Inorganic and Organic Transition Metal Chemistry & Catalysis Principles
1/26
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
27 Terms
What are the types of orbitals in organic chemistry?
1x S orbital and 3x P orbitals, totaling 4 orbitals.
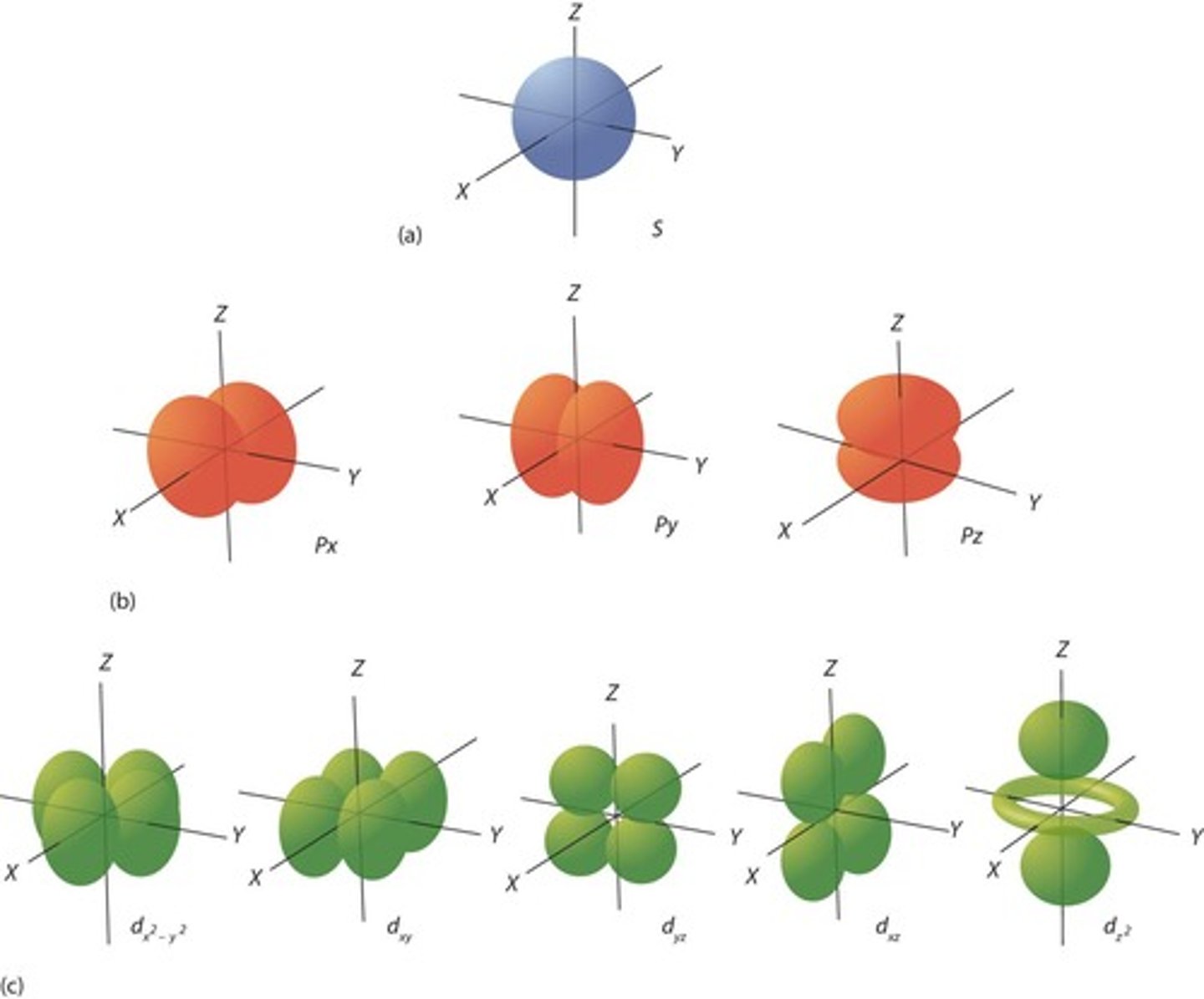
What is the total number of orbitals in inorganic chemistry?
1x S orbital, 3x P orbitals, and 5x D orbitals, totaling 9 orbitals.
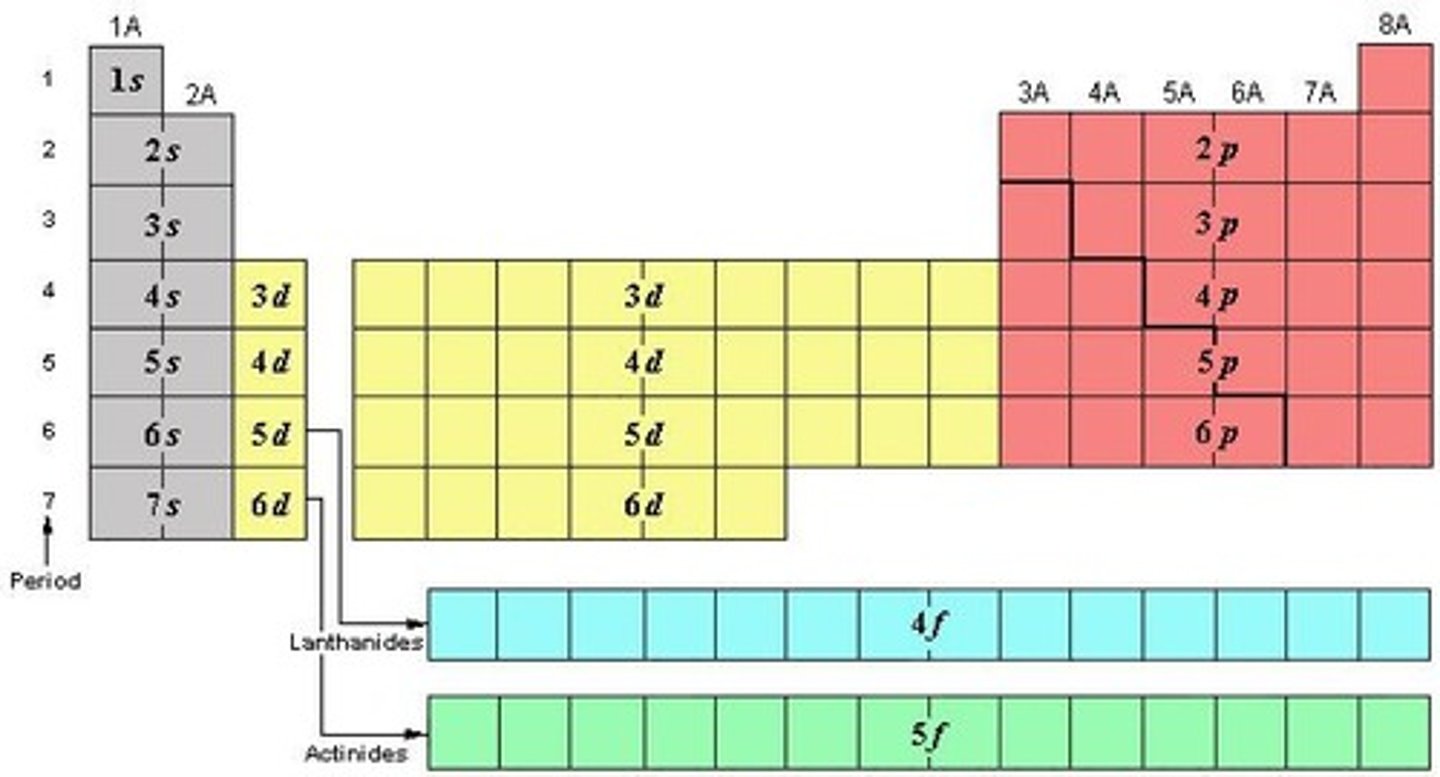
What is the Octet Rule?
Atoms tend to bond in such a way that they have eight electrons in their valence shell.
What is the 18 Electron Rule?
A guideline that states that stable transition metal complexes typically have 18 valence electrons.
What are ligands?
Substituents bonded to transition metals that can alter the behavior of the metal center.
What is the difference between formal charge and oxidation state?
Formal charge assumes equal sharing of electrons, while oxidation state assigns electrons to the more electronegative atom.
How is formal charge calculated?
FC = # valence electrons - ½ (# bonding electrons) - (# nonbonding electrons).
What is a catalyst?
A substance that increases the rate of a chemical reaction without being consumed.
What is the Haber-Bosch process?
A method for synthesizing ammonia from nitrogen and hydrogen, consuming about 1% of the world's electricity.
What are the strategies of catalysis?
Enzymes, organocatalysis, Lewis acid catalysis, photocatalysis, and transition metal catalysis.
What is a catalytic cycle?
The series of steps by which reagents are consumed, products are formed, and the catalyst is regenerated.
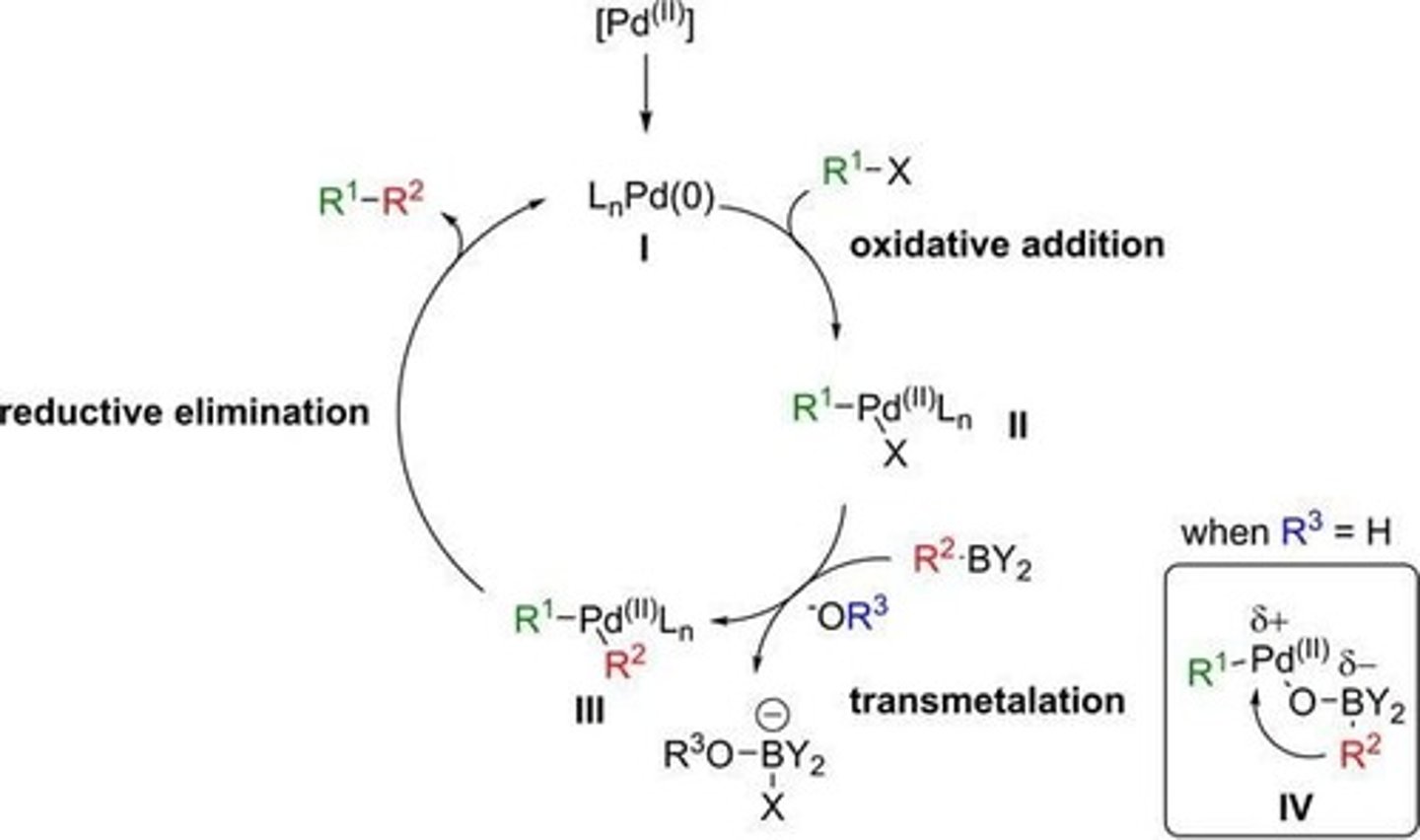
What are the steps in a palladium cross-coupling reaction?
Oxidative addition, transmetalation, and reductive elimination.
What is the significance of Pd(II)OAc2 in palladium catalysis?
It is a precatalyst, not the catalyst itself.
What is the role of bulky phosphines in palladium couplings?
They are critical components that stabilize the palladium during the reaction.
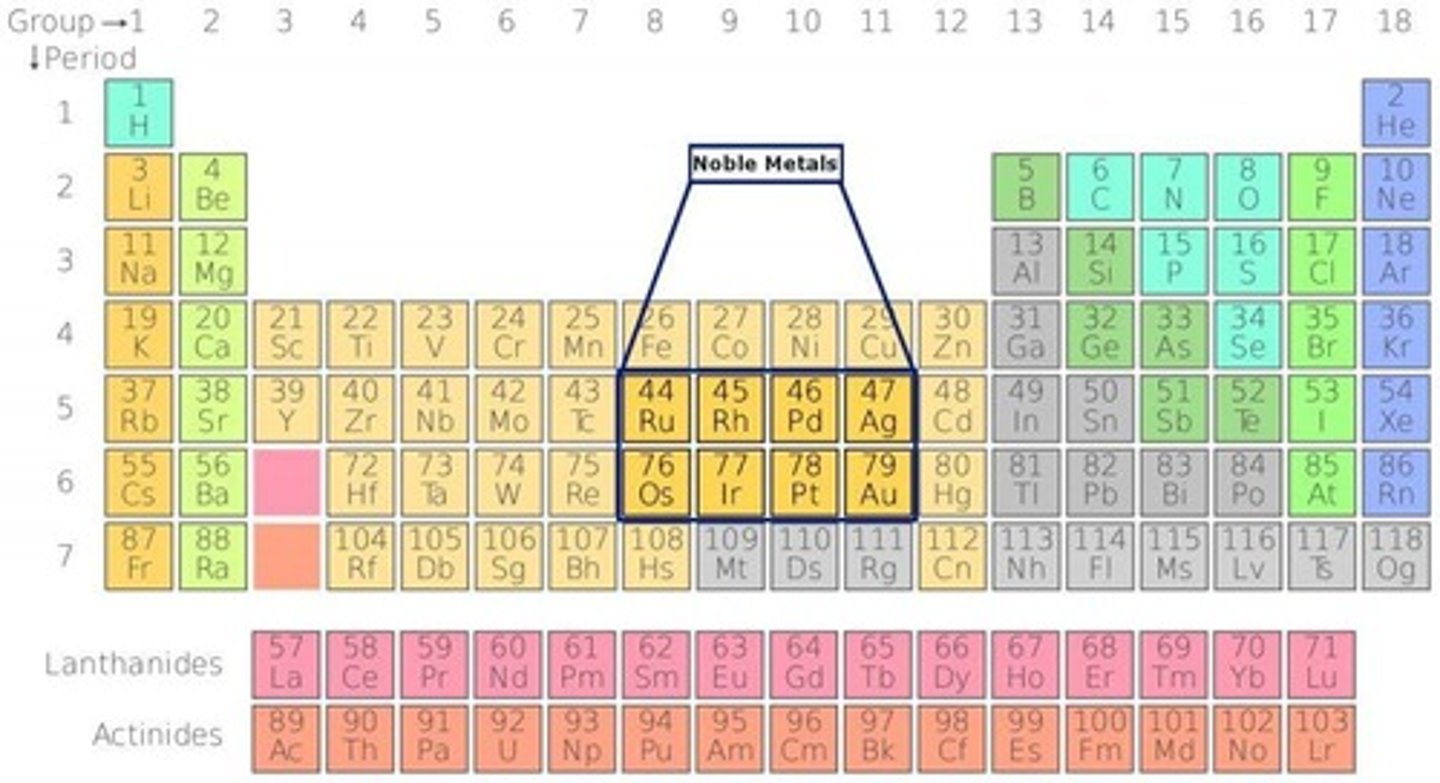
What is the price of palladium compared to other noble metals?
Pd is approximately $5,090.00/mol, while Au is $29,400.00/mol and Pt is $12,300.00/mol.
What is the rate-determining step in a cross-coupling reaction?
The transmetalation step, where the boronic acid R group displaces the X ligand bound to Pd.
What does it mean for Pd(0) to be a d10 metal?
It has a full d orbital, which creates steric strain that can be relieved by forming bonds.
What is the purpose of introducing ligands to a metal?
To alter the properties and reactivity of the metal.
What is the relationship between oxidation state and electron density?
The oxidation state affects how electron density is distributed around the metal.
What is the significance of understanding the oxidation state of a metal?
It helps predict the metal's behavior and reactivity in chemical reactions.
What is the role of ligands in determining the oxidation state of a metal?
Ligands contribute to the overall charge of the complex, affecting the oxidation state.
What is the electronic character of an atom assessed by?
Both formal charge and oxidation state.
What is the impact of changing the oxidation state of a metal?
It alters the metal's chemical behavior and properties.
What is the significance of the Nobel Prizes in Chemistry related to transition metal catalysis?
They highlight advancements in the field, recognizing contributions to understanding and utilizing transition metals.
What is the main advantage of using catalysts in chemical reactions?
They allow reactions to occur more efficiently and often under milder conditions.
What is the main focus of organometallic chemistry?
Using metals to perform organic transformations.
What is the purpose of stabilizing multiple transition states in catalysis?
To make otherwise inaccessible reaction pathways possible.