Ch. 17&18 Cell Cycle and Apoptosis
1/45
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
46 Terms
4 major subdivisions/phases of cell cycle
M: Mitosis = Metaphase-Anaphase Checkpoint → are chromosomes attached
G1: Gap phase = G1 Checkpoint
S: Synthesis → DNA replication
G2: Gap phase = G2/M Checkpoint → pushes to prophase
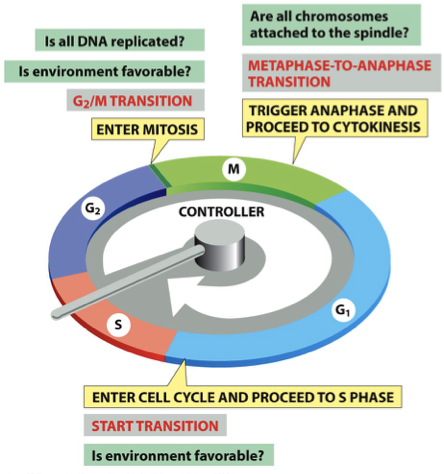
What is Go?
growth arrest = exit cell cycle in G1
quiescence: normal growth arrest
neurons in brain
liver cells enter if chunk is taken out
senescence: induced growth arrest
What happens to Cyclin levels during the cell cycle?
Cdk: cyclin dependent kinase → only active when cyclin is bound
G1 Cyclin
peak concentration at G1 checkpoint
allosterically activiating G1Cdk → phosphorylated
degraded at S phase-Ubiquination in proteosome
M Cyclin
mitotic cyclin
rises during G2 and peaks at G2/M checkpoint
M-Cdk complex
important for M phase/Meta-Anaphase Checkpoint
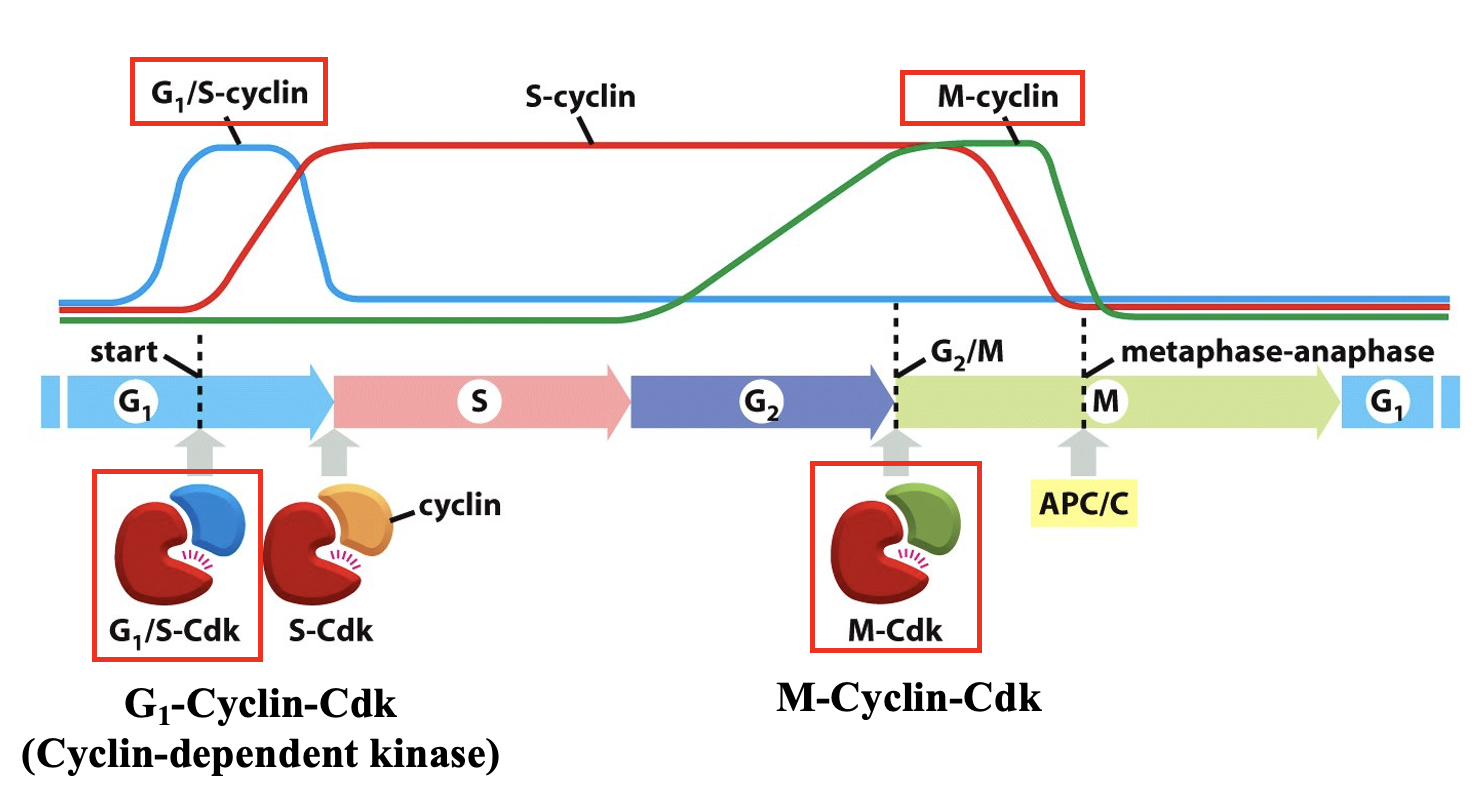
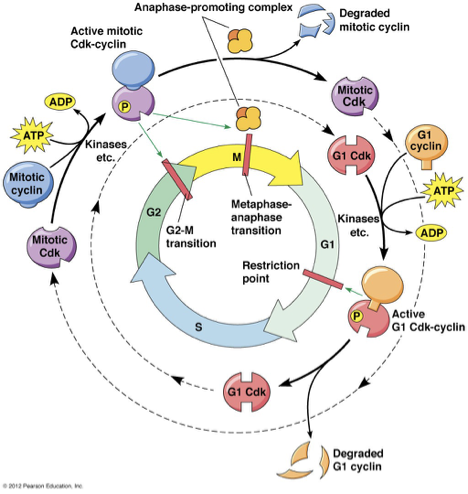
Mammalian Cell Cycle Regulation
prophase?
Targets Mitotic Cyclin Cdk
MAP phosphorylation: mitotic spindle fiber
Condensin phosphorylation: chromosome condensation
Lamin phosphorylation: nuclear envelope breakdown
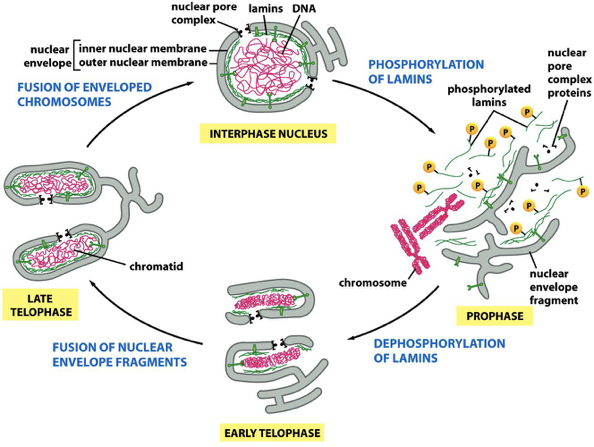
Breakdown and Reassembly of Nuclear Envelope During Mitosis
??
Importance of Cell Death → PCD/Apoptosis
if no PCD in brain = DEATH
brakes for cancer cells
50% of neurons during development of the brain dies
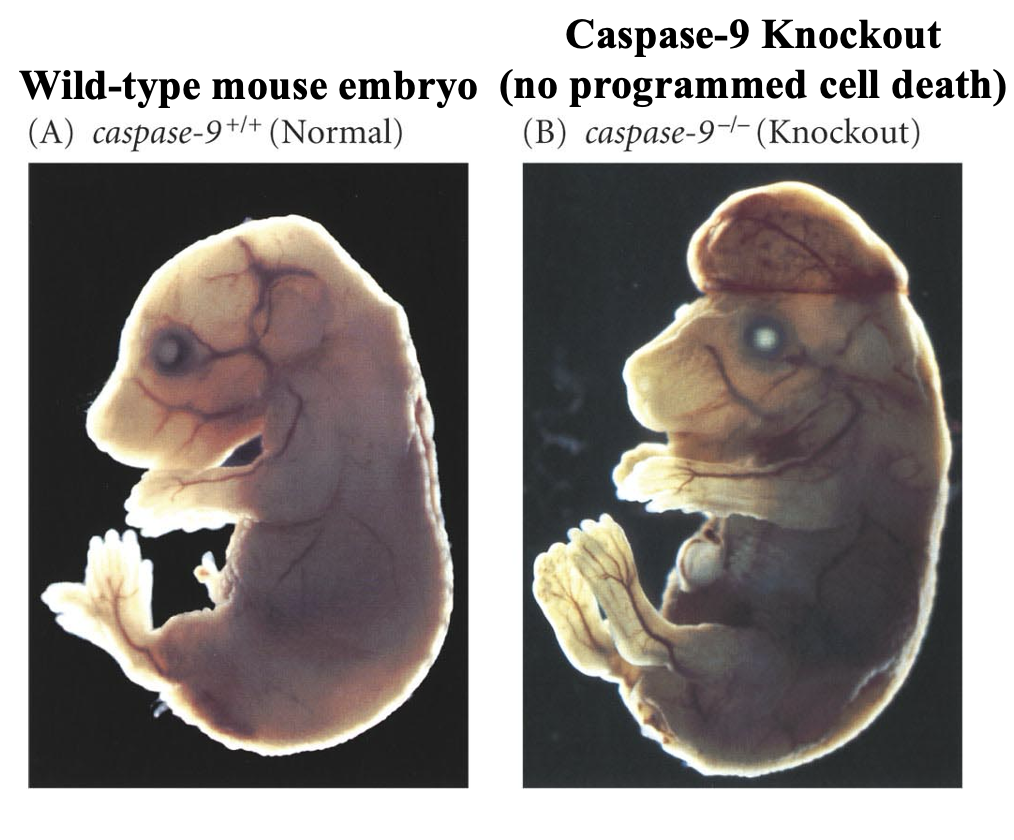
severe development defects and cancer occur if apoptosis is prevented
Ex: Caspase-9 knockout mice has abnormal cells survive causing developmental defects and possibly cancer, while the wild-type embryo removed extra or damaged cells during development
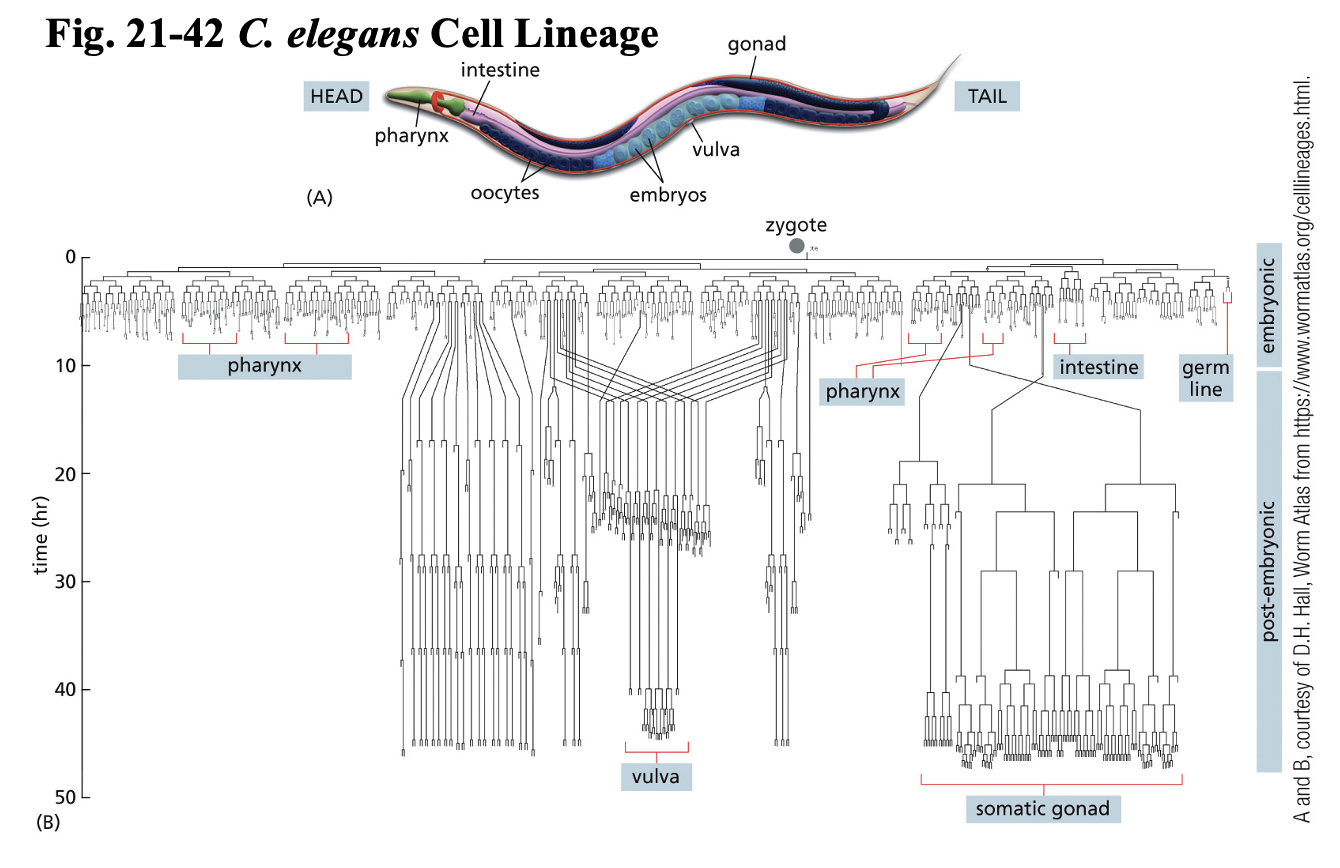
Where was PCD 1st discovered?
C. elegans Nematode Worm → model organism
animal model
transparent embryos
959 somatic cells
Mutant worm → extra 131 cells
led to current understanding of PCD
Cell Death genes discovered = caspase genes
Apoptosis vs. Necrosis
Apop: no swelling/inflammation, neat and tidy cell death
Necrosis: cell swelling → lysis → inflammation
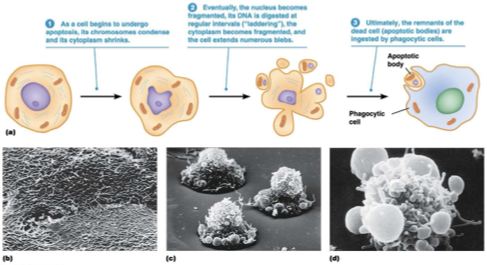
Hallmarks of Apoptosis
shrinkage and detachment from neighbors
phosphotidylserine translocation (inner → outer PM)
membrane blebbing
chromatin condensation
DNA fragmentation between nucleosomes
cell degrades into apoptic bodies → phagocytosed by neighboring cells or macrophages
Induction of Apoptosis
extra cells during development
DNA damage (unrepairable)
cell cycle checkpoint abnormality
What triggers the intrinsic pathway of apoptosis?
Intracellular stress or damage causes mitochondria to release cytochrome c.
What molecule is released from mitochondria during intrinsic apoptosis?
Cytochrome c
What does cytochrome c bind to in the cytosol?
Apaf1 (Apoptotic Protease Activating Factor 1)
What happens to Apaf1 after cytochrome c binds?
Apaf1 changes shape and becomes activated.
What does activated Apaf1 expose?
dATP binding site
Oligomerization domain
CARD domain
What is the role of dATP in apoptosis?
dATP binds Apaf1 and helps assemble the apoptosome.
What is formed when Apaf1 molecules oligomerize?
A wheel-like heptamer called the apoptosome.
What does CARD stand for?
Caspase Recruitment Domain
What is recruited to the apoptosome?
Inactive procaspase-9 monomers
How is caspase-9 activated?
Procaspase-9 monomers dimerize inside the apoptosome.
What does activated caspase-9 do?
It activates executioner caspases.
What is the role of executioner caspases?
They break down cellular components and cause apoptosis.
What is the apoptosome?
A large protein complex made of Apaf1 and caspase-9 that activates apoptosis.
What is the final result of the intrinsic apoptosis pathway?
Programmed cell death (apoptosis)
What does Apaf stand for?
Apoptotic Protease Activating Factor
What does MOMP stand for?
Mitochondrial Outer Membrane Permeabilization
allows cytochrome c to leak out of mitochondria intermembrane space (e- carrier)
Bak and Bak Oligomerization
Regulated by Bcl-2 (B cell lymphoma) family members
What proteins promote MOMP?
Bak and Bax (pro-apoptotic Bcl2 family proteins)
Where is Bak normally located?
Attached to the outer mitochondrial membrane
What activates Bak?
An apoptotic stimulus → extra cell, DNA damaged
What happens to Bak after activation?
Bak changes shape and exposes its BH3 domain and BH3-binding groove.
What allows Bak proteins to oligomerize?
Interaction between exposed BH3 domains and BH3-binding grooves
What is oligomerization?
Multiple proteins joining together into a larger complex
What do Bak oligomers do to mitochondria?
They create openings in the outer mitochondrial membrane.
What is released through the openings caused by MOMP?
Cytochrome c and other proteins from the intermembrane space
What happens after cytochrome c enters the cytosol?
It helps form apoptosomes and activates apoptosis.
How are Bak and Bax thought to form membrane openings?
They likely form large ring-like structures that disrupt the membrane.
What is the role of BclxL?
BclxL is an anti-apoptotic protein that blocks MOMP.
How does BclxL stop apoptosis?
It binds activated Bak and prevents Bak oligomerization.
What happens when Bak oligomerization is blocked?
MOMP and apoptosis are prevented.
Are BclxL proteins pro-apoptotic or anti-apoptotic?
Anti-apoptotic
What are BH3-only proteins like Bad thought to do?
They promote apoptosis by inhibiting anti-apoptotic proteins like BclxL.
How do BH3-only proteins indirectly induce apoptosis?
They block anti-apoptotic proteins, allowing Bak/Bax to cause MOMP.
What is the overall purpose of MOMP in apoptosis?
To release cytochrome c and trigger the intrinsic apoptosis pathway.
Anti apoptotic proteins
keep cell alive
Bcl-2
Bcl-xL
Pro apoptotic proteins
kill cell
Bak: Bcl-2 antagonist/kiler
Bax: Bcl-2 associated x
What does CASPASE stand for?
cysteine-containing aspartate-specific protease