Unit 3.1 Exploring Energy for Living Organisms
1/95
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
96 Terms
All living things need energy to survive, same as at a-
Cellular level
“You are what you eat” means what?
Energy we use and the chemical compounds that make our metabolism work all come from what we eat and drink
Complex system of _________ goes through a constant sequence of _______ that require or release _________
Chemicals; reactions; energy
Define energy
The ability to do work
All living things donate a large part of their existence to what 4 ‘versions’ of the same thing?
Collecting, processing, storing, and using energy
Is energy just related to things like walking and running?
No. Also for thinking, digesting
Define metabolism
Sum of all chemical reactions occurring in a living cell/organism
Where does the word metabolism come from?
Greek work Metabole meaning change
What is an anabolic metabolic reaction?
Metabolic reactions that build larger molecules from smaller molecules/atoms
What is a catabolic metabolic reaction?
Reactions that break large molecules down into smaller molecules/atoms
Is cellular respiration and photosynthesis important? And where do they occur?
Most important, fundamental to all life; take place in the mitochondria and chloroplasts
All living things require-
Energy
Where do living things acquire energy from? Besides physical energy, what is another important use?
Get energy from the food they eat like macromolecules; provide the building material for new cellular matter
What are eukaryotes and prokaryotes?
Types of cells
A eukaryote cell requires what thing for energy?
Glucose
Who has eukaryote cells and give 3 examples
Found in organisms that contain a nucleus surrounded by a membrane and specialized organelles; think vertebrates, protozoa, and trees
What do prokaryotes use for energy?
Glucose and other molecules
Do prokaryotes have organelles? Why or why not? Give an example of a prokaryote
No; they are usually unicellular microorganisms that don’t have a nucleus; bacteria
1 kJ is how many J?
1kJ = 1000J
Define calorie
The amount of energy required to raise the temperature of 1g of water by 1oC
1 calorie is how many kJ?
1cal = 4.184kJ
What does SI stand for?
System of units
How could a person maintain a constant weight?
Energy input from food should match energy output based on the amount you expend while resting plus expending during activities
How would a person lose weight?
Expend more energy than they take it.
How does fat accumulate?
Extra energy ingested but not immediately used is eventually converted to fat
What is the Basal Metabolic Rate (BMR)?
Way to calculate daily energy requirements for basic bodily functions like breathing/digesting/thinking
What is Resting Metabolic Rate (RMR)?
Amount of energy used by a person at rest over a 24 hour period. Energy needed to run the basic energetic needs of organs like heart/lungs/liver/kidneys
How does an active person who eats more keep off weight than a sedentary person who eats less?
Simply by being more active you increase the amount of energy you expend even while resting
What are the 3 common health problems resulting from our modern lifestyle?
Diabetes
Obesity
Cardiovascular disease
Is the rate of diabetes increasing around the world?
Yes, it is now the 4th leading cause of death
How does Type 2 Diabetes develop?
Results from the pancreas’s gradual inability to produce insulin. Cells become resistant to absorbing glucose (sugar) from the blood. A person’s lack of ability to control blood sugar levels
High levels of what cause issues to the kidneys, nerves, feet, digestion?
High blood glucose levels
Diabetes levels are rising in North American youths because of where 2 reasons?
Inactivity and poor diet
Chronically high blood pressure causes what 2 things?
Reduce the ability of cells to respond to insulin and reduce the pancreas’s capacity to make sufficient insulin
How does obesity happen?
Results from the accumulation of excessive amounts of fatty tissue called adipose tissue
Excess adipose tissue and the weight the accompanies it can lead to what 4 things?
Interference with organ system function
Lead to cardiovascular disease
Lead to Type 2 Diabetes
Lead to high blood pressure
What is Body Mass Index (BMI)?
How to calculate body fat. Weight (kg) / Height (M2)
What does a high and low BMI mean?
High BMI means that you are heavier than normal for you height. Low BMI means you’re underweight
What two vague things can affect/lead to cardiovascular disease?
Diet and lifestyle
What are some risk factors that can lead to cardiovascular disease?
High BP, high cholesterol, smoking, stress, alcohol, inactivity, obesity
What are the 2 most common cardiovascular outcomes?
Heart attacks and strokes
How does cardiovascular disease affect the heart?
The heart gets its required energy from blood vessels carrying oxygen, sugars, and other nutrients. Heart disease affects the heart’s structure and functions
What causes a stroke?
Interruption of blood flow in the brain
What is the outcome after a stroke?
Temporary or permanent paralysis; death
Are the numbers of strokes increasing or decreasing?
Decreasing! (3rd leading cause of death in Canada)
What are some factors related to gaining/losing weight?
Cultural background, body image, medications, frequency/intensity of exercise, age, sex, genetics
What does the Canadian Food Guide really emphasize?
Balance among the main types of foods
What is the main difference from the 1977 Canada Food guide to the 2019 guide?
2019 no longer has servings
What are some things the Canada Food Guide offers as guidelines?
Plenty of fruits/veggies, eat protein, make water your drink of choice, whole grain foods, be mindful of habits, cook more often, use food labels, limit food with sodium/sugars/saturated fats, be mindful of food marketing
What is a dietician?
Experts in food and nutrition; help create balanced menus and educate people about healthy eating and lifestyles
How does the Law of Conservation of Energy relate to food?
Energy cannot be created or destroyed so food must become either energy or fat
What is an example of a transfer of energy between living and non-living things (biotic and abiotic) in an ecosystem?
Photosynthesis
What are some forms of energy?
Light, sound, mechanical energy, chemical energy, electrical energy, radiation, heat
Define kinetic energy
Energy that causes objects to move; energy that is being used up
Define potential energy
Energy that is stored for later use
In an example of a ball rolling down a hill at three points (top, mid-roll, bottom), where is kinetic energy present and where is potential energy?
At the top of the hill there is potential, while rolling there is both, and stopped at the bottom there is neither
In the ball rolling example, is energy added if you were to carry the ball back to the top of the hill?
Yes, adding potential energy so that it can be released again as kinetic energy when it rolls down once more
Define thermodynamics
Branch of science that deals with the relationship between all forms of energy
What is the first law of thermodynamics?
Energy can be changed from one form to another but it can’t be created or destroyed. The amount of energy in the universe is always conserved
What is an example of the first law of thermodynamics? (not created or destroyed only changed)
Turning electricity into heat
Whenever energy is transformed into another form there is always-
loss. Not destroyed but unusable for further work
In an equation, reactants and products go on which side?
Reactants → Products
What is the second law of thermodynamics?
Disorder (entropy) in the universe is always increasing. Each time energy is used some will be converted (lost) to heat (random motion)
Which is more likely to occur- order or disorder?
Disorder
What is needed to create or maintain order?
Energy
Give a basic example describing the second law of thermodynamics
A sandcastle can fall apart but it can’t rebuild itself
Define high entropy
High disorder; energy is dispersed, evenly distributed, and unavailable to do work
Define low entropy
Low disorder; energy is concentrated, high organization, and high potential for useful work
What happens to energy during reactions?
The chemical energy stored in the bonds of reactants and products is changed during the course of chemical reactions
What is an endergonic reaction?
A reaction where the reactants have less energy that their products. They will absorb energy- increase in stored energy. Require continual energy input until the reaction is completed. Reaction must go above the target energy level until it settles into a stable energy plateau
Is an endergonic reaction spontaneous or non-spontaneous?
Non-spontaneous
The activation energy of endergonic reactions is usually prohibitively high. What is needed?
Enzymes
What is an exergonic reaction?
Reactants have more stored energy in their bonds than their products release energy. Does not require energy inputs. When the reaction starts the reactants are at a higher amount of energy than the products. Energy is released because the products have less energy.
Are exergonic reactions spontaneous or non-spontaneous?
Spontaneous
Does activation energy need to be overcome in exergonic reactions?
Yes. Initial amount of Ea must be supplied to destabilize the reactant’s bond so that they may be broken and reformed.
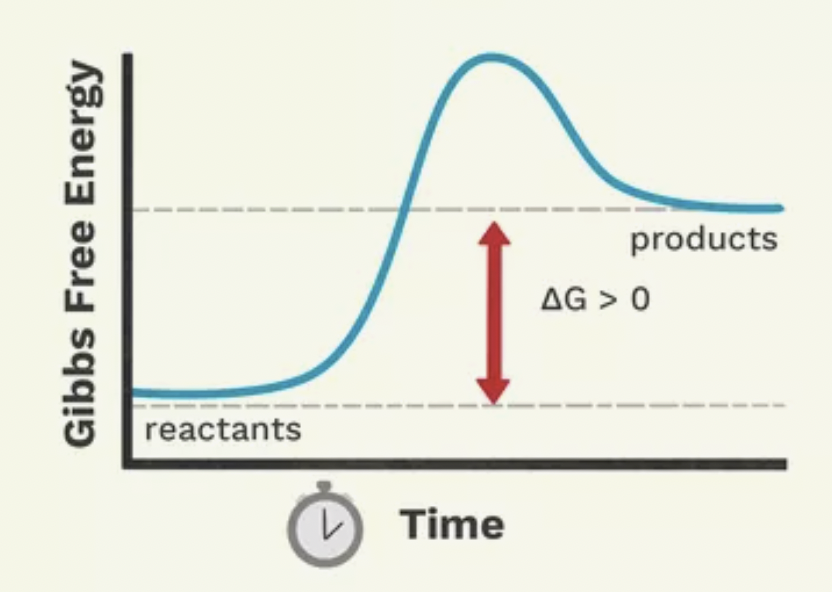
Is this an example of an endergonic or exergonic reaction?
Endergonic reaction
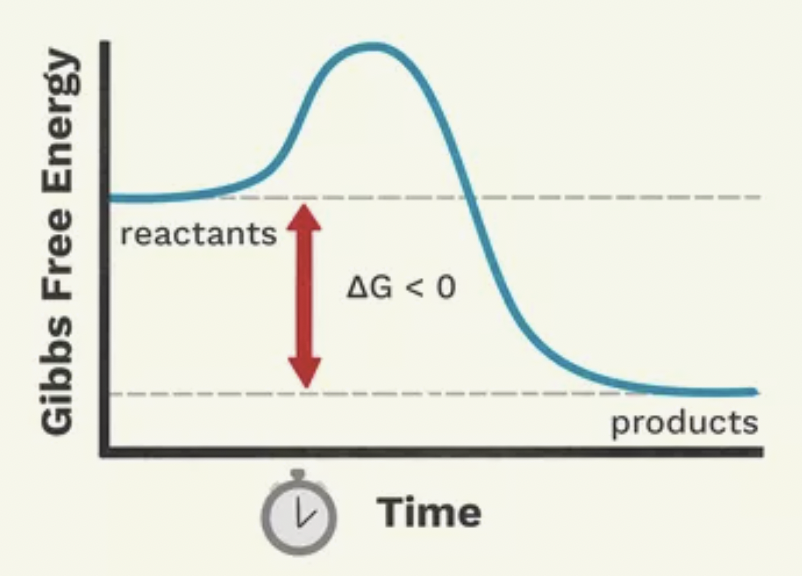
Is this an example of an endergonic or exergonic reaction?
Exergonic reaction
What is mnemonic device to remember redox reactions?
OIL RIG; oxidation is loss of electrons, reduction is gain of electrons
What is oxidation? They are called ____ agents
Loss of electrons and gain of oxygen; substances that are oxidized are ‘reducing agents’ because they cause the reduction of other substances
What is reduction? They are called ____ agents
Gain of electrons and loss of oxygen; Substances that are reduced are ‘oxidizing agents’ because they cause the oxidation of other substances
Can oxidation and reduction happen seperately?
NO. They occur simultaneously
During electron transfer between molecules/atoms, is there energy present?
Yes! Energy can be either stored or released
What happens to energy when a reduced molecule receives an electron from an oxidized molecule?
Reduced molecules are raised to higher energy levels; meanwhile, oxidized molecules have lowered energy levels
Why would a reduced molecule that has gained electrons be raised to to a higher energy level?
Because electrons repel each other; more electrons increases the potential energy of the atom
Can coenzymes/energy carriers become reduced or oxidized?
Yes. Because electrons are shuttled from one enzyme to the next through metabolic pathways (delivering or harvesting energy at each stage)
What is Nicotinamide Adenine Dinucleotide (NAD+) and what is it used for?
It is a coenzyme important in the transfer of energy
What is the purpose of Adenosine Triphosphate (ATP)?
All living things need energy for their metabolism. Energy is carried and released by ATP!
Cells can only get their energy from what?
Glucose
One molecule of glucose is 3012kJ/mol which is WAY more energy required for most enzyme-catalyzed reactions. What do cells do with this extra energy?
Cells save unused energy by storing it into smaller packets called ATP
1 mole of ATP stores and releases 54kJ or energy. Is that close to 1 mole of glucose?
No! Less than 2%
What is ATP “called”?
“The currency of the cell”
What is an analogy of glucose to ATP using money?
Glucose is a $100 bill and ATP is the $1 coins of change you get when you break a large bill; cells make change and store loonies
What is ATP made of?
An adenosine subunit and 3 phosphate groups
Is the release of energy by ATP endergonic or exergonic?
Exergonic *gives off energy*
What is the ATP to ADP to ATP cycle?
ATP → loses phosphate → energy released → ADP → new phosphate → energy absorbed from food
How is New Zealand’s Food Guide different from Canada’s?
Has more broad information but generally the same; more exercise, low-fat milk, lean protein, minimal added fat, low in salt, little added sugar, limit alcohol, cook > buy