period 3 elements continued
1/67
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
68 Terms
structure and bonding of sodium
metallic
structure and bonding of magnesium
metallic
structure and bonding of aluminium
metallic
structure and bonding of silicon
giant covalent,
macromolecule
structure and bonding of phosphorus
P4, molecule, simple covalent, van der waals
structure and bonding of sulfur
S8, simple covalent, van der waals
structure and bonding of chlorine
diatomic, simple covalent
observation of sodium and water
floats and moves on water,
fizzes and may catch fire,
yellow-orange flame
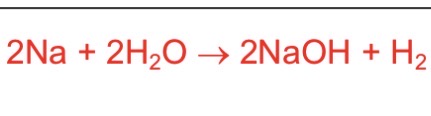
equation between sodium and water
observation between magnesium and water/steam
steam: burns with white flame giving white powder
water: very slow reaction
reaction between magnesium and water
Mg + 2H2O → Mg(OH)2 + H2
reaction between magnesium and steam
Mg + H2O → MgO + H2
which period 3 elements don’t react with water
aluminium, silicon, phosphorus, sulfur
observation of reaction between chlorine and water
dissolves giving very pale green solution
reaction between chlorine and water
Cl2 + H2O ⇌ HCl + HOCl
observation of reaction between sodium and oxygen
burns with yellow-orange flame to produce white powder
equation between sodium and oxygen
4Na + O2 → 2Na2O
observation for reaction between magnesium and oxygen
burns with white flame to produce white powder
equation between magnesium and oxygen
2Mg + O2 → 2MgO
Mg + ½ O2 → MgO
observation of reaction between aluminium and oxygen
burns with white flame to produce white powder
equation between aluminium and oxygen
4Al + 3O2 → 2Al2O3
observation of reaction between silicon and oxygen
burns with white flame to produce white powder
equation between silicon and oxygen
Si + O2 → SiO2
observation between phosphorus and oxygen
burns with vert bright white flame to produce white powder
equation between phosphorus and oxygen
P4 + 5O2 → P4O10
observation between sulfur and oxygen reaction
burns with blue flame and gives off choking gas
reaction between sulfur and oxygen
S + O2 → SO2
which period 3 element doesn’t react with oxygen
chlorine
structure of sodium oxide
ionic
structure of magnesium oxide,
why does it have a higher m.p. than Na2O
Ionic (higher melting point than Na2O as the Mg2+ions are smaller and higher charged than Na+)
structure of aluminium oxide
ionic solid
why is melting point of aluminium oxide not as high as expected
covalent character due to polarising nature of aluminium ions
structure of silicon oxide
giant covalent solid
structure of phosphorus oxide
simple molecular, solid
structure of sulfur dioxide
simple molecular gas,
bent molecule
structure of sulfur trioxide
simple molecular,
trigonal planar structure
what happens when sodium oxide reacts with water
dissolves
equation between sodium oxide and water
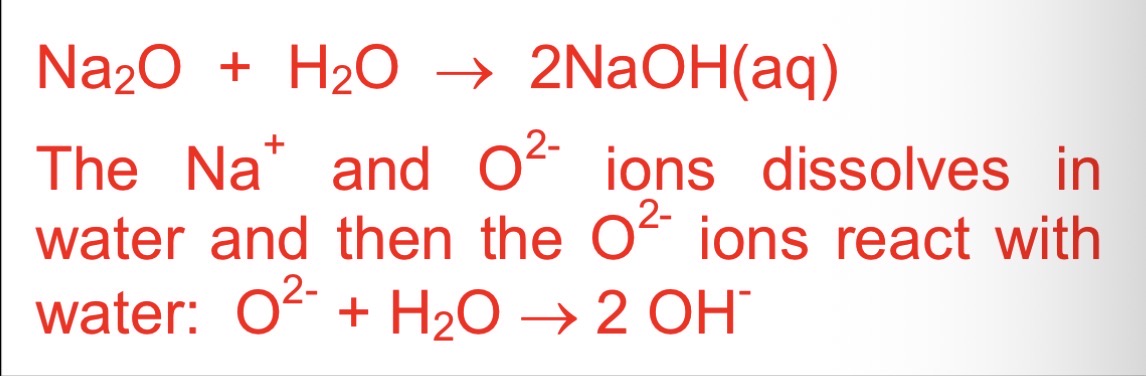
what happens when magnesium reacts with water
slightly soluble
equation between magnesium and water
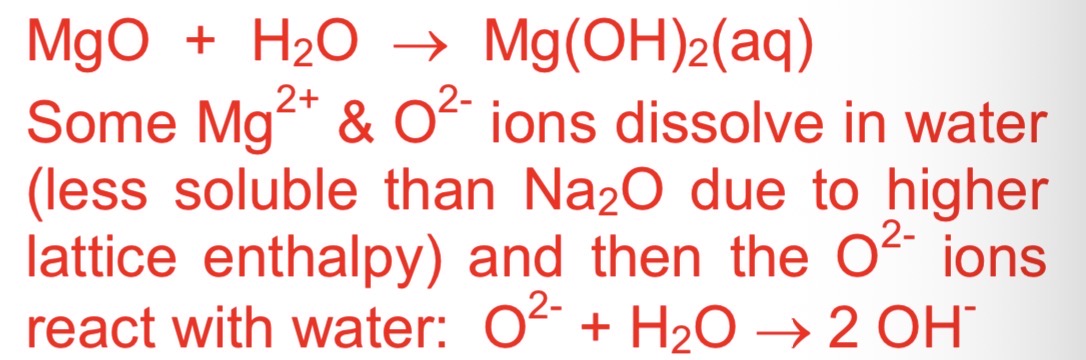
what happens when aluminium oxide reacts with water
insoluble
why is aluminium oxide insoluble in water
very high lattice enthalpy
what happens when silicon oxide reacts with water
insoluble
why is silicon oxide insoluble in water
lattice of atoms linked by strong covalent bonds which require a lot to break
how does phosphorus oxide react with water
violently
equation between phosphorus oxide and water

how does sulfur dioxide react with water
dissolves
equation between sulfur dioxide and water

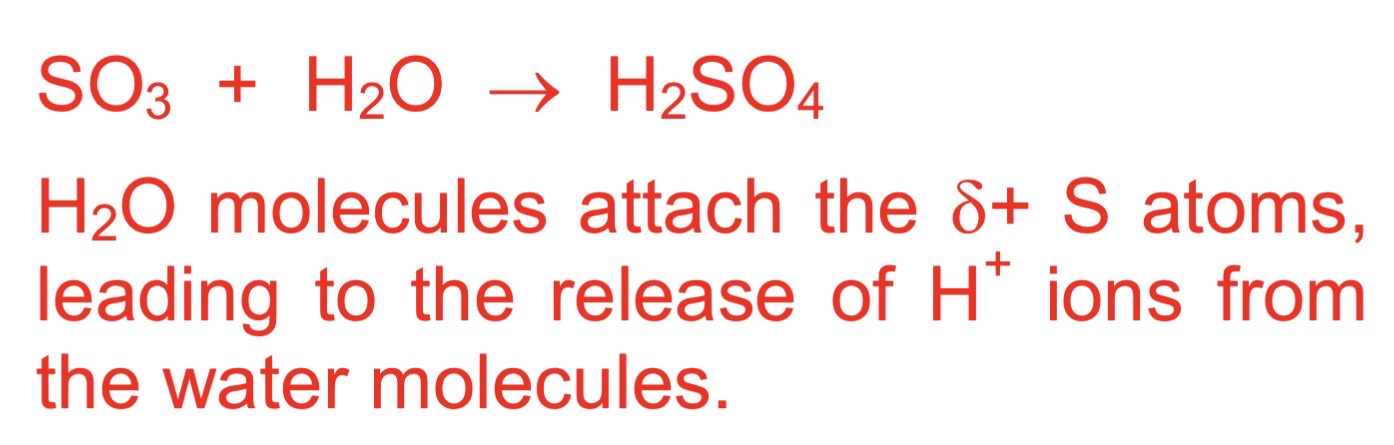
how does sulfur trioxide react with water
violently
equation between sulfur trioxide and water
pH of solution formed when sodium oxide reacts with water
14
pH of solution formed when magnesium oxide reacts with water
10
pH when phosphorus oxide reacts with water
0
pH when sulfur dioxide reacts with water
3
pH of solution formed when sulfur trioxide reacts with water
0
name the basic oxides
sodium oxide,
magnesium oxide
name the acidic oxides
silicon oxide,
phosphorus oxide,
sulfur dioxide,
sulfur trioxide
name amphoteric oxides
aluminium oxide
how does sodium oxide react with an acid
as a base
equation between sodium oxide and acid
Na2O + 2H+ → 2Na+ + H2O
equation between magnesium oxide and an acid
reacts as a base,
MgO + 2H+ → Mg²+ + H2O
equation between aluminium oxide and acids
Al2O3 + 6H+ → 2Al³+ + 3H2O
equation between aluminium oxide and alkali
Al2O3 + 2OH- + 3H2O → 2 Al(OH)4^-
equation between silicon oxide and alkali
SiO2 + 2OH- → SiO3²- + H2O
must be concentrated, hot NaOH
conditions for silicon oxide to react with an alkali
must be hot, concentrated NaOH
equation between phosphorus oxide and alkali
P4O10 + 12OH- → 4PO4³- + 6 H2O
equation between sulfur dioxide and alkali
SO2 + 2OH- → 4 PO4³- + 6H2O
equation between sulfur trioxide and alkali
SO3 + 2OH- → SO4²- + H2O