ANP140 Chapter 24 Water, electrolyte, and acid-base balance
1/64
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
65 Terms
Intercellular fluid
65% of water
Fluid inside cells
Extra cellular fluid
35% of water
fluid outside cells
-tissue fluid 25%
-bloodstream and lymph 10%
Water intake / output
2500 ml/day
water intake
200ml metabolic water
700ml food
1600ml drinking
water output
200ml feces (increase with diarrhea)
300ml Expired air (increase w cold) (insensible water loss)
400ml cutaneous transpiration (insensible water loss)
100ml sweat (increase w temperature)
1500ml urine
urine output
Only way the body can regulate change in h20 balance
- Must have at least 400ml per day to get rid of metabolic waste
Regulation of water intake
increase blood osmolarity
stimulate hypothalamic osmoreceptors
increase ADH
ingestion of h20
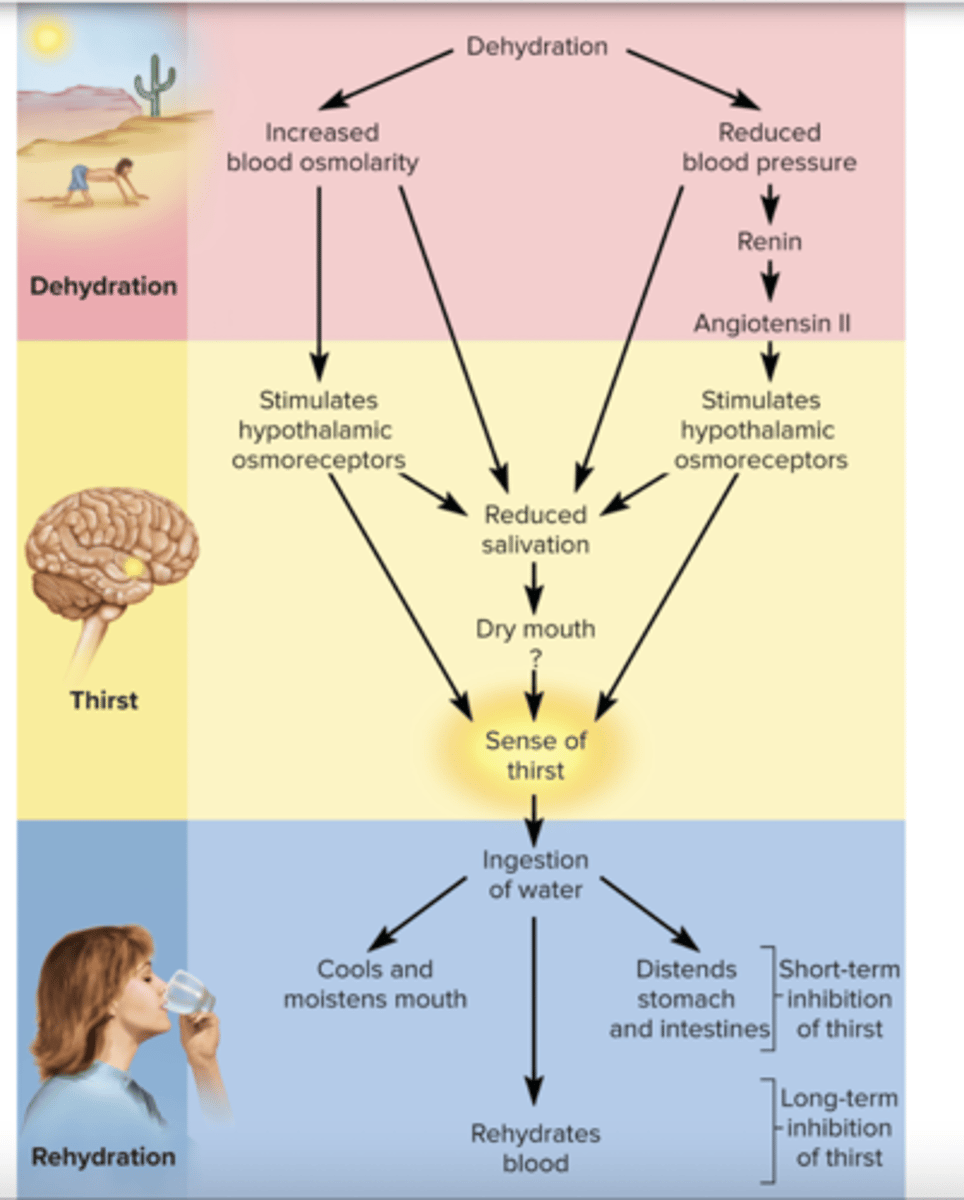
ingestion of Water
1.) cools and moisten mouth
2.) distend stomach
- Short term inhibition, don't drink to much ~ 30 min
- Long term inhibition of thirst
3.) Rehydration of blood
Increase firing of osmoreceptors
increase thirst
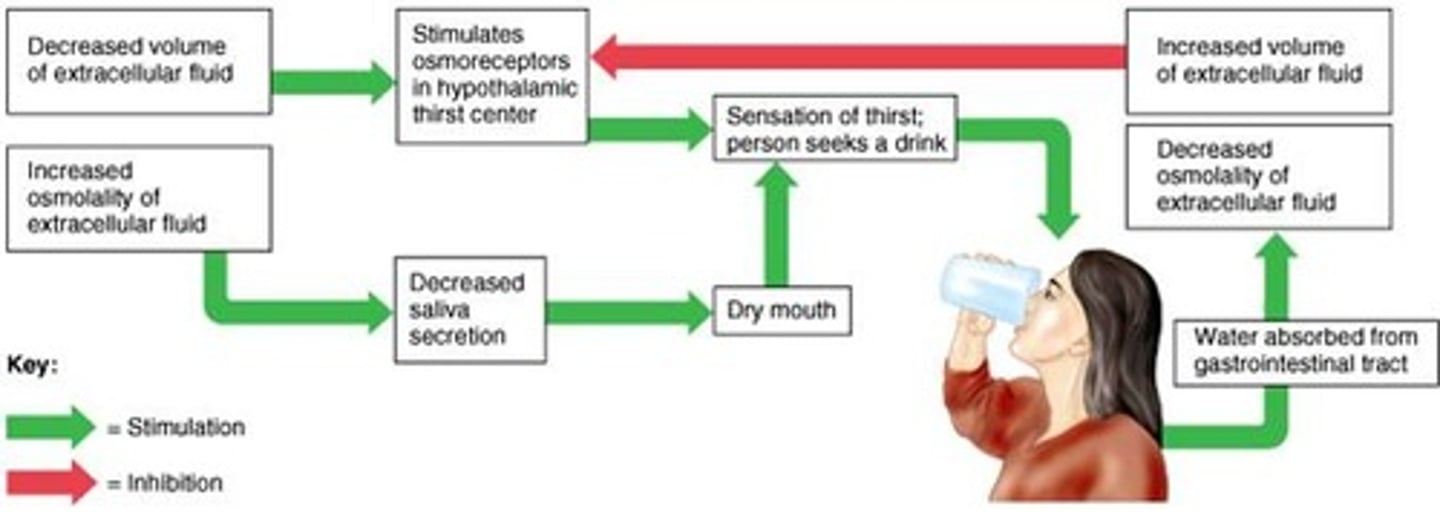
osmoreceptor-ADH mechanism
This mechanism helps regulate water output. Osmoreceptors in the hypothalamus detect high osmolarity, triggering production of ADH as you become thirsty. This causes changes in water permeability in the distal convoluted tubules and collecting ducts so that water is minimized. If low osmolarity is detected (proper hydration), ADH is not secreted in high levels, so water can be lost through urination as the distal convoluted tubule and collecting duct are not permeable to water now
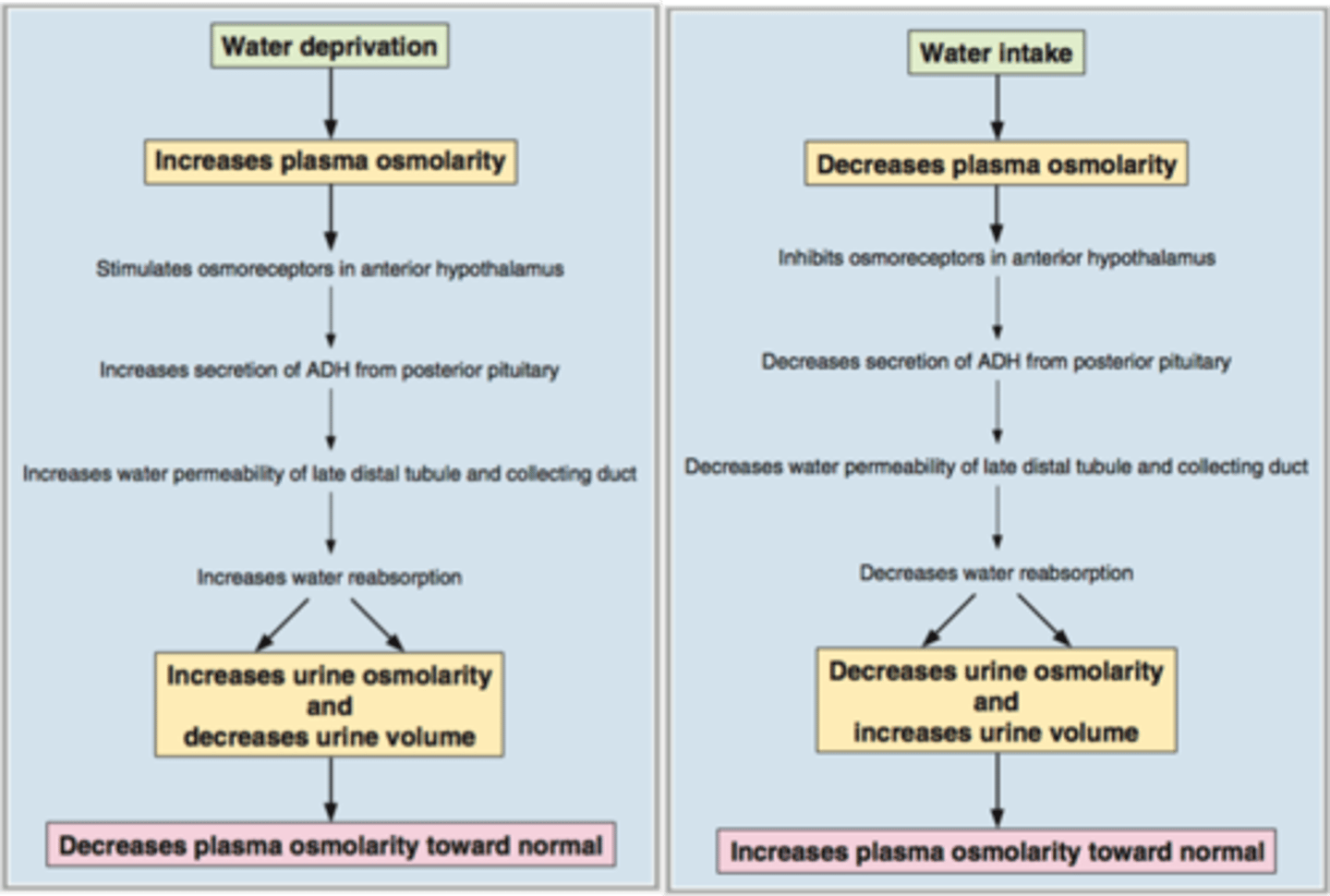
Regulation of plasma osmolarity
ex.) Dehydration
Increase plasma osmolarity
increase osmoreceptors
increase ADH
Increase h20 reabsorption in CD
decrease h20 excretion
- increase urine osmolarity
- decrease plasma osmolarity
Water imbalance
can lead to circulatory shock or neuro dysfunction from decrease BV/BP or dehydration
Hypovolemia
decreased blood volume
ex.) hemorrage
Hypovolemia osmolarity
normal
Hypovolemia causes
loosing h20 and na+ in even amounts
Dehydration
Lack of drinking h20
dehydration osmolarity
increase osmolarity
dehydration casues
More h20 loss then Na+
Dehydration changes cells
cells shrink because h20 leaves
Fluid Deficiency
water out > water in
fluid excess
water in > water out
(less common bc kidneys are good at removing excess)
volume excess
extra h20 and Na+ in even amounts
ex.) renal failure
volume excess osmolarity
Normal
Hypotonic hydration
More h20 the Na+
ex.) drinking lots of water
hypotonic hydration osmolarity
decrease osmolarity
hypotonic hydration cells
swell up
volume excess and hypotonic hydration cause
edema because of increase BP/BV
Pulmonary + cerebral edema
ECF
Na+ and Cl
ICF
K+ and phosphates
electrolyte balance
ECF and ICF should be 300 m0ms/L
Sodium (Na+)
Only need 0.5 g/day
Hypernatremia
high sodium in the blood plasma
- dehydration
Hyponatremia
low sodium in the blood plasma
- excess h20 consumption
sodium imbalances
CNS dysfunction, edema, Increase BP
Potassium (K+)
Small changes in ECF K+ can be deadly due to effect on excitable tissues
Hyperkalemia
excessive potassium in the blood plasma
Cells more excitable, increase RMP
K+ diffuse into cell making it positive
Ex.) Crush injury, cardiac arrest
Hypokalemia
Low potassium levels in the blood plasma
Cells less excitable, decrease RMP
K+ diffuse out of cell making it negative
ex.) Muscle weakness, arrhythmia, vomiting, diarrhea
Normokalemia
A normal level of potassium in the blood
Normal resting membrane potential -70mv
Aldosterone regulates
Na+ and K+ concentrations
Hypercalcemia
increase plasma calcium
Hypercalcemia effects
muscle weakness, arrhythmia
(Hypoparathyroidism (increase PTH))
hypocalcemia
decrease plasma calcium
hypocalcemia effects
cramps, seizures, tetanus, heart failure, asphyxiation
(vitamin d deficiency)
Calcium
reabsorption regulated by PTH
Absorption increases with vitamin D
Acids
release H+ into solutions
bases
accepts h+
Buffors
stabilize pH by removing H+ or adding H+
strong acids
HCl
Fully ionize into H+ + Cl-
weak acids
H2CO3
Can go between H2CO3 and HCO3- + H+
Balanced pH
H+ equal HCO3-
increasing acidic
decrease pH
Increase H+
increase basic
Increase pH
decrease H+
sources of gaining H+
increase CO2
metabolism of proteins
decrease HCO3- (diarrhea, urine)
sources of losing H+
decrease CO2
metabolism
decrease H+ (vomiting, urine)
High protein diet
excess H+ ions so urine is acidic
buffer system ICF
1.Phosphates
H2PO4- <--> HPO4- + H+
Buffer system ECF
2.) Bicarbonate
CO2 + H2O <--> H2CO3 <--> HCO3- + H+
protein buffer system
Primary ICF buffer (hemoglobin)
Also buffers ECF (plasma protein)
3/4 buffer capacity
-COOH <--> - COO + H+
-NH3+ <--> -NH2 + H+
mechanisms to maintain acid base balance
buffers, respiratory, renal
1.) Buffers
first line of defense
short term
fast/speedy - (msec-sec)
Low buffering capacity
2. Respiratory
Change H+ levels by changing ventilation to alter CO2
intermediate Buffer capacity
intermediate speed - (sec-min)
3.) renal
Excrete excess H+ and HCO3-
longer term
high buffer capacity
slow speed (hours to days)
Renal Control of pH
Kidneys filter HCO3- but cannot reabsorb directly
- Filtered HCO3- is recycled into new HCO3- then reabsorbed
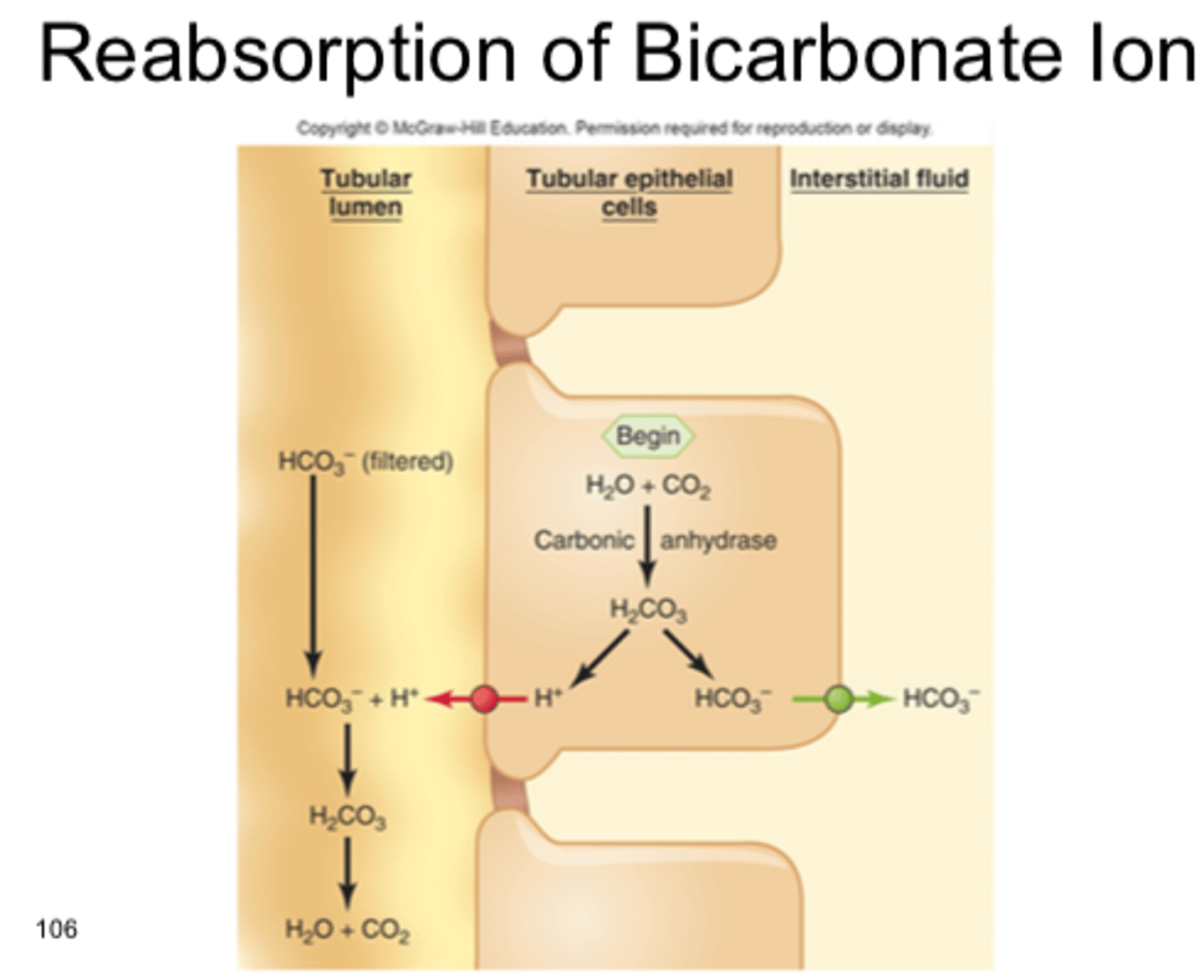
excess HCO3- (alkalosis)
-HCO3 > H+
Excess HCO3 has no H+ to combine with and is excreted
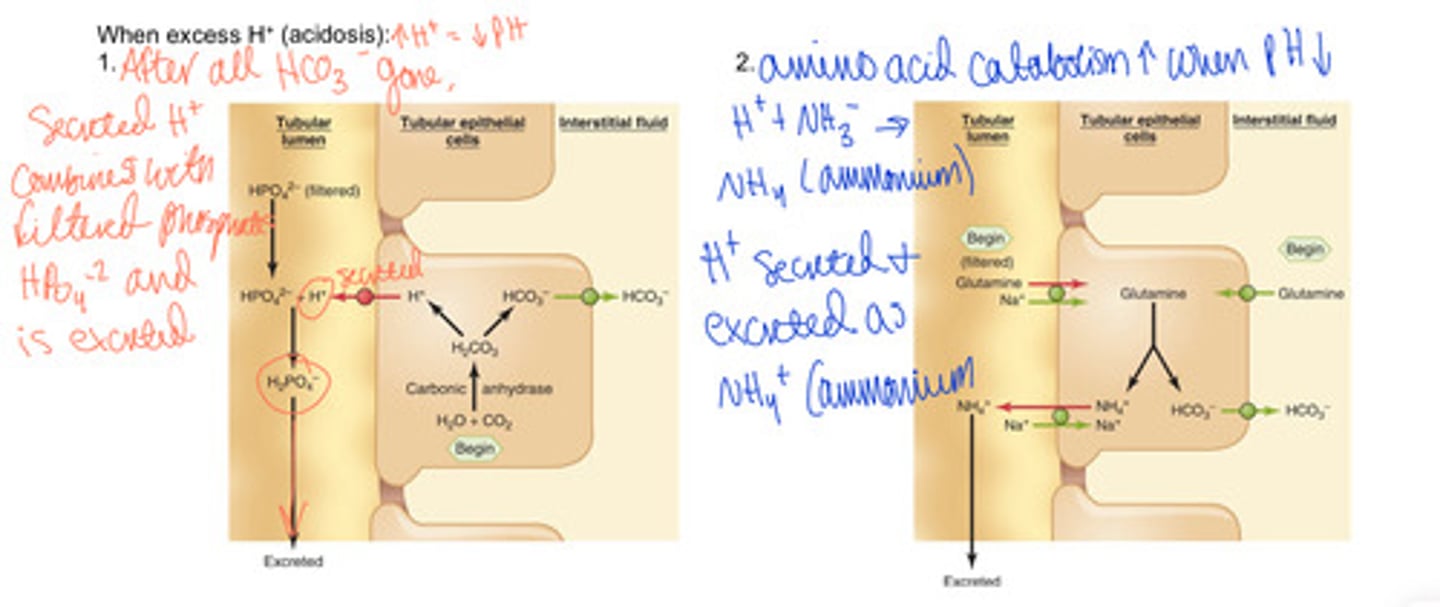
excess H+ (acidosis)
H+ > HCO3-
1.) secreted H+ combine with phosphates and are excreted
2.) amino acid catabolism increases when pH decrease, Excess H+ is excreted as ammonium (NH4+)