CHM2045 Exam 3
1/36
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
37 Terms
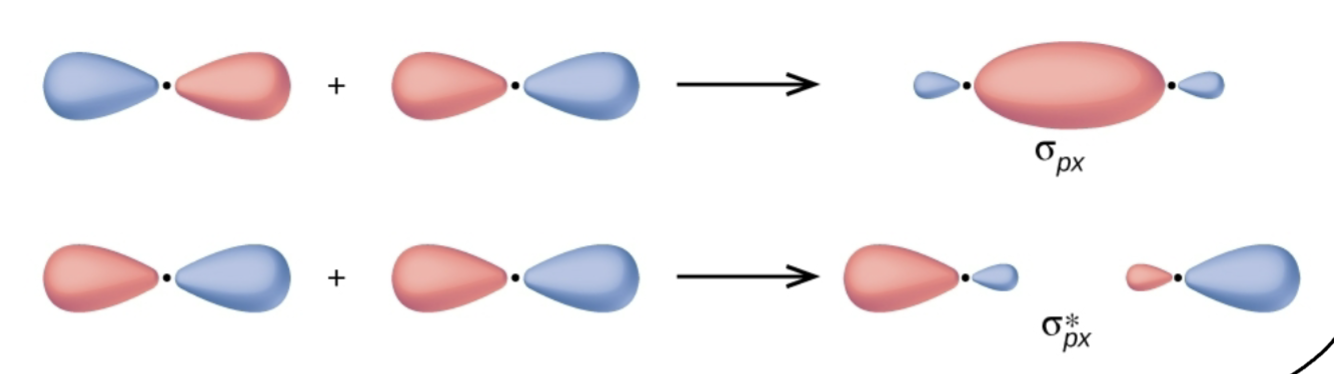
What kind of bonding?
Sigma
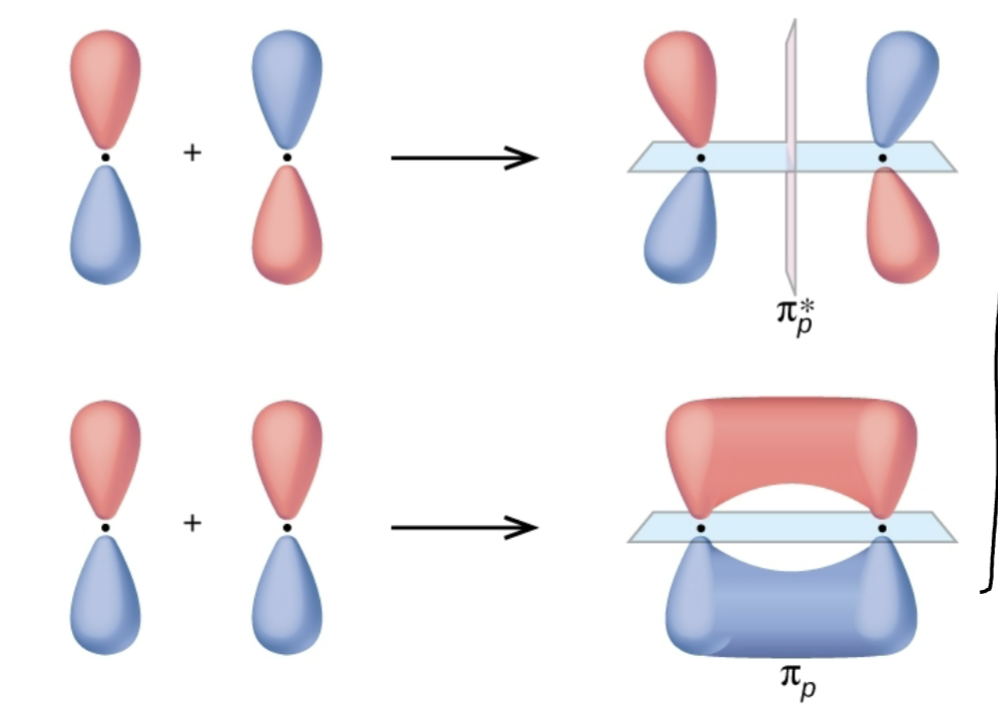
What kind of bonding?
Pi
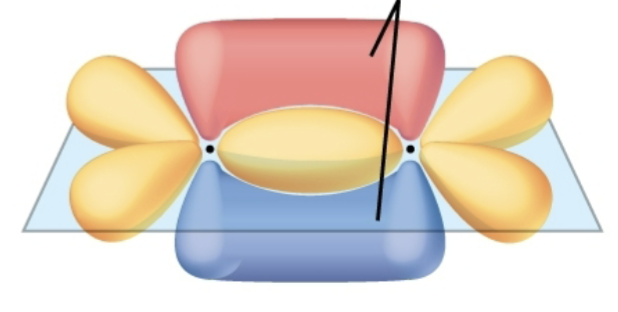
Bond order?
Double bond
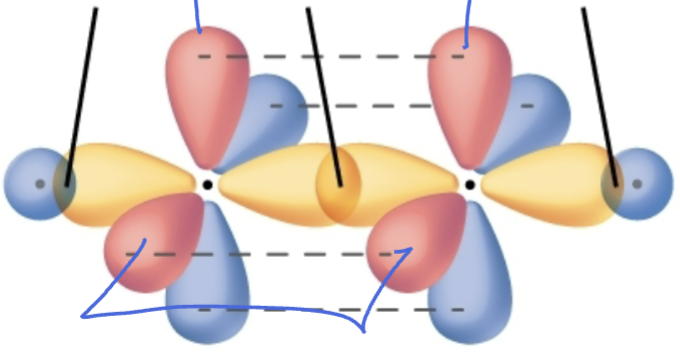
Bond order?
Triple bond
Four principles of MO theory

What does adding wave functions produce?
A bonding MO; constructive interference
What does constructive interference do?
Promotes bonding
What does subtracting wave functions produce?
Antibonding MO; destructive interference
What does destructive interference do?
Decreases bonding
How do you rank the stability of MO bonds?
Highest bond order > lowest bond order
MO bond order
½ (# e- in bonding orbitals - # e- in antibonding orbitals
Why doesn’t He2 exist?
Bond order = 0
What does unstable mean?
Bond order = 0; does not exist
How does number of bonding e- vs. antibonding e- affect bonds?
More bonding vs. nonbonding = stronger bonds
Paramagnetic
Electrons not paired
Diamagnetic
Electrons paired
Why does Ne exist as a monoatomic gas?
MO bond order
Benzene formula
C6H6
Why is resonance not needed in MO theory?
Electrons delocalized amongst bonds
AX2 molecular geometry
linear
AX3 molecular geometry
trigonal planar
AX2E1 molecular geometry
bent
AX4 molecular geometry
tetrahedral
AX3E1 molecular geometry
trigonal pyramidal
AX2E2 molecular geometry
bent
AX5 molecular geometry
trigonal bipyramidal
AX4E1 molecular geometry
seesaw
AX3E2 molecular geometry
t shaped
AX2E3 molecular geometry
linear
AX6 molecular geometry
octahedral
AX5E1 molecular geometry
square pyramidal
AX4E2 molecular geometry
square planar
AX3E3 molecular geometry
t shaped
AX2E4 molecular geometry
linear
Are lone pairs equatorial or axial?
Equatorial
In Cl2CO, what direction is the overall dipole?
Toward the O
A liquid boils when…
its vapor pressure equals the external pressure. (use for Clausius-Clapeyron equation)