Amines
1/38
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
39 Terms
What is an amine?
A molecule derived from ammonia where at least one hydrogen has been replaced by an R group.
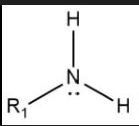
Draw the general displayed formula of a primary amine.
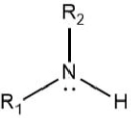
Draw the general displayed formula of a secondary amine.
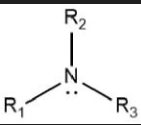
Draw the general displayed formula of a tertiary amine.
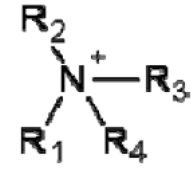
Draw the general displayed formula of a quaternary ion derived from a tertiary amine.
What is an aromatic amine?
An amine where at least one of it’s R groups contains benzene.
What is an aliphatic compound?
A compound that is not aromatic.
What can quaternary ammonium salts be used to make?
Cleaning products.
What are cationic surfactants?
Long hydrocarbon tails on some quaternary ammonium salts.
How do quaternary ammonium salts work as cleaning products?
1) The non-polar hydrocarbon tail is attracted to (non-polar) oil (grease).
2) The polar salt head is attracted to (polar) water.
3) Water and oil can now mix allowing oil (grease) to be removed from the thing that needs cleaning.
Describe and explain the polarity in a quaternary ammonium salt.
Quaternary ammonium salts have polar head (where the positive ammonium ion part is bonded to the negatively charged ion) and a non-polar hydrocarbon tail.
Why do cationic surfactants work as conditioner and fabric softener?
The positively charged ammonium ion part is attracted to negatively charged hair fibres/clothes fibres, this removes static so softens the material.
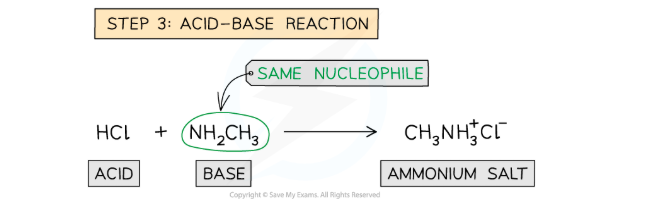
True or false? Amines are strong bases.
False. AMINES ARE WEAK BASES.
Why do amines act as bases?
They have a lone pair of electrons (on the nitrogen) that allows them to accept protons (aka act as a base).
What kind of bond forms between a proton and an amine and why?
Dative covalent bond because both electrons come from the nitrogen.
Draw the general displayed formula for the reaction between a primary aliphatic amine and a proton.
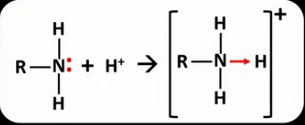
What will affect the strength of an amine as a base?
The availability of the lone pair of electrons on the nitrogen.
True or false? The higher the electron density, the more available the lone pair of electrons on the nitrogen is.
True.
Order ammonia, aromatic amines, and primary aliphatic amines (R group is alkyl group) from weakest to strongest as a base.
Aromatic amines → ammonia → primary aliphatic amines.
Why are aromatic amines the weakest bases out of all amines?
Benzene is an electron withdrawing group meaning the ring of delocalised electrons pulls electrons away from the nitrogen. This reduces electron density at the nitrogen and therefore decreases the availability of electrons at the nitrogen, so aromatic amines are less basic.
Why are primary aliphatic amines (where the R group is an alkyl group) the strongest bases out of all amines?
Alkyl groups are electron pushing groups (have positive inductive effect) meaning the electrons are pushed towards nitrogen. This means that nitrogen is more electron dense and therefore has a higher availability of electrons, so primary aliphatic amines are more basic.
Are amines nucleophiles or electrophiles? Why?
Amines are nucleophiles. They are attracted to positively charged species due to their lone pair of electrons on the nitrogen.
Give two ways in which you can make an aliphatic amine.
React a halogenoalkane with excess ammonia.
Reduce a nitrile.
Draw the mechanism for the reaction between chloroethane (a halogenoalkane) and excess ammonia. What is the name of this mechanism? Talk me through the reaction.
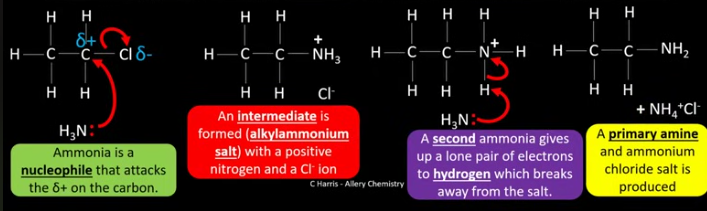
Nucleophilic substitution.
What is a disadvantage of producing aliphatic amines by reacting halogenoalkanes with excess ammonia?
Primary, secondary, tertiary, and quaternary amines are formed so the product of the reaction is impure.
Why are secondary, tertiary, and quaternary amines also formed when a halogenoalkane is reacted with excess ammonia?
Any amine formed in the initial reaction has a lone pair of electrons so will attract nucleophiles and start another reaction, it can also react with any excess halogenoalkanes.
What is the cheapest way to make primary amines in industry? What is the name of this reaction? What conditions are needed for this reaction?
Reduce nitriles using hydrogen and a nickel/platinum catalyst.
Catalytic hydrogenation.
High temperature and high pressure needed.
Why is the reduction of nitriles to produce primary amines favoured in industry?
The reaction only produces primary amines so it gives a pure product.
Give the balanced symbol equation for the reaction between a nitrile and hydrogen to form a primary amine.
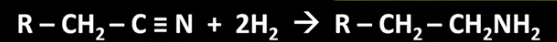
What is a way in which you can reduce nitriles without using hydrogen?
Add LiAlH4 (lithium aluminium hydride) and dilute acid.
True or false? LiAlH4 is cheap.
False. LiAlH4 IS EXPENSIVE.
What does [H] stand for?
Reducing agent.
What is LiAlH4 dissolved in before reacting with a nitrile?
A non-aqueous solvent (like dry ether).
Give the balanced symbol equation for the reaction between a nitrile and a general reducing agent to produce a primary amine.

Give two uses of aromatic amines.
Manufacture of dyes.
Pharmaceuticals.
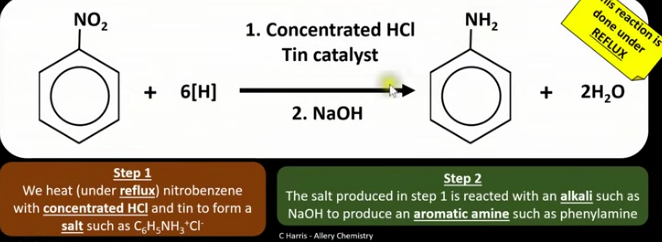
How can you make aromatic amines?
Reduce nitro compounds with a reducing agent.
Draw the balanced equation for the reaction between nitrobenzene and a reducing agent. Give the conditions of this reaction.
Draw and name the mechanism for the reaction between ammonia and an acyl chloride.
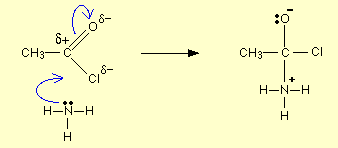
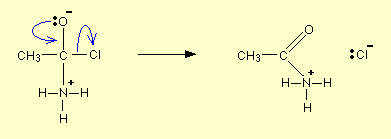
Nucleophilic addition-elimination
Draw and name the mechanism for the reaction between a primary amine and an acyl chloride.
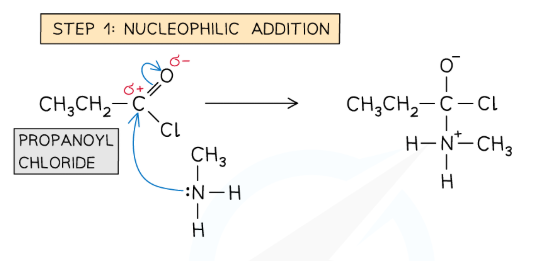
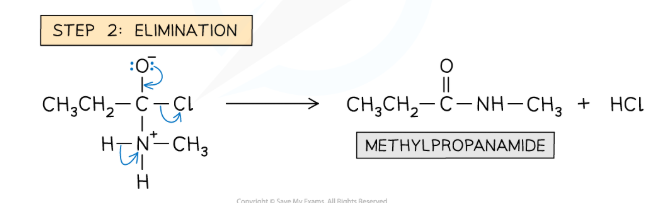
Nucleophilic addition-elimination