Chemistry - Rates of Reaction
1/9
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
10 Terms
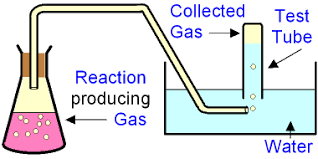
Collection under water
Errors:
Some gas escapes, especially before the bung is inserted
Inserting the bund displaces air
Some gases dissolve in water (CO2 and O2 particularly)
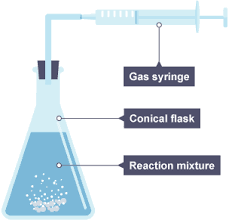
Using a gas syringe
Errors:
Some gas escapes, especially before the bung is inserted
Inserting the bund displaces air
Temperature is not constant (of gas) [tends to get hotter → expansion]
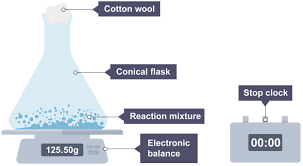
Measuring change in mass
BEST METHOD
Errors:
Some gas escapes, especially before the cotton wool goes in
Some gas may dissolve in the liquid
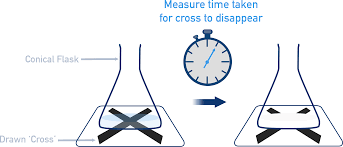
Measuring the time taken for cross to be invisible
Errors:
Relies on judgement / eyesight / size of cross
Surface area
More particles of solid are exposed so more frequent collisions
Concentration
More particles in the same volume of liquid = more frequent collisions
Pressue
More particles in the same volume of gas so more frequent collisions
Temperature
Particles gain more kinetic energy so move faster so collide more frequently. This also means that a higher percentage of particles have higher energies than the activation energy so a higher percentage of collisions are successful
Adding a catalyst
Provides an alternative reaction route with a lower activation energy so a higher percentage of particles have higher energies than the activation energy so a higher percentage of collisions are successful
Catalyst
A substance that increase the rate of reaction but is chemically unchanged at the end of the reaction (not used up)