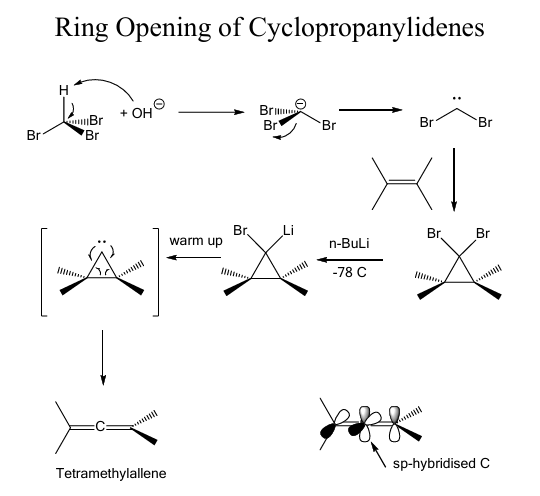
O5: Reactive intermidiates
1/37
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
38 Terms
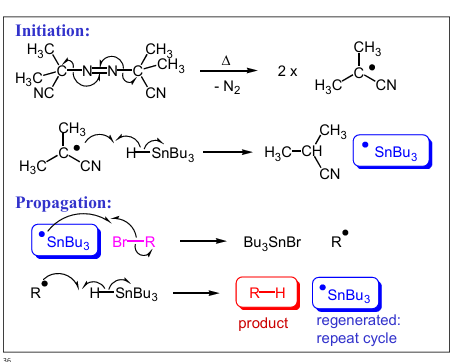
What does a Tin hydride reduction rxn do?
state reagents
It is a type of radical chain rxn that remove the functional group,
eg. R-Br → R-H
“AIBN”and “Bu3SnH”
Show the full rxn of turning
R-Br → R-H
Showing the mechanisms and naming the steps
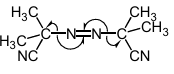
draw AIBN
write a Barton-McCombie Dehydroxylation via Xanthates including type of alcohols and reagents , draw xanathate formation
alcohols included for formation 2nd and 3rd alcohols
for breaking 2nd and 3rd with O-FG
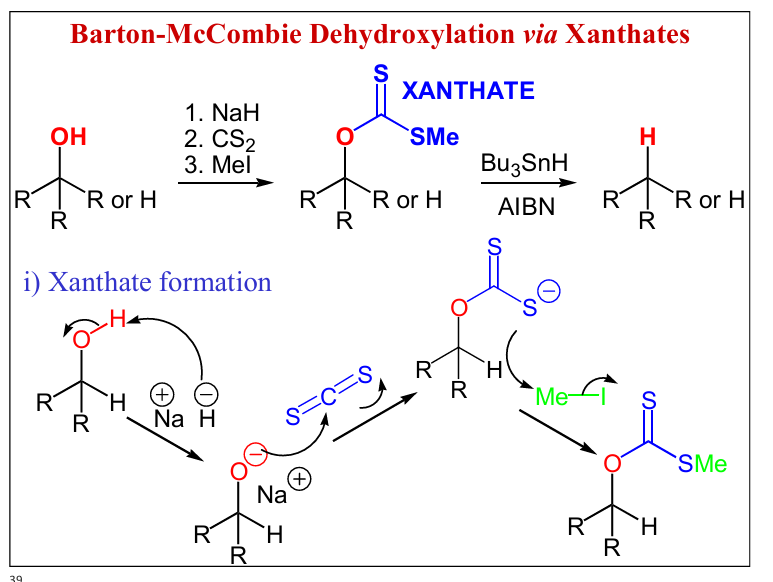
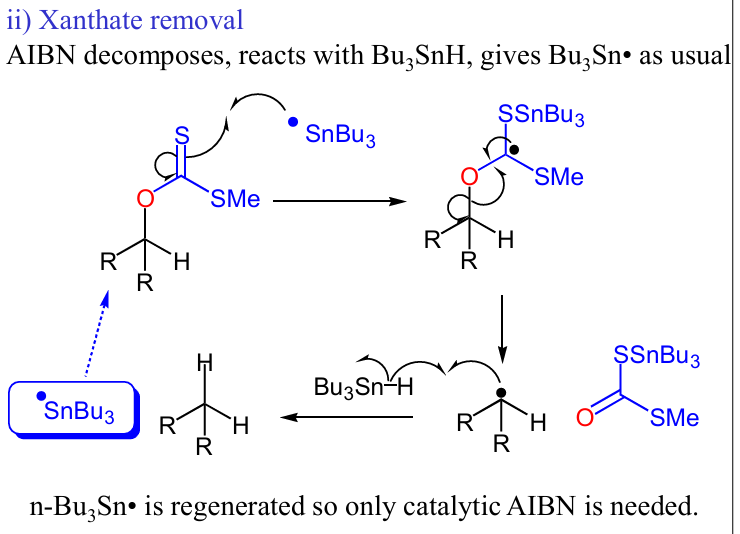
Draw a xanthate removal
Talk about EWG and EDG and their effect on radical alkene rxn
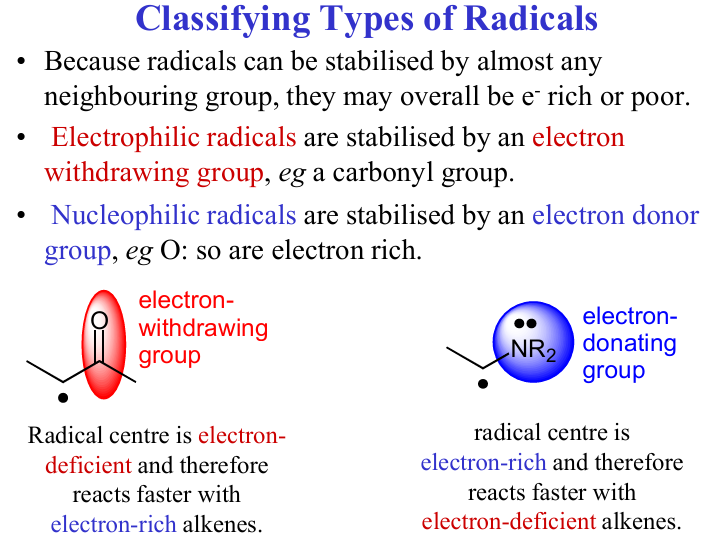
Give examples of EWG and EDG and how do you personally tell them apart
EWG have bond with a C or another atom next to an electronegative atom , and Halogens
While EDG have an atom bond next to eg, H, or a lower electronegative group, or a phenyl
EWGs: -NO2, -CN, -COOH, -CHO, -CF3, halogens
EDGs: -OH, -NH2, -OR, -CH3, -C6H5 (phenyl in some contexts)
What slows down radical rxns state 4 things
More stable radicals = slower rxn
Conjugation is stablising = more conjugation slower rxn
EDG stabilise by hyperconjugation = slower rxn
EDG faster rxn by induction, but if they allow reasonance then slower rxn
Sterics: bulky groups stabilise radicals while smaller make rxn faster
What radicals have fastest rxn ( primary, secondary,……) and why
Primary radicals (Fastest)>2> 3
Due to primary having the highest bond dissociation Energy ( lower BDE means Higher stability)
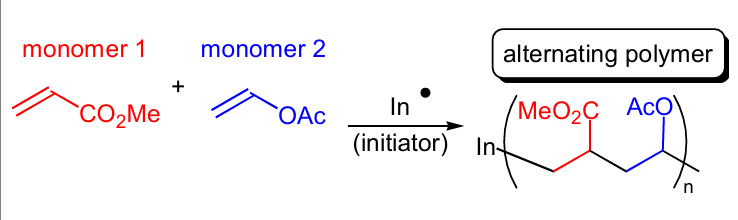
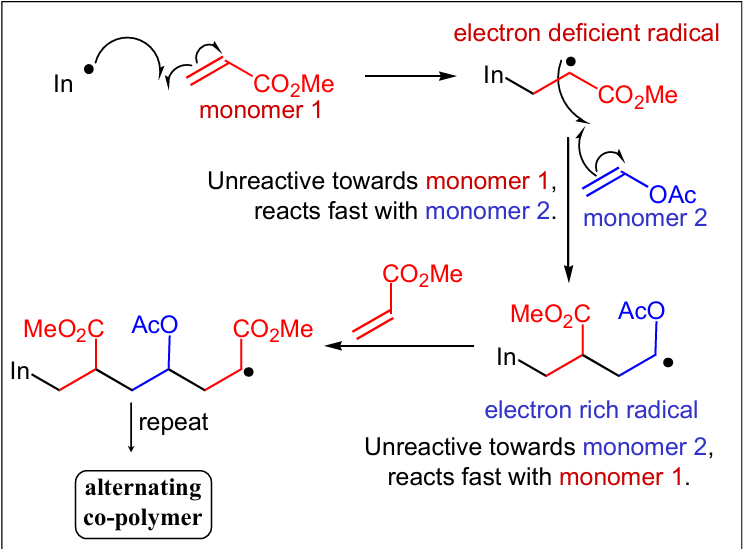
Show the mechanism behind the polymer effect
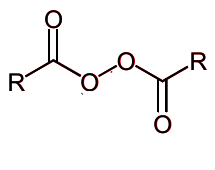
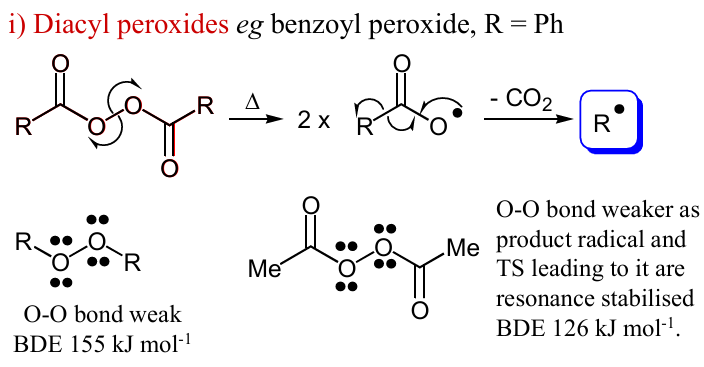
Draw the mechanism if you add heat to this
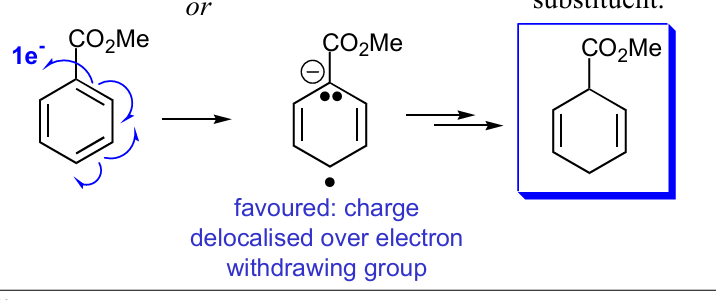
What is the favoured path for molecules rxn with e- with EWG, what would change if the group was OMe for example
the double bond on the left will bond to the e-, causing negative on ortho left and radical on meta right then takes proton
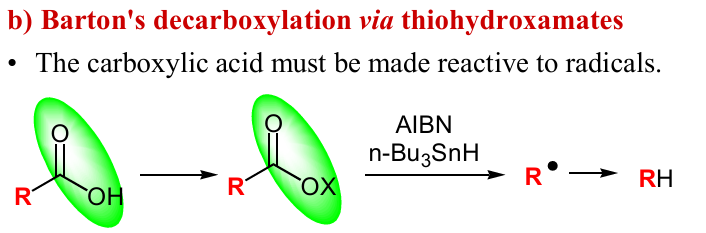
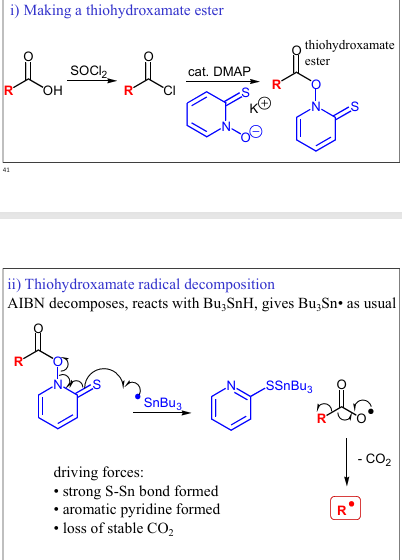
Give reagents and products to forming the OX, and give the mech to getting RH
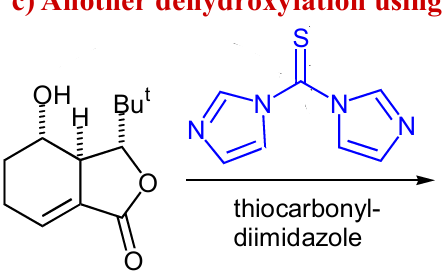
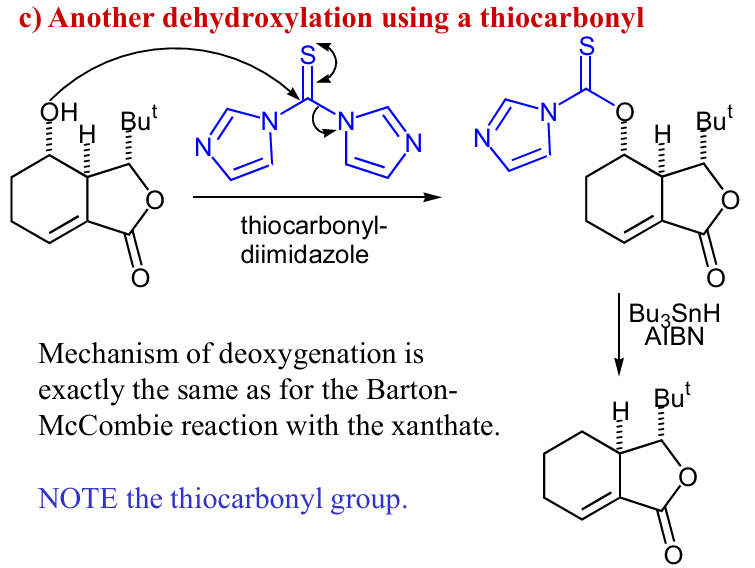
draw rxn mech and product, and how could we get rid of the OX to form another product (reagents only)

What reagents should you use for this intermolecular addition, show mechanism as well
HSnBu3 and AIBN
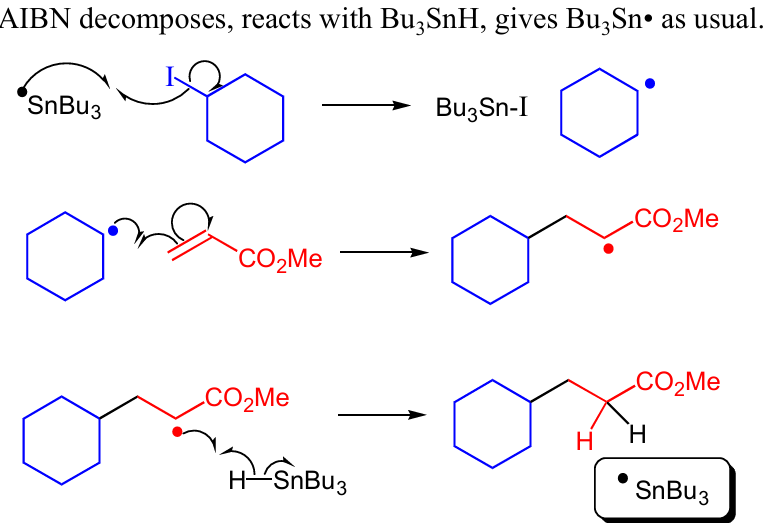
write out eg. of a radical clockwise of R - H
turns RH to ROH
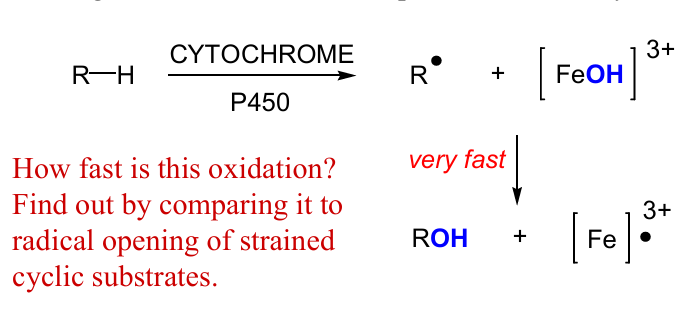
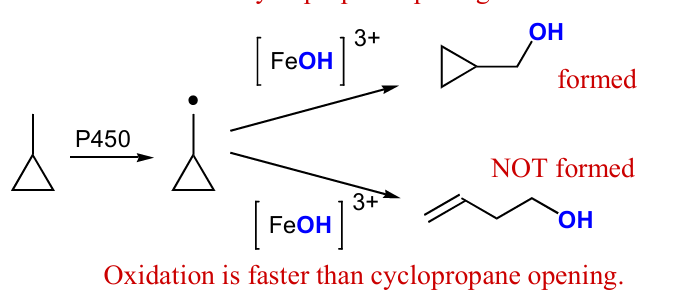
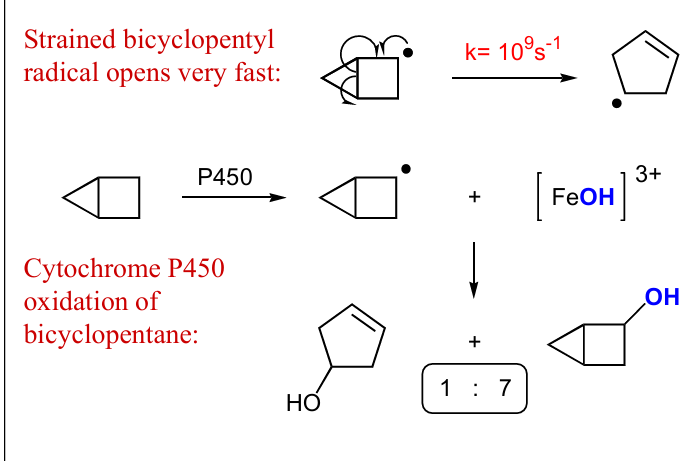
use P450, draw mech and products that formed and not formed
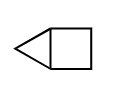
Do the same for this
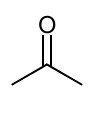
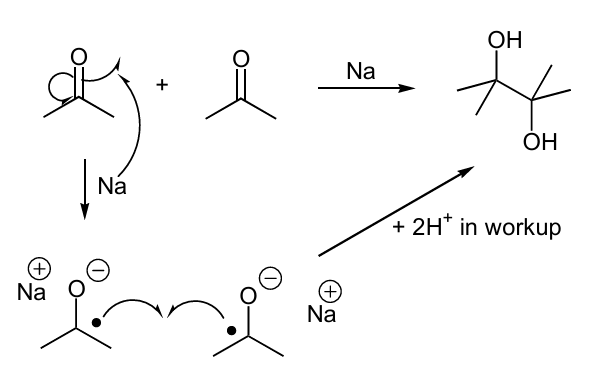
Using this do a Pinacol coupling rxn
notice 1 O up and the other is down

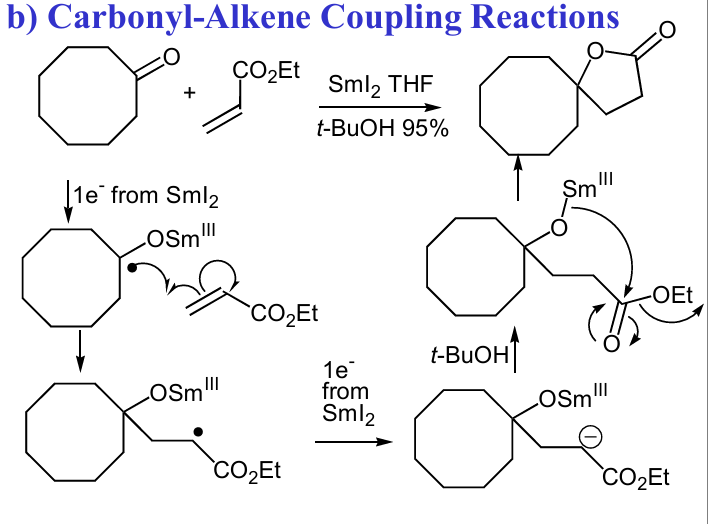
Give reagent and mech for this Carbonyl-Alkene Coupling Reactions
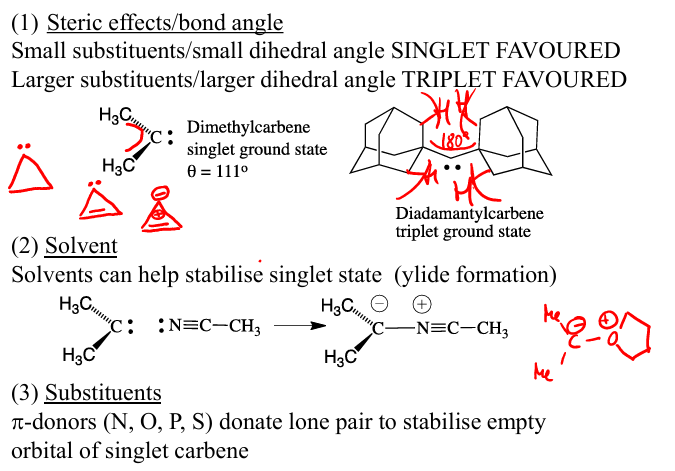
Orbital wise draw a singlet and a triplet carbene describe each of the e-s, how would triplet carbene bond to 2Cp
What stabilises the empty p orbital of a singlet
Triplet carbene will use one for sigma bonding to Cp as X
and the other one bonding with a pi bonding, a Cp as L2X
EWG pi interactions such with halogen stabilises the empty p orbital of a singlet

what R groups are prefered for each of the carbenes,and what can stabilie singlet carbenes
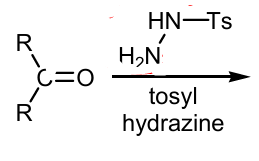
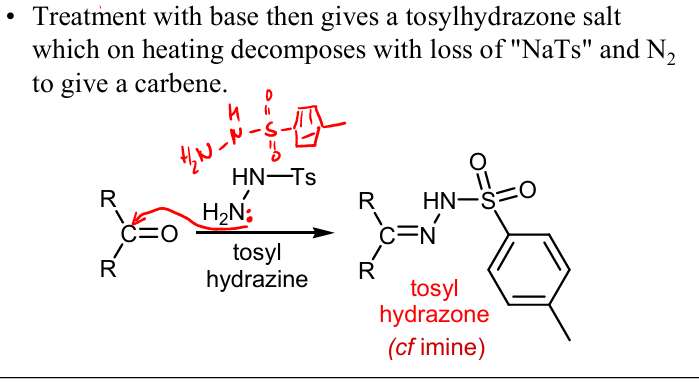
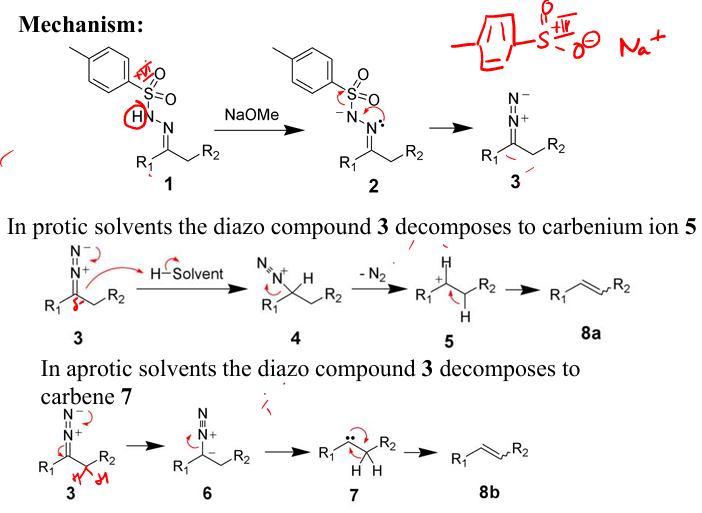
Show mechanism drawing the full product for the Generation from tosylhydrazones
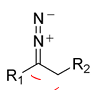
From the the Bamford-Stevens Reaction show how can you isolate this diazo compound and show how it will react in protic vs aprotic to give E alkenes
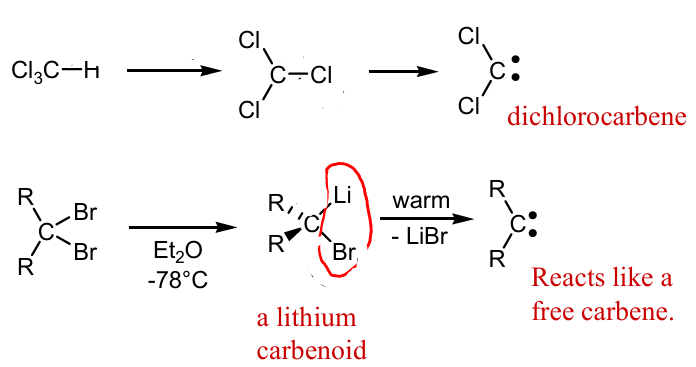
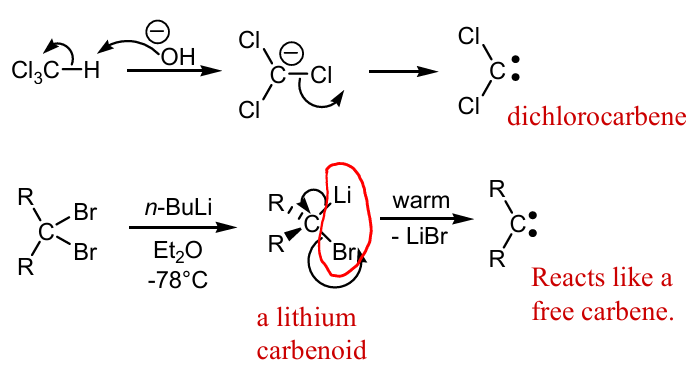
Show missing reagents and mechanism to making these carbenes
How would singlet carbene react with E and Z alkene, what would triplet show differently and why draw example
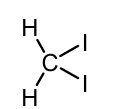
Turn this to a zinc carbenoid show reagents and product
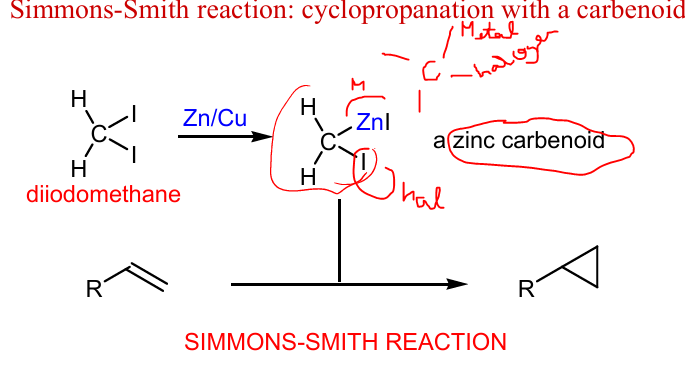
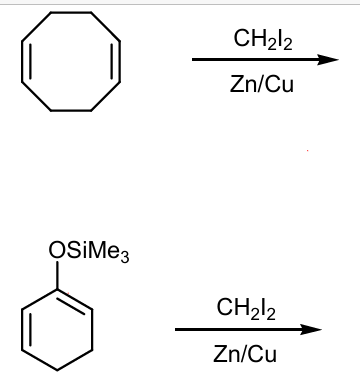
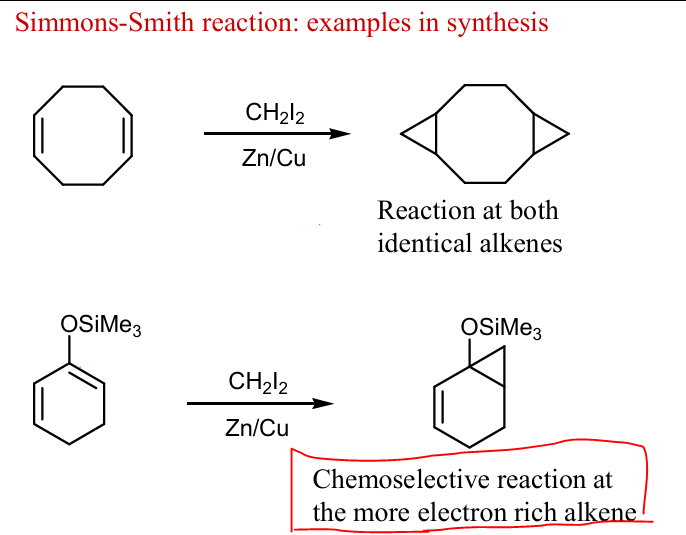
Draw products of these Simmons-Smith reactions
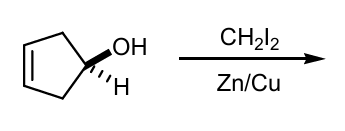
Draw product with stereohemistry and why?
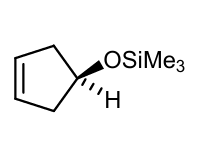
Draw product with stereohemistry (Why?) of a Simmons-Smith reactions
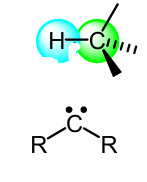
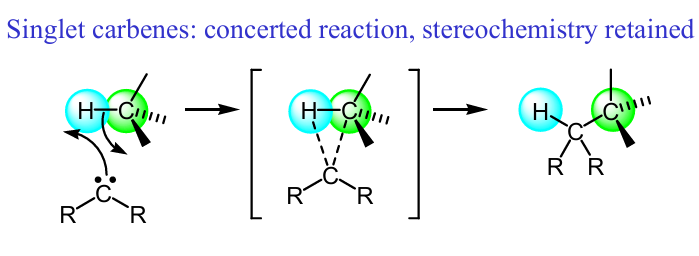
Show the mech of this singlet carbene rxn
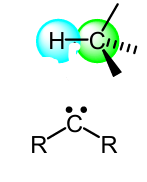
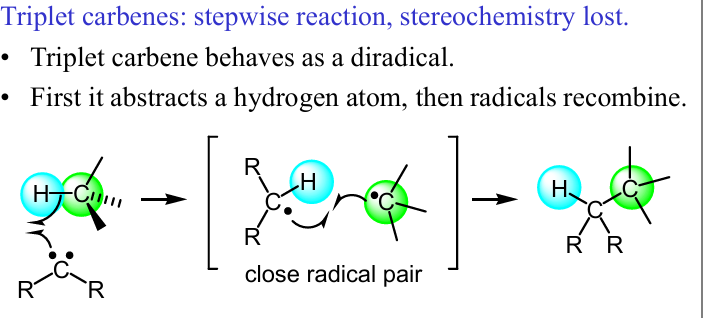
Show the mech of this tripletrcarbene rxn
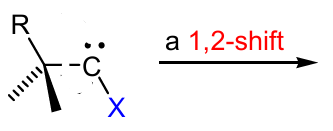
Show a carbene rearrangement reaction
can stick H, aryl, alkyls,…
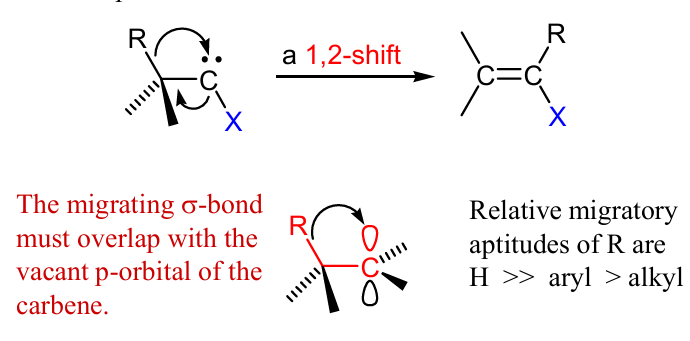
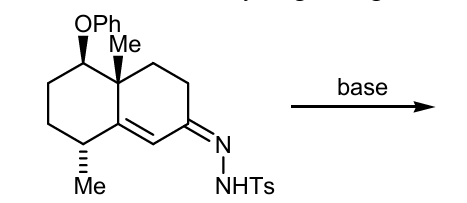
Show mech and product
double bond NNHTs, breaks to become carbene ( remember)
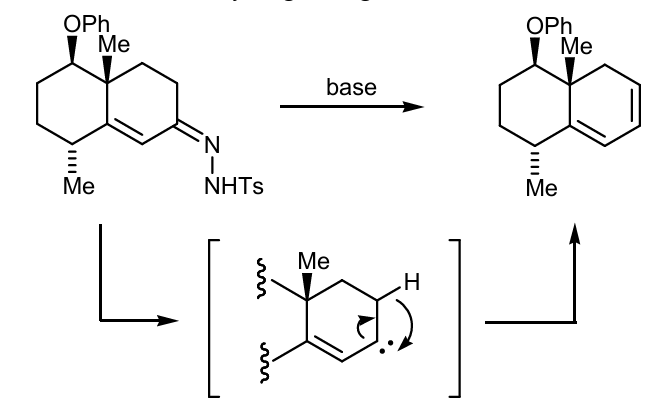
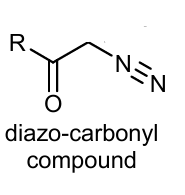
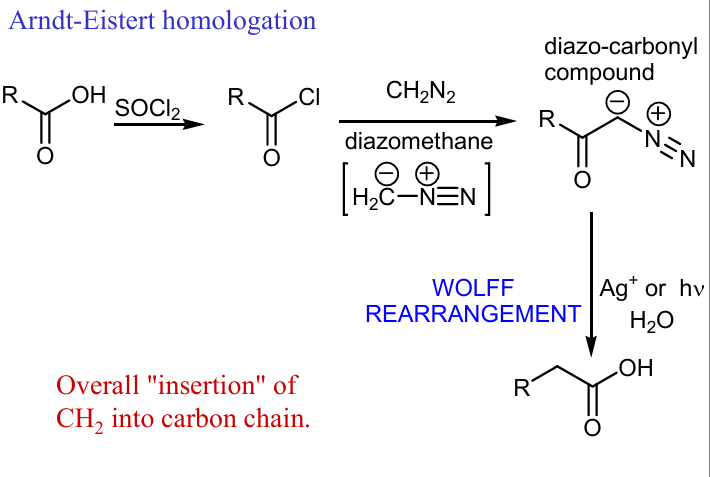
Show wolf rearegement of this
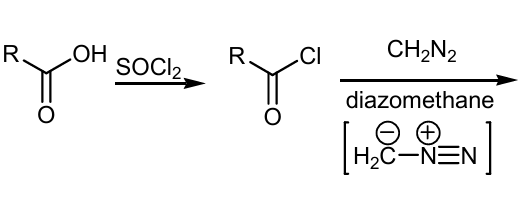
Continue the rxn using wolff rearengement
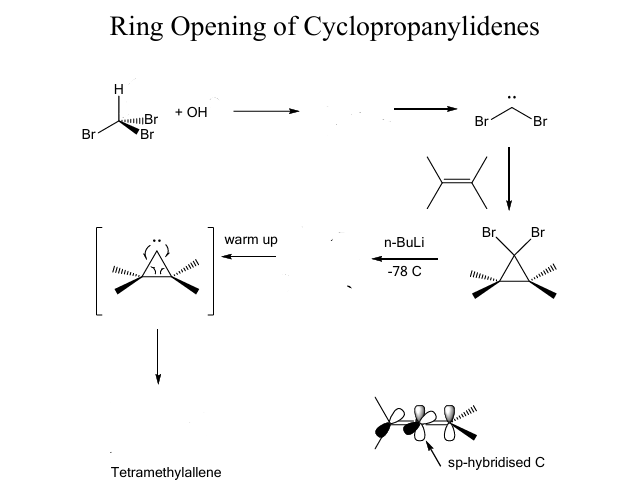
Show missing products and their mech