Electron arrangements
1/18
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
19 Terms
How are electrons arranged?
In principal quantum shells
What is used to number the quantum shells?
Principle quantum numbers
Does the energy of the principal quantum shells increase or decrease as they increase?
Energy increases
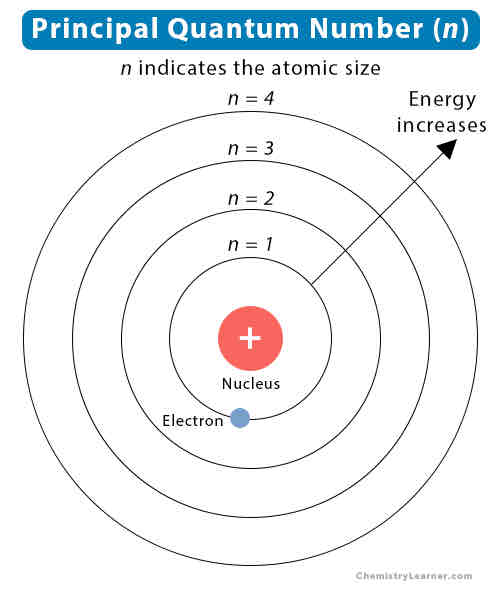
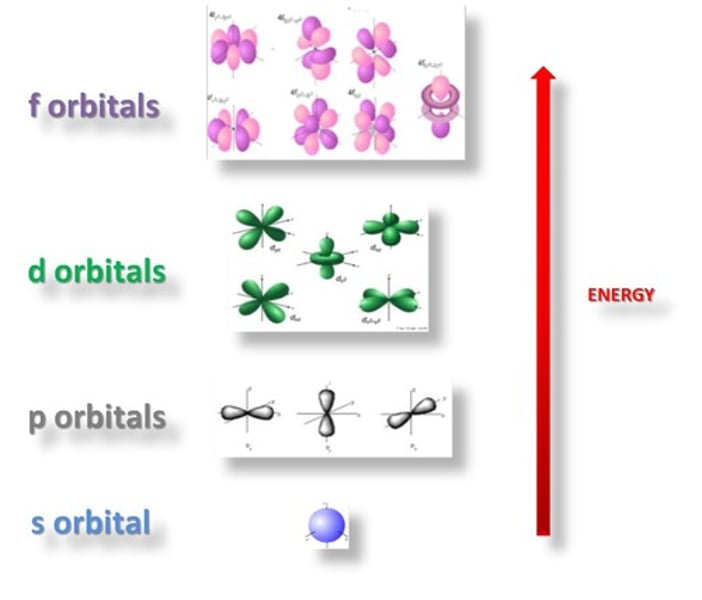
What are principle quantum shells split into?
Sub-shells
What are the sub-shells called?
s, p, d, f
What do sub-shells contain?
Orbitals
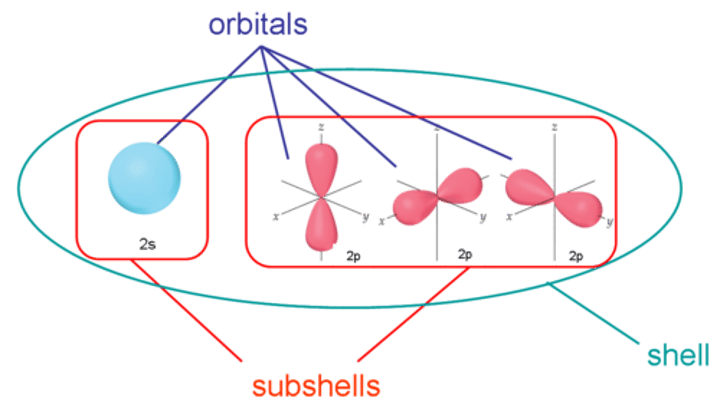
What are orbitals?
Volumes of space in which electrons are most likely to be found
What sub-shell is in the 1st energy level?
1s
What sub-shells are in the 2nd shell?
2s, 2p
What sub-shells are in the 3rd shell?
3s, 3p, 3d
What sub-shells are in the 4th shell?
4s, 4p, 4d, 4f
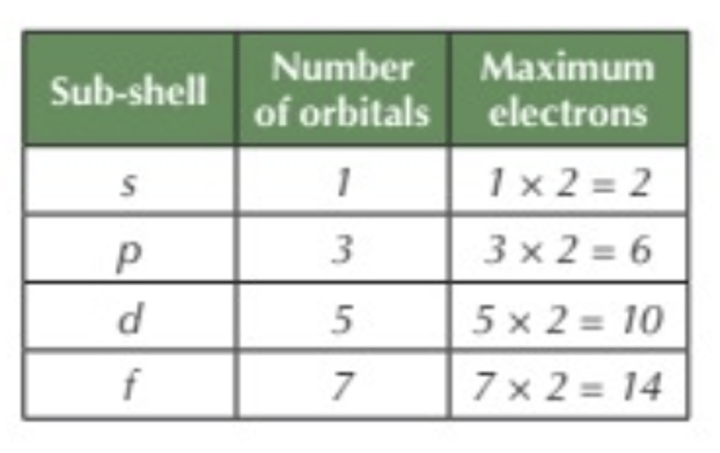
How many electrons can occupy each orbital?
2 electrons
How many orbitals are in the s sub-shell?
1 orbital
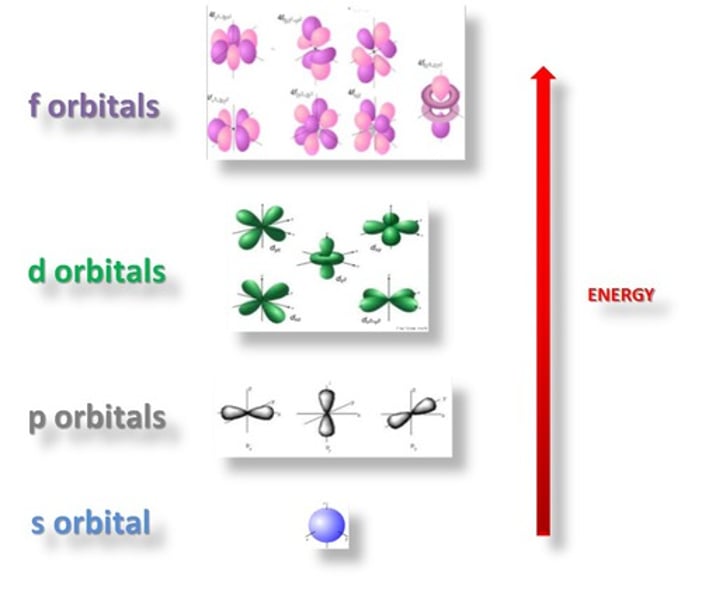
How many orbitals are in the p sub-shell?
3 orbitals
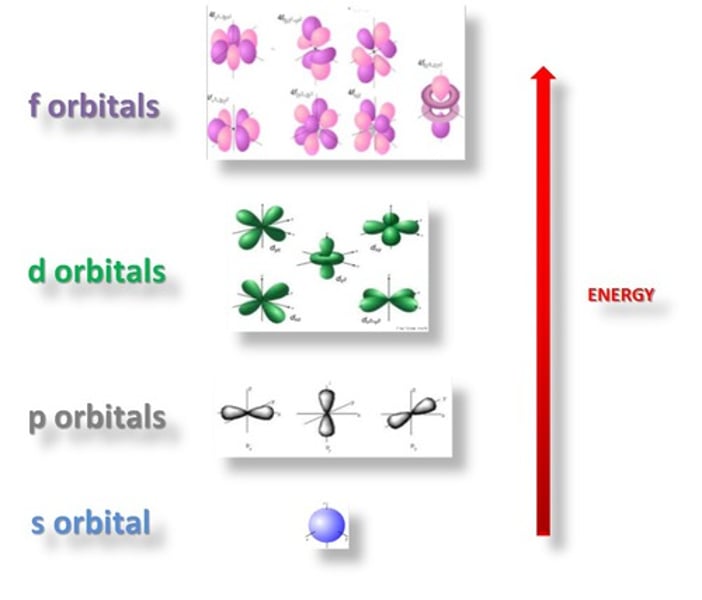
How many orbitals are in the d sub-shell?
5 orbitals
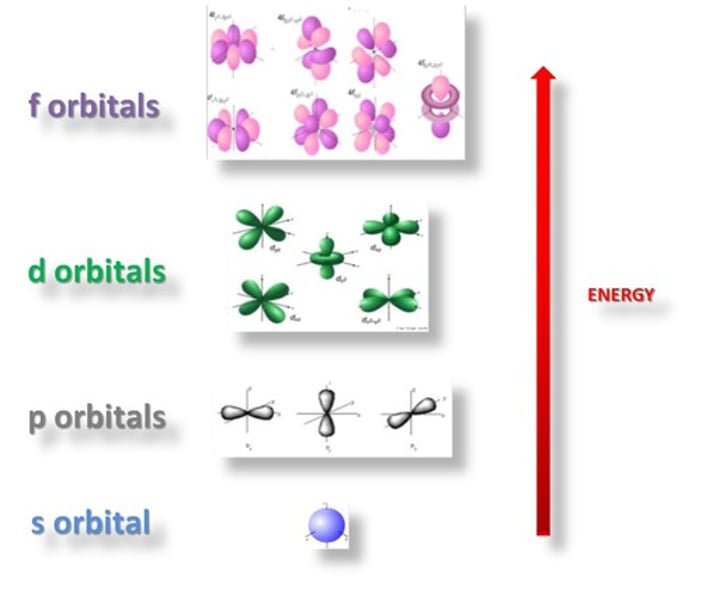
How many orbitals are in the f sub-shell?
7 orbitals
Maximum amount of electrons in each sub-shell
Order in which electrons fill orbitals

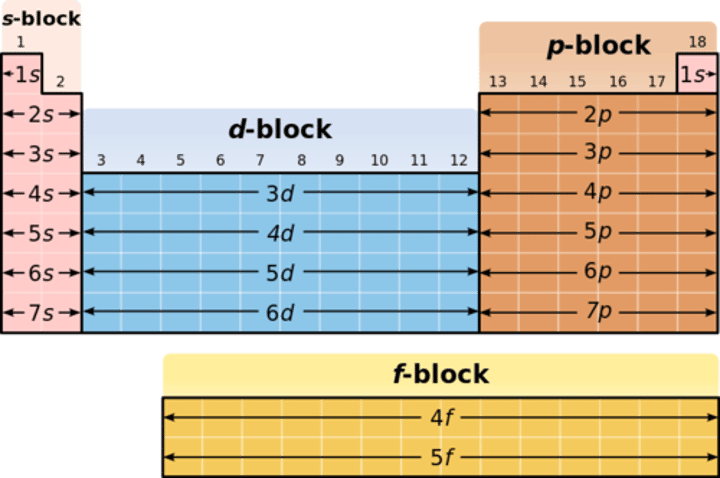
What blocks is the periodic table divided into?
Four blocks: s, p, d and f blocks