Chemistry Sc5, Sc6, Sc7 - Bonding, structures, properties (Year 9 Spring term)
1/39
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
40 Terms
ion
an atom or groups of atoms with an electrical charge due to the gain/loss of electrons
Cation
A positively charged ion
Anion
A negatively charged ion
Ionic bonding is between
metals and non-metals
electrostatic forces
forces of attraction between oppositely charges particles
what happens to the ending of non metal ions
add -ide (oxygen > oxide)
Ionic Compound
No overall charge (neutral) e.g Aluminium Chloride
-ide compounds
contain only 2 elements
-ate compounds
contain oxygen and 3 or more elements
Polyatomic ions
groups of 2 or more atoms which have become charged
Ammonium
NH4+
Nitrate
NO3-
Hydroxide
OH-
Carbonate
CO32-
Sulfate
SO42-
Sulfite
SO32-
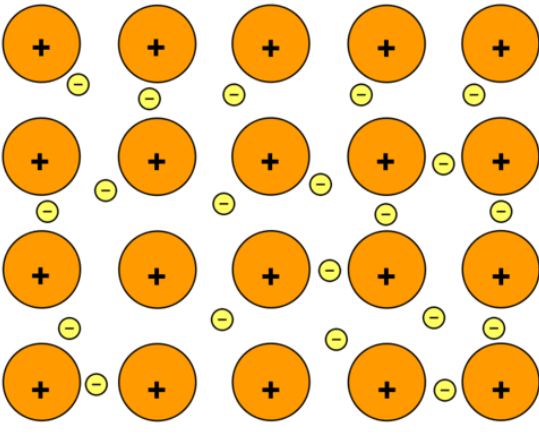
ion arrangement
packed together in a regular repeating arrangement cfalled a LATTICE
alternating cations and anions
held together by electrostatic forces
Properties of Ionic Compounds
-high melting/ boiling points -require lots of energy to break -Only conduct electricity when molten or aqueous as the ions are free to move
many soluble in water
hard but brittle
covalent bond
a shared pair of electrons
what does covalent bonding occur between
non-metals
Valency
How many covalent bonds an atom will make 8 - group number = valency
How are molecules held together
atoms in molecules are held together by strong electrostatic forces of attraction between the positive nuclei and the negative electrons
Simple Molecular (covalent) structure
small distinct groups of atoms
Giant covalent structure
billions of atoms held together in a lattice structure
simple molecular properties
low melting/boiling points
a few are soluble in water
most do not conduct electricity
why do simple molecules have low melting/boiling points
because it does not take much energy to overcome weak intermolecular forces
why do simple molecules not conduct electricity
Because molecules are neutral so cannot carry charge
Giant covalent properties
high melting/boiling points
do not conduct electricity ( apart from graphite and graphene due to delocalised electrons)
insoluble in water
polymer
monomers joined in a chain -longer polymers have more intermolecular forces -> higher melting/boiling points
Diamond (Structure, Properties, Uses)
Structure- giant covalent Properties: -high melting point -insoluble in water -does not conduct electricity (no free electrons) -4 strong covalent bonds Uses: used in cutting tools (hard)
Fullerene (structure, properties, uses)
Structure - simple molecule
-low melting point
-soft + slippery
-does not conduct electricity (delocalised electrons can't move between molecules)
-3 covalent bonds
Uses: carbon nanotubes can conduct electricity (electronics)
Graphite
structure - giant covalent (layers of graphene) -high melting point
insoluble in water
good conductor(layers allow delocalised electrons to be free to move)
Uses: electrodes in electrolysis, pencil leads (slippery)
Graphene structure, properties and uses
structure: giant covalent- single layer sheets of carbon in hexagonal lattice
-high melting point -very strong
-good conductor of electricity (delocalised electrons)
Uses: added to plastics/carbon fibre (strong)
typical properties of metals
-solids with high melting points
-shiny
-malleable
-high density
-good conductors of electricity
typical properties of non-metals
-solid/liquids/gases with low melting point -brittle -low density
poor conductors
why are metals malleable
because layers of ions can slide over each other
why can metals conduct electricity
because delocalised electrons flow through the structure carrying a charge
delocalised electrons
not associated with a particular atom or bond
metallic bonds
strong electrostatic forces of attraction between positive metal ions and negative delocalised electrons
metal structure
giant metal lattice -electrons in the outer shell are delocalised > move throughout the structure -metal atoms are + since they lose electrons