Unit 6 Nomenclature
1/23
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
24 Terms
what functional group has highest priority?
Carboxylic acids
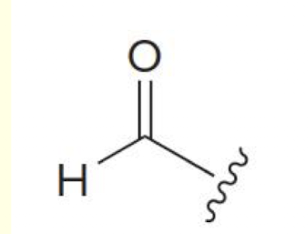
how to name when functional group is aldehyde:
choose longest carbon chain containing the CHO group
Parent name will end in -al
number from end nearer the CHO group
** if attached to a ring, call it a carbaldehyde (ring name + carbaldehyde)
how to name when functional group is ketone:
**ketones stay below aldehydes in priority
select longest chain containing the carbonyl group
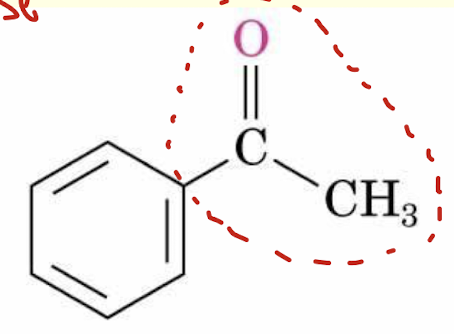
Parent name ends in -one
number from end nearer the carbonyl group
when both an aldehyde and C=O add in “oxo—”
aldehyde C is #1 in chain (ending is now —al)know the acyl groups, and enal vs. enone
***acyl groups as substituents get -yl OR -oyl
molecules with alkene + aldehyde =
enal
molecules with alkene + ketone =
enone
formyl group (acyl group)
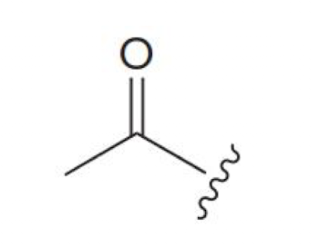
acetyl group (acyl group)
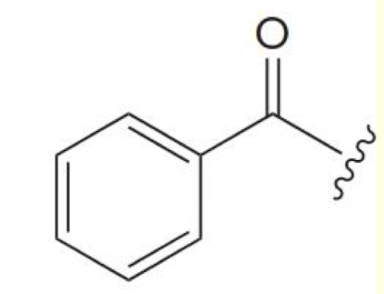
benzoyl group
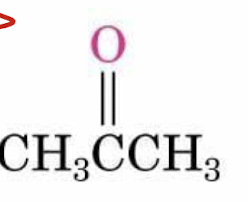
acetone structure (non-IUPAC name for this kind of ketone)
acetophenone structure (non-IUPAC name for this kind of ketone)
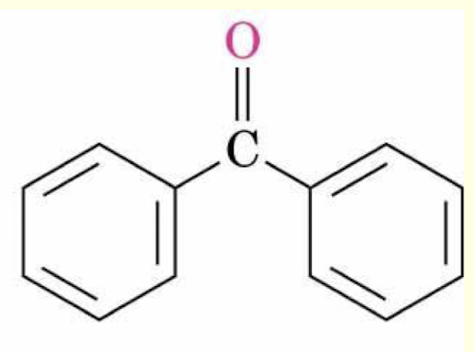
benzophenone structure (non-IUPAC name for this kind of ketone)
how to name when RCOOH (carboxylic acid) present (highest priority functional group):
choose longest carbon chain containing the COOH group
Parent name ends in —oic acid
non iupac: -ic acidnumber from end nearer the COOH group
if ring present:
- name using the suffix —carboxylic acid
- the CO2H carbon itself is not numbered in this case (just the ring is)
if two carboxylic acids present:
name using the suffix —dioic acid
if carboxylate salt (such as RCOONa, aka it lost the H )
suffix = —ate
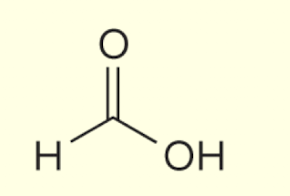
methanoic acid (non IUPAC common carboxylic acids to know)
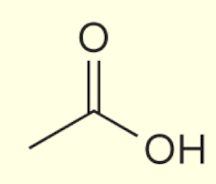
ethanoic acid (non IUPAC common carboxylic acids to know)
benzoic acid (non IUPAC common carboxylic acids to know)
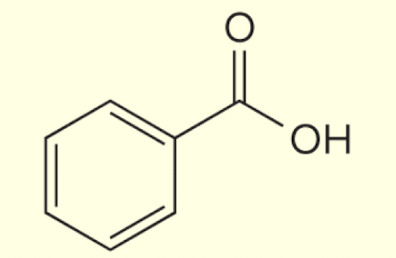
how to name when RC=-N (=- is triple bond) present :
named by adding —nitrile as a suffix to the alkane name with nitrile carbon numbered C1
some are named as derivatives of carboxylic acids, know both non iupac and iupac way
non-iupac naming for nitrile as derivative of carboxylic acid:
replace —ic acid or —oic acid with —onitrile
iupac naming for nitrile as derivative of carboxylic acid:
replace -carboxylic acid with —carbonitrile
how to name when acid halide RCOX present :
replace the —ic or —oic acid ending with —yl or the —carboxylic acid ending with —carbonyl
then specify the halide
** oic = oyl and ic = yl
how to name when acid anhydrides present (combine 2 RCOOH (by dehydrating them together) to make RCO2COR’):
if symmetrical, replace “acid” with “anhydride” based on the related carboxylic acid
if unsymmetrical, cite the two acids alphabetically and add anhydride at the end (as separate word always)
how to name when Ester present (RCOOR’):
name R’ and then, after a space, the carboxylic acid with the “—ic/—oic acid” ending replaced by “—ate” or “—oate”
AKA: name what is attached to the “O” first then what is attached to the “C=O”
name the carboxylate anion second
how to name when amide (RCONH2) present :
for unsubstituted —NH2 group: replace —oic acid or —ic acid with —amide
or replace the —carboxylic acid ending with —carboxamideif N is substituted: identify the substituent groups (with N— before each) and then the parent amide