Unit 10
1/13
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
14 Terms
Stereocenter
An atom within a molecule where switching any two of its substituents gives a new stereoisomer
Assigning Absolute Configuration of an Alkene
Split the alkene down the middle (perpendicular to the sigma bond)
Rank the priority of R1 vs R2 using the CIP rules
Rank the priority of R3 vs R4 using the CIP rules
Assign E or Z
Z
Together
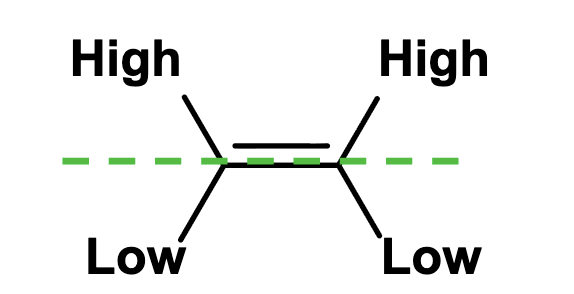
E
Opposite
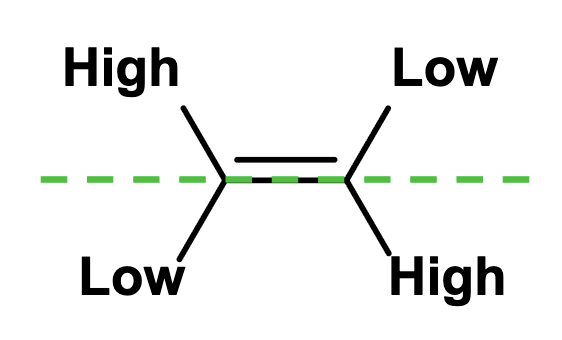
CIP Rules
Look at the atoms directly attached to each of the double bond carbons and rank them in order of decreasing atomic number
If a decision can’t be reached by ranking the first atoms in the substituent, look at the second, third, fourth, etc. atoms away from the double bond carbons until a difference is found.
Multiple bonded atoms are considered equivalent to the same number of single bonded atoms
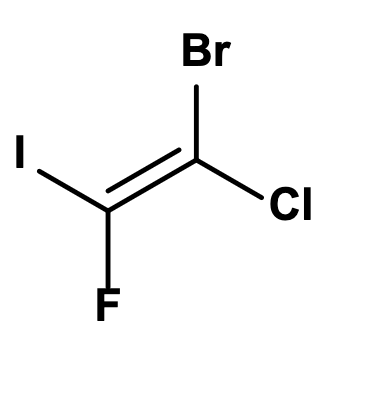
Assign E, Z, or neither
Z
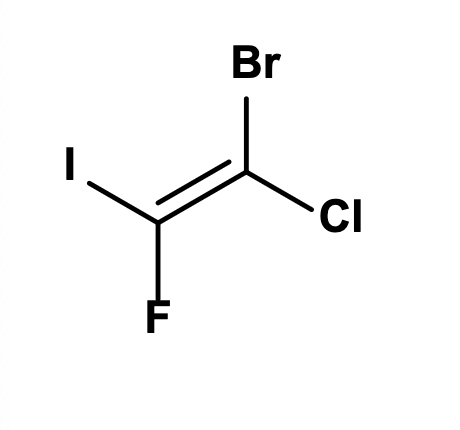
What is the IUPAC name of the alkene above?
(Z)-1-bromo-1-chloro-2-fluoro-2-iodoethene
Asymmetric Centre
All asymmetric centers are stereocenters, but not all stereocenters are asymmeteric centres
Absolute Configurations of Asymmetric Centres (R and S)
Prioritize substituents using CIP rules
Orient molecule so that #4 priority is pointing straight back into the page
Draw an arrow from priority #1 to #2 to #3
R Configuration
Arrow goes clockwise
S Configuration
Arrow goes counter-clockwise
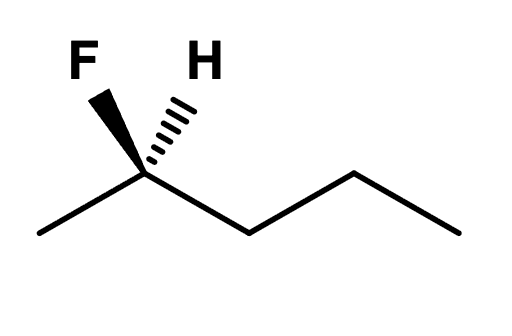
Label each asymmetric centre in the molecule below as R or S.
R