Organic Chemistry Exam 1
0.0(0)
Studied by 6 peopleCard Sorting
1/93
Earn XP
Description and Tags
Last updated 2:18 PM on 9/20/22
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
94 Terms
1
New cards
Cyclopentane
________ (108, only slight angle strain, exhibits some torsional strain, envelope form relieves some torsional strain)
2
New cards
Cyclopropane
________ (60, severe angle strain that can be alleviated by orbitals bending but this increases energy due to inefficient overlap of orbitals.
3
New cards
Alkyl Halide
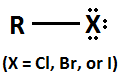
4
New cards
Alkene
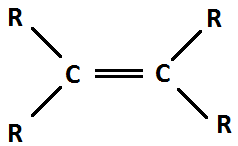
5
New cards
Alkyne
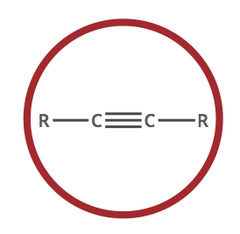
6
New cards
Alcohol
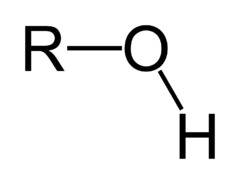
7
New cards
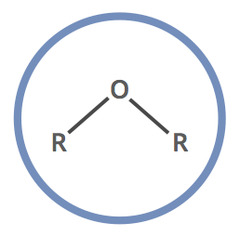
Ether
8
New cards
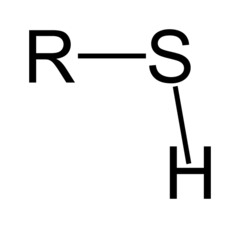
Thiol
9
New cards
Sulfide
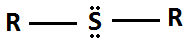
10
New cards
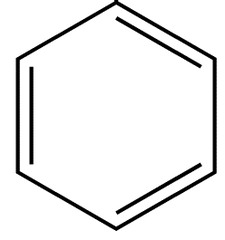
Arene
11
New cards
Ketone
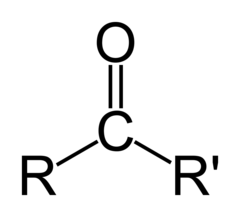
12
New cards
Aldehyde
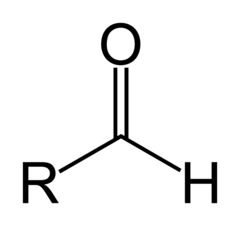
13
New cards
Carboxylic Acid
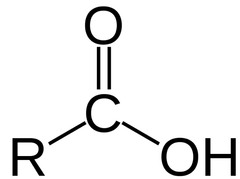
14
New cards
Acyl Halide
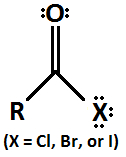
15
New cards
Anhydride
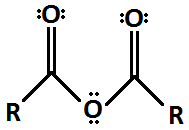
16
New cards
Ester
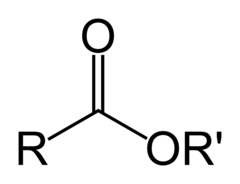
17
New cards
Amide
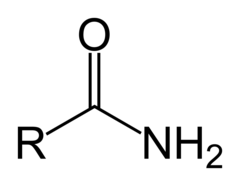
18
New cards
Amine
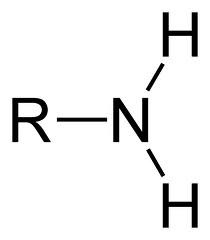
19
New cards
Valence Bond Theory
A covalent bond is formed from the overlap of atomic orbitals. It is also the sharing of electron density.
20
New cards
Atomic Orbital Theory
Overlap of electron cloud
21
New cards
Molecular Orbital Theory
Linear combination of atomic orbitals. Molecular Orbitals are associated with an entire molecule. Treats bonding electrons as being part of the entire molecule.
22
New cards
Antibonding Molecular orbital
Destructive interference, higher energy orbital
23
New cards
Bonding Molecular Orbital
Constructive Interference, lower energy orbital
24
New cards
Hybridization
Steric Number: 2, 3, 4
Steric Number: 2, 3, 4
4-109.5, sp3, 1 sigma bond 3 pi bonds
3-120, sp2, 1 sigma bond 2 pi bonds
2-18-, sp, 1 sigma bond 1 pi bond
3-120, sp2, 1 sigma bond 2 pi bonds
2-18-, sp, 1 sigma bond 1 pi bond
25
New cards
Hybridization
Specific orbitals available for bonding
26
New cards
Bond Strength versus Length
Strongest to Weakest: triple, double, and single
Longest to Shortest: single, double, and triple
Longest to Shortest: single, double, and triple
27
New cards
Sigma Bonds
Head to head over lap of s orbitals
28
New cards
Pi Bonds
Side to Side overlap of p orbitals. They prevent bond axis from rotating, meaning double bonded carbons have no isomers involving rotation. Triple bonds also cannot rotate but it doesn't matter because of their linear geometry.
29
New cards
Molecular Formula
No structure info, just lists the molecule number and their amount
30
New cards
Lewis Structure
Draw all bonds
31
New cards
Partially Condensed Structure
C-H bonds are not explicitly drawn, all other bonds and lone pairs are drawn
32
New cards
Condensed Structure
Single bonds are not drawn. Groups of atoms are clustered together.
33
New cards
Heteroatoms
Atoms other than carbon and oxygen
34
New cards
Formal Charge
Number of valence electrons - (bonds + number of lone pairs)
35
New cards
Hydrocarbon
Contains only C and H
36
New cards
Alkyl Group
Straight branch chain of C and H atoms
37
New cards
R
x
Z
x
Z
R : Rest of molecule
X : Represents common halogen
Z Represents any group of atoms
X : Represents common halogen
Z Represents any group of atoms
38
New cards
Wedge
Dash
Curvy Line
Dash
Curvy Line
Wedge : Represents a group coming out of the page
Dash : Represents a group going behind the page
Curvy Line : 50% behind and 5-% in front
Dash : Represents a group going behind the page
Curvy Line : 50% behind and 5-% in front
39
New cards
Acyclic
Cyclic
Bicyclic
Cyclic
Bicyclic
Acyclic : No rings
Cyclic : One ring
Bicyclic : two rings
Cyclic : One ring
Bicyclic : two rings
40
New cards
Parent Chain
Select the longest chain to be the parent chain. If two are the same, choose the on with more substituents.
41
New cards
Naming Substituents
add -yl to parent name for substituent name.
42
New cards
Misconceptions
-Prefixes are not used to alphabetize except for iso
-No formula for calculating possible isomers
-No formula for calculating possible isomers
43
New cards
Isopropyl
1-methylethyl
44
New cards
Sec-butyl
1-methylpropyl
45
New cards
isobutyl
2-methylpropyl
46
New cards
tert-butyl
1,1-Dimethylethyl
47
New cards
isopentyl or isoamyl
3-methylbutyl
48
New cards
neopentyl
2,2 - dimethylpropyl
49
New cards
Assembling Names
-Number all carbon atoms
-First substituent receives lower number, with a tie, the 2nd should be as low as possible
-Further tie, assign alphabetically
-First substituent receives lower number, with a tie, the 2nd should be as low as possible
-Further tie, assign alphabetically
50
New cards
Naming Bicyclic
Bicycloalkane : two rings connected to one another
Identify two bridgeheads
Start numbering from bridgeheads, longest to shortest
Identify two bridgeheads
Start numbering from bridgeheads, longest to shortest
51
New cards
Constitutional Isomers
Must use all atoms, both structures must be real, must be different compounds. To check if you drew the same isomer twice, use IUPAC naming
52
New cards
Stability of Isomeric Alkanes
Because branched alkanes have a larger heat of combustion their bonds are stronger and more stable
53
New cards
Newman Projections
Achieved due to the rotation about the vertical axis of a c-c single bond
54
New cards
Stereoisomer
Same connectivity but different spatial arrangement
55
New cards
Degenerate
Same amount of energy
56
New cards
Staggered v. Eclipsed
Staggered :
- Possess favorable interactions between an occupied bonding and antibonding sigma orbitals(hyperconjugation)
-60 degree bond angle
-lowest in energy
-Gauche interactions between methyl groups that are right by each other in the conformation (increased energy due to steric interaction)
Eclipsed :
- In the highest energy, the largest molecules are eclipsing each other (steric interaction)
-Experience torsional strain due to the rotation
- Possess favorable interactions between an occupied bonding and antibonding sigma orbitals(hyperconjugation)
-60 degree bond angle
-lowest in energy
-Gauche interactions between methyl groups that are right by each other in the conformation (increased energy due to steric interaction)
Eclipsed :
- In the highest energy, the largest molecules are eclipsing each other (steric interaction)
-Experience torsional strain due to the rotation
57
New cards
Cycloalkanes
Ideal angle 109.5. Anything that deviates from that is angle strain. Cyclopropane ( 60, severe angle strain that can be alleviated by orbitals bending but this increases energy due to inefficient overlap of orbitals. Torsional strain due to eclipsed conformation) Cyclobutene ( 90, less angle strain but more torsional bc four sets of eclipsing hydrogens, can adopt a puckered formation without gaining strain ). Cyclopentane ( 108, only slight angle strain, exhibits some torsional strain, envelope form relieves some torsional strain ). Cyclohexane ( 120, nearly strain free. Chair and boat conformation. ). Cycloheptane ( 129 ). Cyclooctane ( 135 ).
58
New cards
Chair
No angle strain, no torsional strain
59
New cards
Half chair
Some angle strain and some torsional strain
60
New cards
Boat
No angle strain, torsional strain and steric strain because of flagpole hydrogens.
61
New cards
Axial v. equatorial
Axial : parallel to vertical axis
Equatorial : along the equator of the ring ( it is favorable to put large groups in equatorial positions because it lowers their potential energy)
Equatorial : along the equator of the ring ( it is favorable to put large groups in equatorial positions because it lowers their potential energy)
62
New cards
Wedge v Dash
Wedge : Up
Dash : Down
Dash : Down
63
New cards
Ring flip
Conversion of one chair conformation to the other by rotating all C-C bonds
64
New cards
Cis v trans
Cis : same plane
Trans : opposite plane
Trans : opposite plane
65
New cards
Comparing stability
Add up A-Values for axial positions
66
New cards
Haloalkane
Halogen as a substituent
67
New cards
Organohalide
Organic Compound Containing a hydrogen
68
New cards
Stereoisomers
same molecular formula, same connection, different 3D arrangement. Cannot convert one to the other
69
New cards
Superimposable
An object and its mirror image are identical
70
New cards
Chiral object
An object that is not superimposable
71
New cards
Achiral object
An object that is superimposable
72
New cards
A carbon is a chiral center when
It is a tetrahedral bearing four different groups. In a ring it is when the paths around the ring are different
73
New cards
Enantiomer
The non-superimposable mirror image to a chiral compound
74
New cards
Assign priority based on
the atomic number of the substituents, with 1 being the largest priority. If there is a tie, keep moving outwards until something breaks the tie
75
New cards
If priority 4 is on a dash(S and R are what)
R is clockwise and S is counterclockwise
76
New cards
If priority 4 is on a wedge(S and R are what)
R is counterclockwise and S is clockwise
77
New cards
If priority 4 is on plane
Swap 4 with the dashed substituent and then swap the other two
78
New cards
Chiral center on rings
If each side of the carbon is the same (as in there is an internal mirror plane) then it is achiral. If each side is different then it is chiral.
79
New cards
Why is there a dipole moment in trans-1,3-Dichlorocyclobutane?
Cyclobutane adopts a puckered conformation to alleviate torsional strain, causing the dipoles to not fully cancel each other.
80
New cards
Are trans or cis isomers more stable?
Trans isomers. This is because with cis isomers, the bulky atoms are trying to occupy the same space.
81
New cards
Optically Active
A compound is optically active when compounds rotate the plane of polarized light. It is a consequence of chirality
82
New cards
What's the difference in rotation between enantiomers?
Enantiomers will rotate the plane in equal amounts but in opposite directions.
83
New cards
Specific rotation of a molecule is equal to
Observed rotation/(concentration * wavelength)
84
New cards
Enantiomerically Pure
A solution containing a single enantiomer
85
New cards
Racemic mixture
A solution containing equal amounts of both enantiomers, this will be optically inactive
86
New cards
Enantiomeric Excess
Occurs when both enantiomers are contained in unequal amounts
87
New cards
Percent enantiomeric excess equals
rotation of mixture / rotation of pure enantiomer * 100
88
New cards
Enantiomer
Nonsuperimposable mirror image
89
New cards
Diastereomer
Non superimposable not mirror images (Cis trans isomers)
90
New cards
Maximum number of stereoisomers
2^n (n is the number of chiral centers)
91
New cards
Meso compounds
Compounds with a chiral center that are achiral
92
New cards
Presence of a plane of symmetry means
That a compound is achiral and optically inactive
93
New cards
Allenes
Compounds containing two adjacent c=c bonds. Since central carbon is using 2 different p orbitals, it causes a twist, causing chirality
94
New cards
Z v E
If the priority group is on the same side of the double bond, it is Z, if it is the opposite side, it is E.