.g., testosterone, estrogen)
Which Nobel Prize winners are credited with deciphering the mechanisms of cholesterol and fatty acid metabolism?
Konrad Bloch and Feodor Lynen.
According to the "receipt," how many molecules of Acetyl-CoA are required to synthesize one molecule of cholesterol?
18 Acetyl-CoA. (Note: Every single carbon in the final 27-carbon structure originates from these units).
What is the total ATP and NADPH cost to produce one cholesterol molecule?
18 ATP and 16 NADPH.
Why is Oxygen listed on the receipt for cholesterol synthesis?
Oxygen molecules are required, primarily for the later stages in the ER where squalene is cyclized and modified.
Which stages of cholesterol synthesis occur in the cytosol?
1. Synthesis of mevalonate (including the committed step).
2. Synthesis of activated isoprenoids.
Which stages of cholesterol synthesis occur in the Endoplasmic Reticulum (ER)?
1. The condensation of isoprenoids into Squalene.
2. The cyclization and final conversion of Squalene –> Cholesterol.
What is the committed step of the entire cholesterol biosynthetic pathway?
The conversion of HMG-CoA –> mevalonate.
True or False: Cholesterol can be degraded by the body to be used as a source of fuel/energy.
False. Cholesterol is not a source of fuel and is not degraded for energy.