equations not given
1/24
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
25 Terms
fractional saturation (weak binding)
F = [L]total / (KD + [L]total)
What is the equation for Δo?
Δo= E(eg) − E(t2g)
Rate =
Rate = change in conc / change in time
Unit: M.s-1 or mol L-1s-1
Write the rate of A→ B
with respect to A and with respect to B
-d[A]/dt
d[B]/dt
what is the rate law of A and B in this reaction?
A→ B
Rate law for A:
d[A]/dt = -K[A]
Rate law for B:
d[B]/dt = K[A]
what is the time constant + its equation?
Avg time for A to be converted into B in a reaction
τ = 1/k
In this equation:
A + B —> C
What is the rate equation with respect to A,B and C?

P + L ←→ P:L
what is the rate law with respect to P?
d[P]/dt = -kon [P][L]+ koff [PxL]
What is the arrhenius equation?
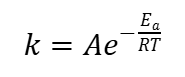
Whats the equation for k1 and k2 in a 2-step equation at a steadt state ?
k1[A]ss = k2[B]ss
What is the equation for the rate law of A in a reversible reaction?
d[A]/dt = -kf[A]+ kr[B]
what is the ratw law for the following equation:
A + A + B —> C
With respect to A / B
½ d[A]/dt = d[B]/dt = -k[A]2[B]
What is the linearised form of the arrhenius equation?
lnk = [-Ea/ R] 1/T + lnA
What is the pseudo-first-order rate constant
k′=kon[L]0
What is the equation for the observed rate constant in pseudo-first-order ligand binding?
kobs=kon[L]0+koff


What is the dissociation constant KD in terms of kinetic constants?
KD=koff/kon


U=
U = q + w
DU =
DU = q - w
H =
H = U + pv
DH =
DH = DU + Dpv
DS =
DS = DQ/T
in J.K-1
Kc =
Kc = [C]c[D]d / [A]a[B]b
wavelenght =
wavelength = hc/E