Chem Equilibrium
1/51
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
52 Terms
Reversible Reactions
A chemical reaction in which the products can react to re-form the reactions.
When are these reactions reversible?
products can reform into reactants simultaneously with the forward reaction, achieving a state of chemical equilibrium.
Dynamic Equilibrium
Amt of products & amt of reactants r constant. Its changeable.
Change scenario changes the amt. Reaction hasn’t stopped.
a state in a reversible process where the forward and reverse reactions or processes occur at the same rate, resulting in no net change in the system
What does double arrow show?
It indicates the overall reversibility of the reaction.
Requirement of =librium
Unless at least one of the substances involved escapes or is removed from the reaction system. For a reaction to reach equilibrium, it must occur in a closed system where no reactants or products can leave. Gas is being produced in container.
Products favored
When the products of the foward reaction are favored, there is a higher concentration of products than of reactants at =librium.
Smelly classroom vs ice cream :)
Equilibrium Start-Ups
When the products of the reverse reaction are favored, there is higher concentration of reactants than of products at equilibrium.
Both forward and reverse reactions occur to nearly the same extent before chemical equilibrium is established.
(Neither reaction is favored, and considerable concentrations of both reactants & products are present at =librium).
Favoring differences
Products of the forward reaction favored: This shifts the equilibrium to the right (more products form).
Products in Forward Right
Products of the reverse reaction favored: This shifts the equilibrium to the left (more reactants form).
Products in Reversed Left
******Sometimes it can be none!!!!
Equilibrium Expression
nA + mB → xC + yD
Concentrations of C & D = zero. A & B. = maximum
Over time the rate of the forward reaction decreases as A & B are used up.
Rate of the reverse reaction increases as C & D are formed.
When these 2 reaction rates become =, equilibrium is made
Graph analzation
y = rate if reaction (time it takes to be formed or lost)
Reaction rate of forward reaction decreases with time as reactants are consumed, while the reaction rate of the reverse reaction increases as products are formed, leading to equilibrium. The graph shows how the rates of both the forward and reverse reactions change over time, ultimately converging at equilibrium.
Law of Mass Action
A principle stating that at equilibrium, the rate of the forward reaction is equal to the rate of the reverse reaction, and the concentration ratio of products to reactants raised to their respective stoichiometric coefficients is constant.
The constant is K. It is independent of the initial concentrations. IT is dependent of the temperature of the system.
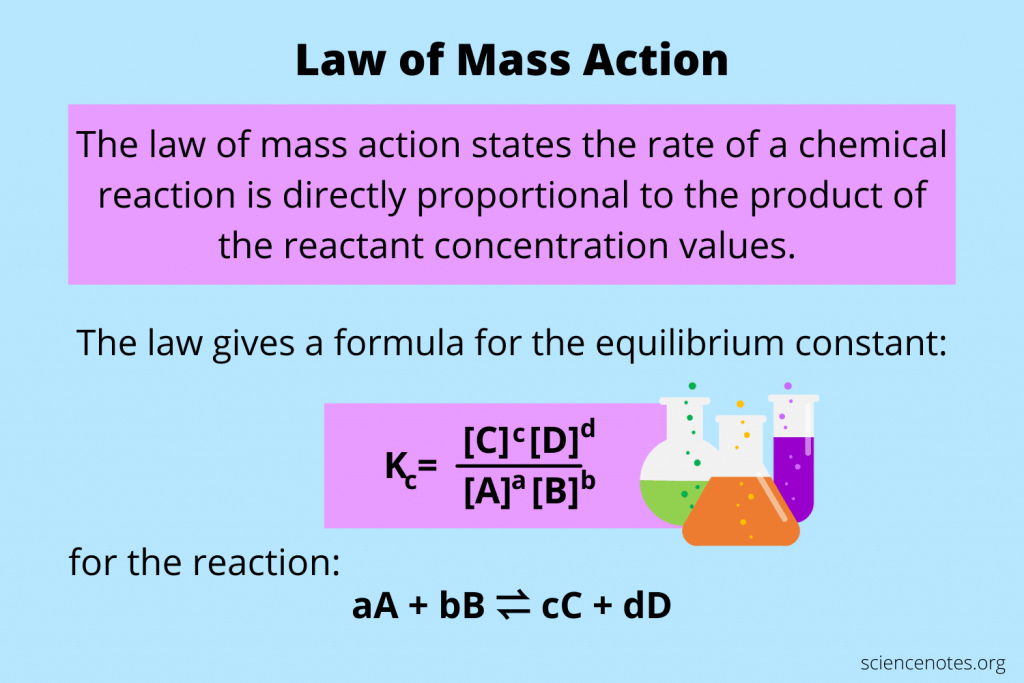
What will the problems have?
SOLIDS & LIQUIDS WILL NEVER BE IN THESE TYPE OF PROBLEMS!!!!!!!!!! THEY ARE OMITTED.
Assume acquees solutions or gases
Only substances that can actually change are included in K!!!
K size
K small → Reactants r favored
K large → Products r favored
Reaction Quotient
Which way will it go to reach equilibrium. Depends on how much substance you have.
Reaction Quotient Direction
Q > K → Reverse Reaction. Reaction shift to the left. More reactants
Reverse, Left, Reactants
K > Q → Forward Reaction. Reaction shifts to the right. More products.
Forward, Right, Products
Le’chatlier Principle.
If a system at equilibrium is stressed, it will shift in a direction that will minimize its stress.
Changing concentration (Adding/removing species)
Changing pressure/volume (Affects gas reactions)
Change temp (Shifts k itself)
Add a catalyst (Reaches equilibrium faster, but doesn’t change K)
Decreasing volume & increasing pressure
Decreasing volume & increasing pressure, gaseous equilibrium will move in the direction of fewer moles of gas, as the system seeks to alleviate the pressure increase. XXXXXXXXXXXXX
Speed of Chemical reactions. BEYOND THIS IS KINETICS.
The speed of a chemical reaction on the energy pathway that a reaction follows and the changes that take place on the molecular level when substances interact.
Equilibrium
How far a reaction goes
Problem of high KE
Only ions or molecules with very high kinetic energy can overcome repulsive forces and get close enough to react.
Reaction mechanism.
Chemical equations describe reactions, but do not show the reaction mechanism. Reaction mechanism is the step-by-step sequence of reactions by which the overall chemical change occurs.
A reaction that appears from its balanced equation to be a simple process may actually be the result of several elementary steps, each with its own transition state and activation energy.
They can check and see if everythings work as intended in a reaction mechanism by assessing the individual rate laws of each elementary step, ensuring that they align with the observed kinetics.
Intermediates
Species that appear in some steps but not in the net equation.
Homogenous Reaction
Reaction whose reactants & products exist in a single phase.
Types of Mechanism
Plausible → Steps of mechanism must add up to overall reaction
Intermediate → Produced in one of steps and consumed in the other
Collision Theory
Explaining how reactions occur. Set of assumptions regarding collisions an reactions.
Hit each other the right way/Oriented properly & with sufficient energy to form products/Hard enough.
Chemical reaction produces new bonds which are formed between specific atoms in the colliding molecules.
Activation Energy
An initial input of energy is needed to overcome the repulusion forces that occur between reactant molecules when they are brought very close together. It involves breaking reaction bonds and producing new bonds during the transformation of reactants into products.
Bond breaking vs forming
Bond breaking = Endothermic.
Bond forming = Exothermic
Activation Complex
In the brief interval of bond breakage and bond formation, the collision complex is in a transition state.
Kinetic-Molecular Theory
States that the speeds and therefore the kinetic energies of the molecules increase as the temperature increases.
Collisions between molecules must possess sufficient energy to form an activated complex on a reaction will not take place
Raising the temperature of a random reaction provides more molecules that have the necessary activation energy and cause an increase in the reaction rate.
Rate-Influencing Factors
Rate depends on particular reactants and bonds involved.
In heterogeneous reactions, the reaction rate depends on the area of the reaction substances.
Increasing surface area → more particles in contact → faster rate
Heterogenous Reaction
Involve reactants in two different phases. Slower than homo bc homo has same and those are faster.
Temperature
Increase in temp increases the average KE of the particles in a substance: this can result in a greater number of effective collisions. Increases reaction rate.
Concentration
Incerased increases paritcles that can collide, which in turn raises rate.
Catalyst
Substance that changes the rate of a chemical reaction without itself being permanently consumed. Catalysts lower the activation energy, making reactions proceed faster.
Provides alternate energy pathway in which the potential-energy barrier between reactants and products is lowered.
Catalysis
The action of a catalyst. There used, but never used up to its full potential.
Homogenous Catalyst
Catalyst in hte same phase as all the reactants and products in a reaction system
Heterogenous Catalyst
When its phase is different from that of the reactants.
Rate Law
Equation that relates reaction rate and concentrations of reactants.
The relationship between the rate of a reaction and the concentration of one reactant is determined experimentally.
K Value
Increases as the temperature increases, but the relationship between reaction rate and concentration almost always remains unchanged.
Rate Law
R = k[A]x[B]y
Powers to which a reactant concentration is raised is called the order in that reactant.
First order, 2nd order??????????
The value of n is said to be the order of the reaction with respect to A, so the reaction is said to be the “nth order in A”
Order
An order of one for reactant means that the reaction is directionally proportional to the concentration of that reactant
An order of two means that the reaction rate is directly proportional to the square of the reactant.
0 Order = Doesn’t effect the rate regardless of anything.
Sum of all the reactant orders is called the order of the reaction/overall order.
*****The orders in the rate law may or may not match the coefficients in the balanced equation.
HELLO QUIZ TMRW: Why can pure solids & liquids be ignored in Kc expressions
Their concentrations don’t change during the reaction - they’re essentially constant, so they’re absorbed into the K value.
K Info:
K > 1 → Products favored
K < 1 → Reactants favored
K = 1 → Rough mix of both
Reaction Quotietn
Q is calculated the same way as K, but using current concentrations
Q < K → Reaction shifts right
Q > K → Reaction shifts left
Q = K → Equilibrium
How does volume effect concentration?
Increasing the volume of a solution (while keeping solute amount constant) decreases its concentration, and decreasing the volume increases concentration
Temperature
Increase in temp leads to spreading of molecules. Yes, an increase in temperature causes molecules to move faster, spread apart (thermal expansion), and generally reduces the number of combined or bound molecules by weakening intermolecular forces
Pressure & moles
Increasing pressure shifts toward the side with fewer moles of gas.
Volume & pressure
Volume decreases as pressure increases. This relationship is described by Boyle's Law, stating that pressure and volume are inversely proportional for a given amount of gas at a constant temperature.
Homogenous vs Heterogeneous
Homogeneous equilibrium occurs in a single phase, while heterogeneous equilibrium involves substances in different phases.
Substance rules
Adding more substance shifts away from it
Removing substance shifts towards it
K Rules
Lots of product molecules + few leftover reactants = Big K
Lots of reactant molecules leftover = Small k
Temp & K
Endothermic → Raising temp increases K, Lowering temp lowers K
Exothermic → Raising temp lowers K, Lowering temp increases K