Lecture 2 (slide 76-153)
1/91
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
92 Terms
HER2+
gBRCA mut
bekendste moleculaire subtypes borstkanker
HER2+ similar principle as in lung cancer: activation of growth factor receptor signaling
principe HER2+ als moleculair subtype borstkanker
NEU / ERBB2
synoniemen HER2
Herceptin
Kadcyla
oudere en nieuwe therapie voor borstkanker tegen HER2+ subklasse
Trastuzumab: monoclonal antibody against HER2 receptor
wat hebben herceptin en kadcyla gemeen?
kadcyla: also emtansine (a chemotherapeutic agent), which is also released into the cell
verschil kadcyla en herceptin
the antibody acts as a carrier for the chemotherapy agent = antibody-drug conjugate (blocking effect + additional chemotherapeutic effect)
principe antibody drug conjugate (ADC)
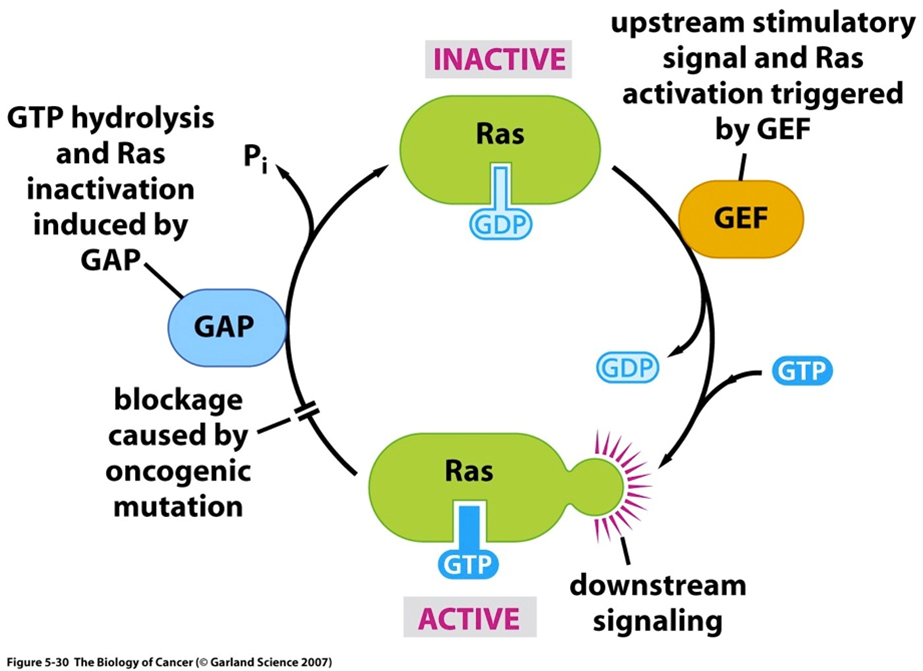
RAS cycle
guanine nucleotide exchange factor: switches RAS on
GEF
GTPase activating protein: switches RAS off
GAP
Hotspot activating mutations often occur at G12, G13 or Q61
oncogene mutaties in RAS
Oncogene
is RAS an oncogene or tumor suppressor?
NF1 (neurofibromatosis 1) is an example of a GAP, so a tumor suppressive gene
NF1 in context van RTK downstream pathways
HRAS
NRAS
KRAS
three main RAS isoforms
KRAS: very important
HRAS: not mutated at all
rol HRAS en KRAS in pancreatic cancer
KRAS has a strong preference for the codon 12 mutations
voor wat voor mutaties heeft KRAS een sterke voorkeur?
central nervous system tumors
voorbeeld tumor met weinig RAS mutaties
melanoma (skin cancer) has over 30-40% of patients with BRAF activation. So very important oncogene in skin cancer!
belangrijk oncogene in skin cancer
Know that patients with a BRAF hotspot mutation usually do not overlap with RAS mutations. Additional mutation in the same pathway gives little extra selective benefit
bestaat er overlap in BRAF en RAS mutaties?
Vemurafenib (BRAF inhibitor)
Trametinib (MEK inhibitor)
belangrijke therapieën tegen melanoma
V600E
belangrijke activating mutatie in BRAF
colorectal carcinoma
waar kan je nog BRAF mutaties vinden (buiten melanoma)?
Requirement of combination therapy
A: In the BRAF V600E cancer situation, the MAPK cascade drives proliferation with feedback regulation present
B: With BRAF inhibition (e.g. vemurafenib), mutant BRAF is blocked, but feedback release can reactivate upstream EGFR/RAS signaling and restore pathway activity, causing resistance
C: Therefore broader pathway blockade is needed, combining BRAF inhibition with EGFR and/or MEK/ERK inhibitors to suppress reactivation
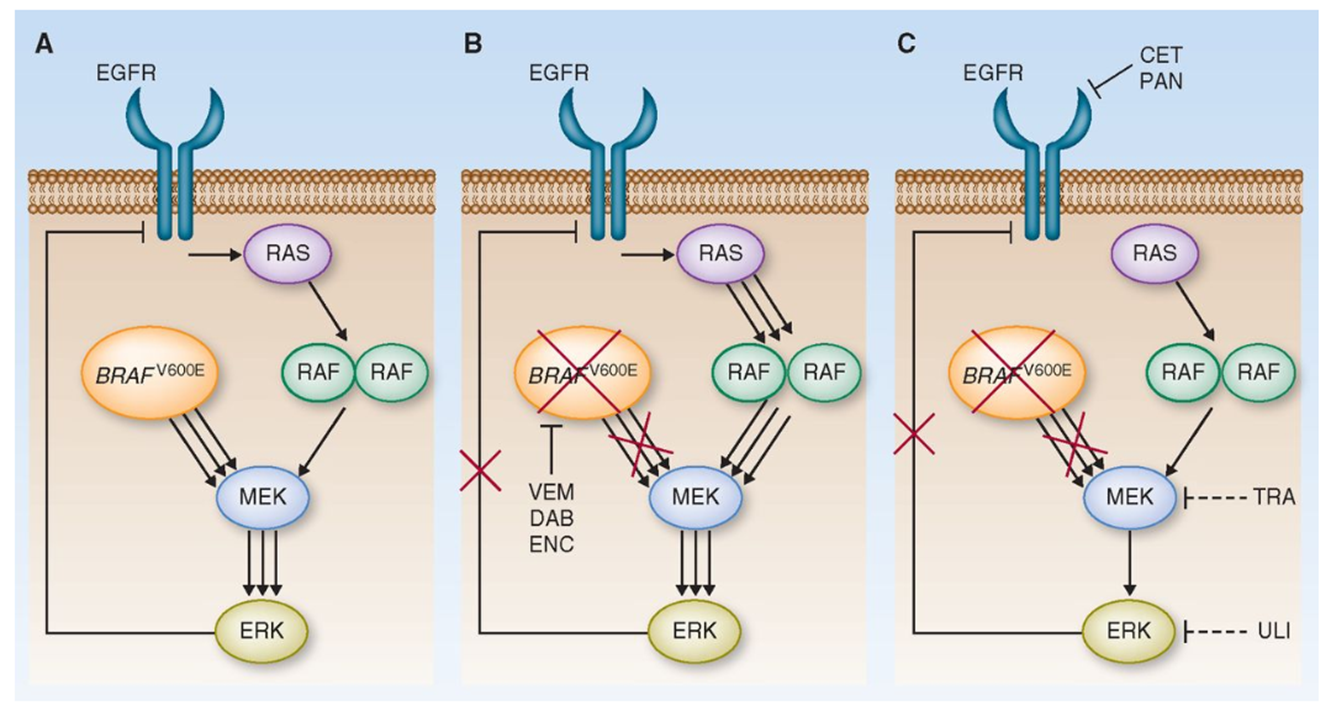
waarom is combination therapy nodig bij BRAF V600E mutated tumors?
AKT PI3K pathway
naast RAS/MAPK nog een belangrijk pathway in kanker
Phosphatidylinositol 3-kinase
PI3K
a heterodimer lipid kinase consisting of an 85-kD subunit bound to a 110-kD catalytic subunit
hoe is PI3K opgebouwd?
cell growth
effect AKT PI3K pathway
PTEN is an important tumor suppressor in this pathway, antagonizing PI3K signaling
tumor suppressor in AKT PI3K pathway
Proteus syndrome
overgrowth syndrome
Usually germline cancer predisposition involves tumor suppressor genes rather than activating oncogenes
This case is an exception: postzygotic germline/mosaic activating mutation in the PI3K/AKT pathway (often AKT1), not inherited from the parents in the classic sense
waarom is Proteus syndrome zo belangrijk in onze context?
MAX is an important dimerization partner of MYC, promoting transcriptional activation
belangrijke partner MYC
MAD can compete with MYC for MAX binding, leading to repression / reduced mitogenic signaling
MAD in context MYC en MAX
RB is a key gatekeeper of the G1/S transition. E2F transcription factors drive passage through the R-point and are normally suppressed by RB
belang RB
RB is inactivated, E2F released, cell goes past the R-point
effect phosphorylatie van RB
CDK4 is an important example in many tumor types because, together with cyclin D, it promotes progression through G1 toward S-phase
hoe gaat een cel van G1, naar de S-phase?
The extent of RB phosphorylation increases through the cell cycle, with additional kinases such as CDK2 contributing later
kinases die actief worden later in de cell cyle
which gene encodes p21?
CDKN1A
p21 is an upstream inhibitor of CDK2 (and CDKs), so acts as a tumor suppressive regulator
is p21 een oncogene of suppresor?
Cyclin-dependent kinases (CDKs) can act as oncogenic drivers when overactive
zijn CDKs oncogenes of suppressors?
CCND1
which gene encodes cyclin D1?
More Cyclin D1 (cofactor of CDK4/6) → more CDK4/6 activity → more proliferation.
wat maakt van CCND1 een oncogene?
Glioblastoma
important kind of brain tumour
drives amplifcation/deletion of cell cycle regulators
Chromothripsis can lead to amplification of EGFR and CDK4, promoting strong proliferative signaling
waarom leidt chromothripsis tot glioblastoma?
Cell-cycle control is not an isolated event: cyclin-CDK complexes are tightly linked to DNA-damage signaling
waarom is cyclin-CDK complexes niet het enige belangrijke bij cell cycle regulation?
Fusion tyrosine kinase from t(9;22), classic driver of CML.
Wat is BCR-ABL
SAC is an important checkpoint controlling progression from metaphase to anaphase, ensuring chromosomes are correctly attached to the spindle
belang SAC
Spindle assembly checkpoint
afkorting SAC
APC/C is a very important complex here. Together with cofactor CDC20, it functions as an E3 ubiquitin ligase
APC/C–CDC20 promotes degradation of securin and cyclin B. Loss of cyclin B lowers CDK1 activity
Later, APC/C with cofactor CDH1 degrades additional mitotic regulators such as FOXM1, AURKA, AURKB and PLK1, helping mitotic exit and preventing reactivation of CDK1
belang APC/C in SAC
CDK1 together with cyclin B is a key driver of mitosis. High CDK1-Cyclin B activity keeps the cell in mitosis until chromosomes are properly aligned
belangrijke driver van mitosis
Pro-mitotic transcription factor.
G2/M transition and mitosis.
Often overexpressed and drives proliferation.
wat is FOXM1 en zijn belang in kanker?
pathways with eg p53 and RB stops cells with too short telomeres (senescence), but mutations can make cells go past this checkpoint, going to crisis.
Crisis is also overcome by activating telomerase or other mechanisms (TERT complex)
hoe overkomen kankercellen te korte telomeren?
Catalytic reverse transcriptase subunit of telomerase.
wat is TERT?
RNA template used by telomerase.
wat is TERC?
p53
het belangrijkste gen dat zorgt voor senescence
They develop drugs targeting senescent cells (senolytics) and drugs targeting telomerase/telomere maintenance
Therapie tegen tumoren met verhoogde telomerase activiteit
increased transcription, gain/amplification, mutation or genomic rearrangements
Promoter mutations can create new ETS/TCF transcription factor binding sites, leading to higher TERT expression and increased telomerase activity
Enhancer hijacking
hoe kan TERT meer actief worden?
ALT (alternative lengthening of telomeres)
alternatieve methode voor telomere maintenance
telomerase-independent telomere maintenance through homologous recombination-based mechanisms
ALT does not rely on telomerase enzyme activity and is often associated with more genomic instability
=> Telomerase-dependent versus telomerase-independent telomere maintenance
mechanisme ALT (Alternative Lengthening of Telomers)
Extrachromosomal circular C-rich telomeric DNA.
wat zijn c-circles?
ALT-positive tumors.
waar vind je c-circles?
Telomerase-positive telomeres are often more heterochromatic / compact, with repressive chromatin marks
ALT-positive telomeres are relatively more open / altered chromatin and show increased structures such as G4 DNA and R-loops
chromatin difference tussen ALT- en telomerase-positives
ATRX helps maintain normal telomeric chromatin structure (linked to H3.3 deposition); loss or mutation of ATRX is frequently associated with ALT-positive tumors
belang ATRX bij ALT- en telomerase-positives
TERRA is a telomeric non-coding RNA and is often increased/associated with ALT, together with markers such as C-circles and APBs
TERRA
Nucleotide subsitution
Promotor hijacking
gene fusion
enhancer hijacking
focal amplification
disruption of insulated neighborhoods
Verschillende types van oncogene activatie
Inactivating mutations are the most important mechanism of tumor suppressor loss; enhanced degradation is less common but possible
belangrijkste mechanisme van tumor suppressor loss
Tumor suppressor genes are often linked to hereditary cancer predisposition syndromes when one mutant allele is inherited in the germline
Familial predisposition means increased risk, not certainty, of developing a certain tumor type
tumor suppressor genen en erfelijkheid
A lot of tumor suppressor genes need to have both alleles inactivated for full loss of function
Knudson two hit hypothesis
Hypophosphorylated RB = active brake; hyperphosphorylated RB = released brake
hypo- and hyperphosphorylated RB1
13q14: Retinoblastoma
In this case, the translocation places the RB1 region on the derivative X chromosome. If that derivative X becomes inactivated in a subset of cells, RB1 can be functionally silenced
Loss/inactivation of RB1 leads to retinoblastoma (a retinal eye tumor)
typische kanker door RB1 translocation, ook belangrijk geweest voor de ontdekking
Finding a disease gene based on chromosomal location.
positional cloning
p16 and 15
suppressor CDK4
p53 is a sequence-specific DNA binding protein that regulates transcription
p53
two N-terminal transactivation domains
conserved proline-rich domain
a central DNA binding domain
C terminus encoding its nuclear localization signals
oligomerization domain needed for transcriptional activity.
onderdelen p53
DNA-damage by UV → phosphorylation of p53 / changes in MDM2 → p53 increase → activates p21 → inhibits cyclin-CDK complexes → stops cell cycle for DNA-repair
cascade DNA damage by UV en p53
Ubiquitinates p53 for degradation.
rol MDM2
inhibits MDM2
=> increases p53
effect ARF
p53 is often active as a tetramer
wat voor eiwit is p53? (in zin van opbouw)
p16 (INK4A) and p14 (ARF) (both tumor suppressors)
belangrijke eiwitten komende van CDKN2A
inhibiting CDK4/6
rol INK4A
E2F is a key transcription factor that promotes G1/S cell-cycle progression by activating genes needed for DNA replication and proliferation. Normally it is restrained by RB. When E2F becomes abnormally elevated, cells sense this oncogenic signal and induce ARF, which stabilizes p53 and can trigger apoptosis or growth arrest.
on / off switch
belang E2F
Fine-tuning of p53 response.
belang PTM’s bij p53
single hit is sufficient for accelerating cancer formation
is de two hit hypothesis geldig bij p53?
~50% of tumors
komen p53 mutaties frequent voor?
>75% of mutations (>15.000 known) are missense rather then nonsense
wat voor mutaties heb je bij p53?
Partial reduction of function
hypomorphs
gain of new oncogenic function
neomorphic mutant
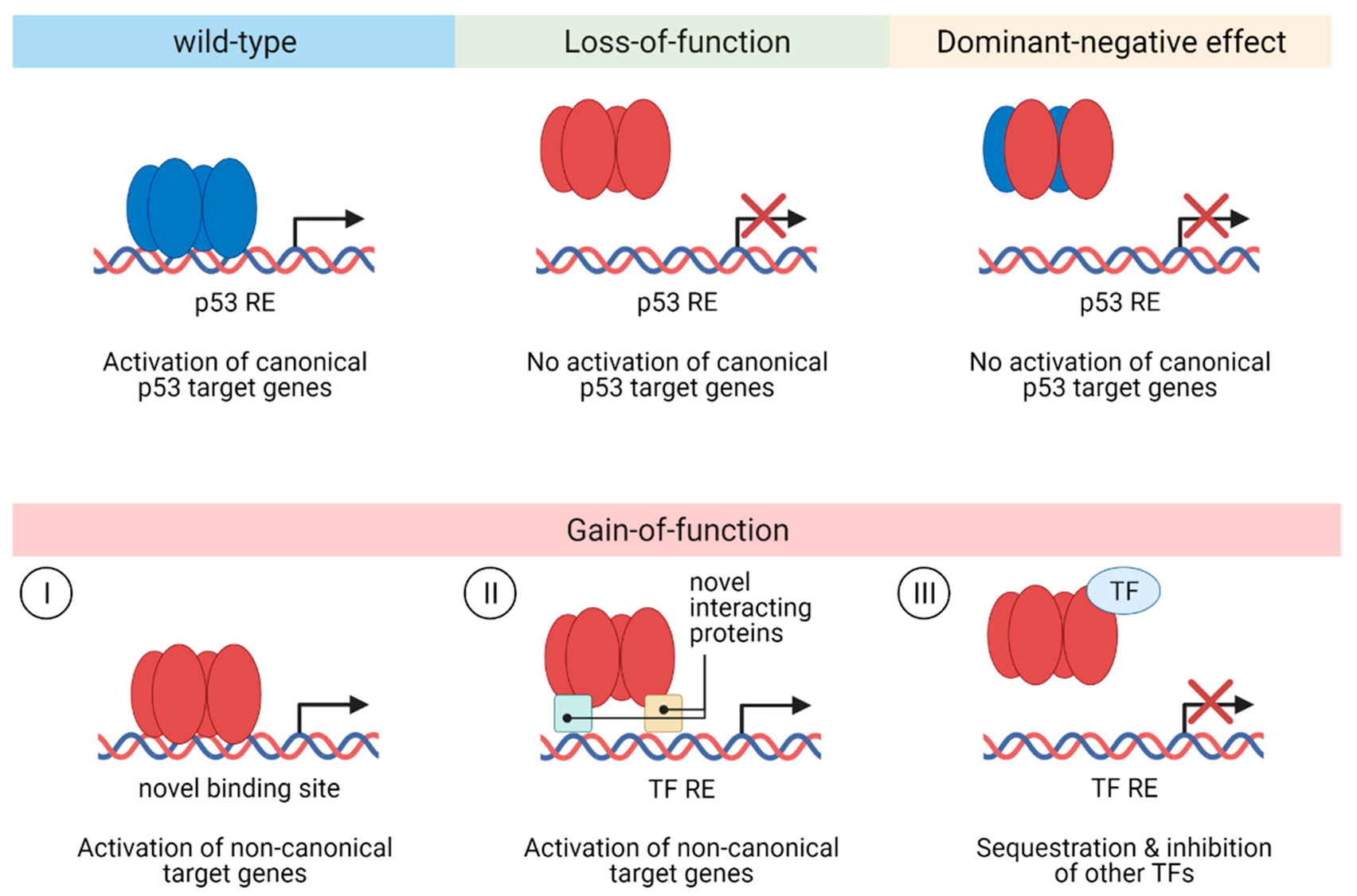
GOF present, but less frequent
Mogelijke mutaties bij p53
adenomatosis polyposis coli – colon cancer
APC (de kanker, niet het complex)
FAP (Familial Adenomatous Polyposis)
HNPCC: hereditary nonpolyposis colon cancer:
two syndromen dat zorgt voor een hoog risico in colorectal cancer
APC, it normally controls Wnt by beta-catenin, but mutation in APC leads to a lot of Wnt, so a lot of growth
gen belangrijk in FAP
germline mutations leading to mismatch repair defect
oorzaak HNPCC
Gorlin syndrome (Sonic Hedgehog pathway)
syndroom gelinkt met medulloblastoma