BIOL-L 112 Ch 4
1/20
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
21 Terms
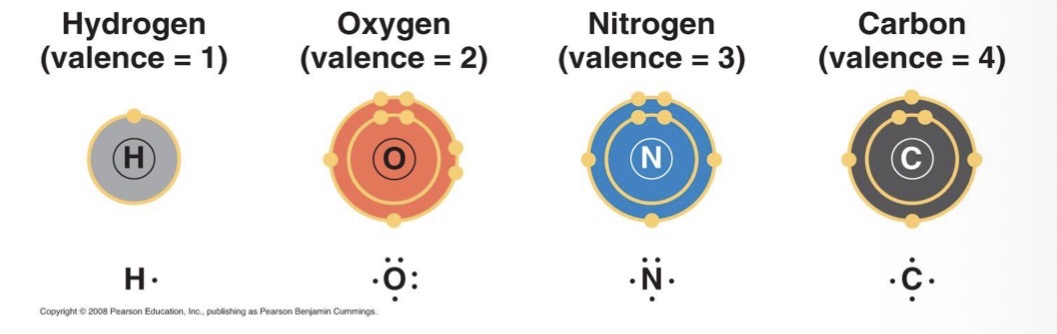
Valence
The number of unpaired electrons required to complete the atom’s outermost shell (or the number of covalent bonds an atom can form)
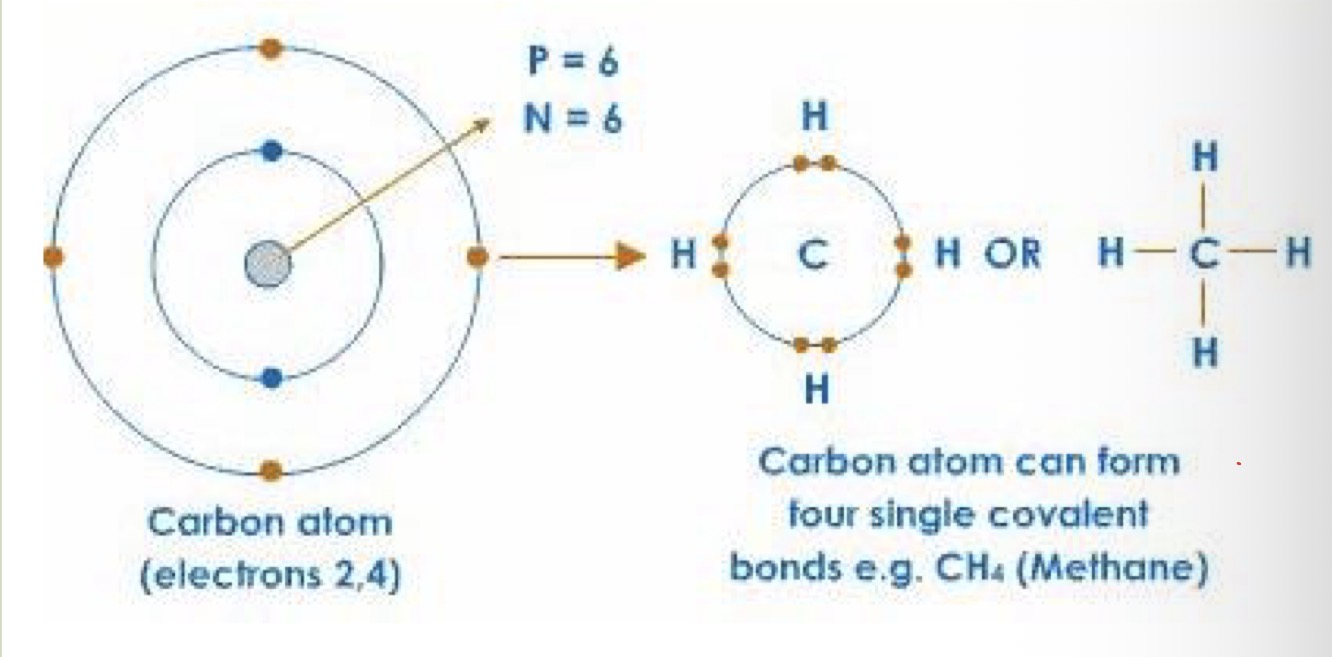
What are the 4 major atomic components of organic molecules?
CHON (carbon, hydrogen, oxygen and nitrogen)
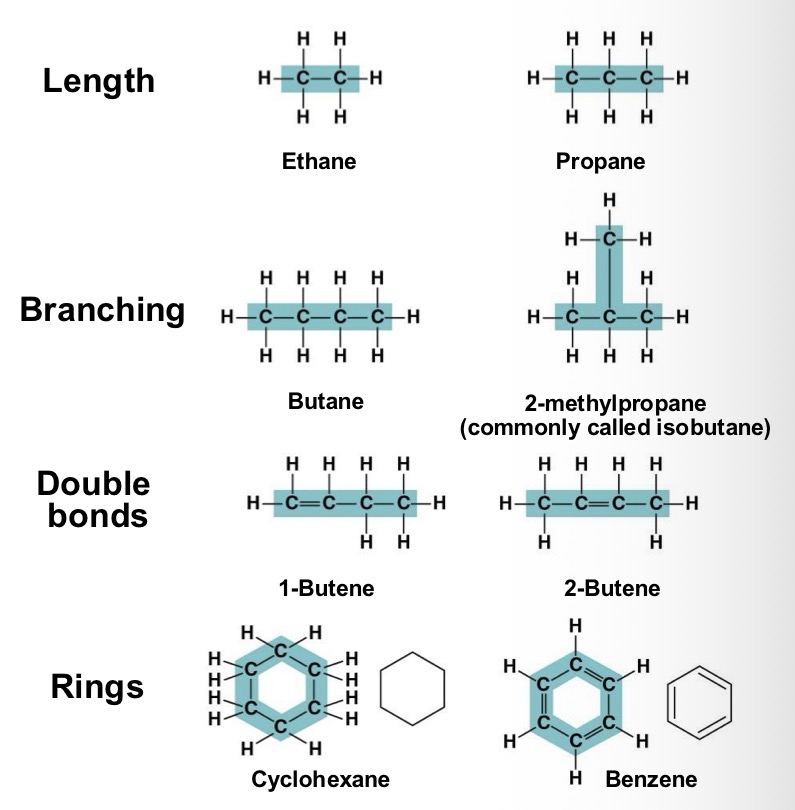
Carbon chains form the skeletons of most organic molecule. Skeletons can vary in length and shape. Why does this variation matter?
It contribute to molecular diversity
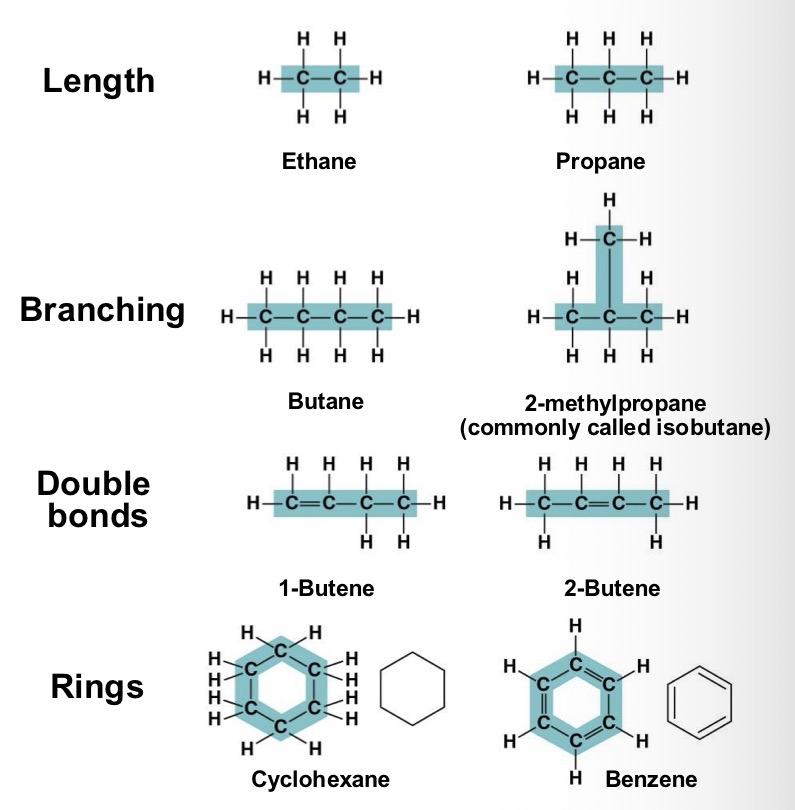
All the molecules shown are what? And how present are they?
Hydrocarbons. They are rare in living organisms, however many organic molecules have regions consisting of only carbon and hydrogen?
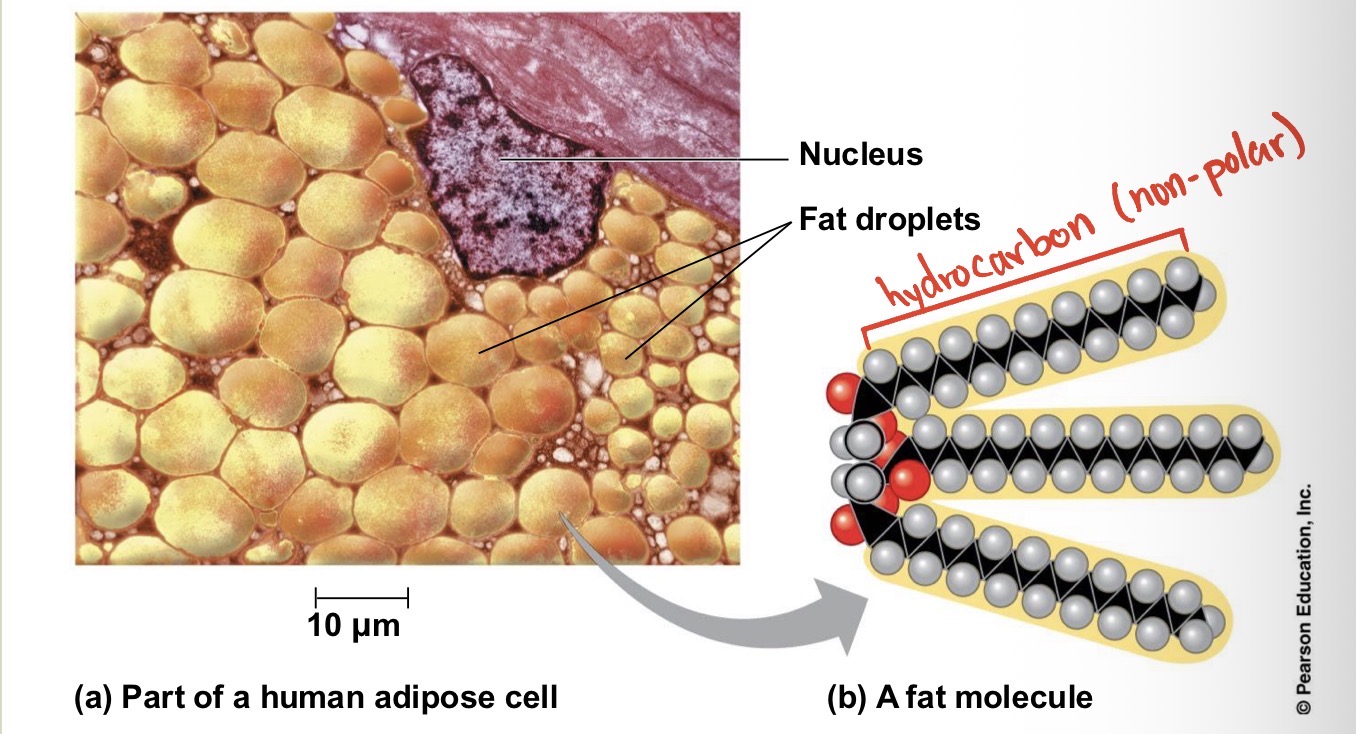
Many organic molecules, such as fats, have hydrocarbons. What can the hydrocarbon tail of a fat molecule do?
It can be broken down to release a large amount of energy
Isomers
Compounds with the same molecular formula, but different structures and properties
Structural isomers
Different covalent partners
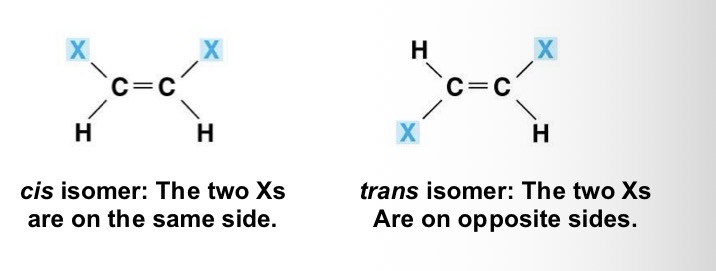
Cis-trans isomers
Different arrangement about a double bond
Enantiomers
Different arrangement around asymmetric carbon
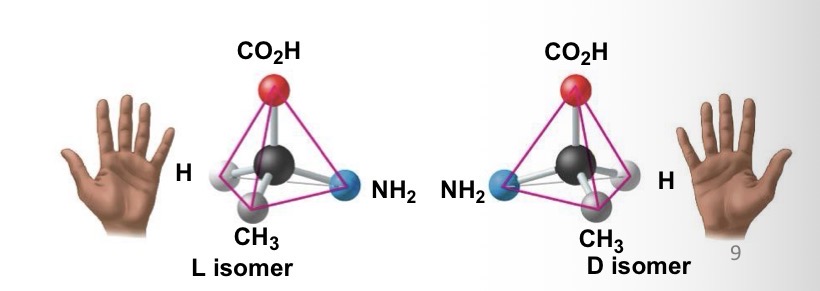
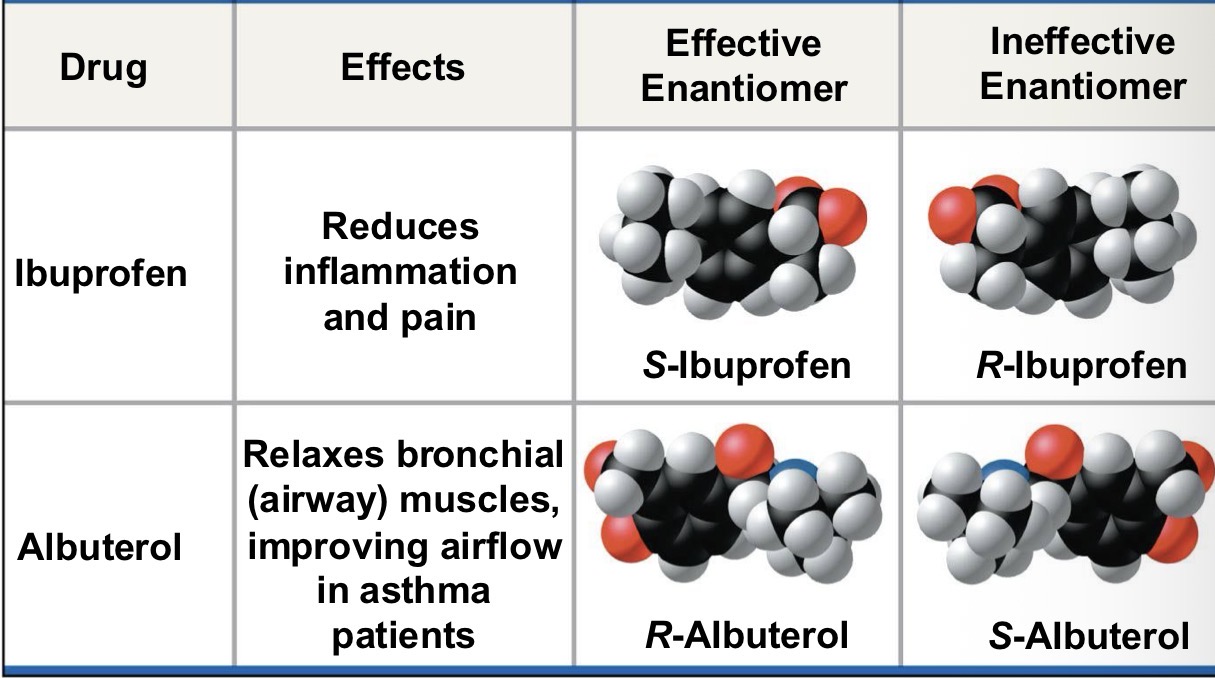
Ibuprofen and Albuterol are what?
Enantiomer.
ibuprofen is sold as a mixture of S and R form
S form is 100X more effective than R form
Albuterol is sold as R form
S form counteracts R form
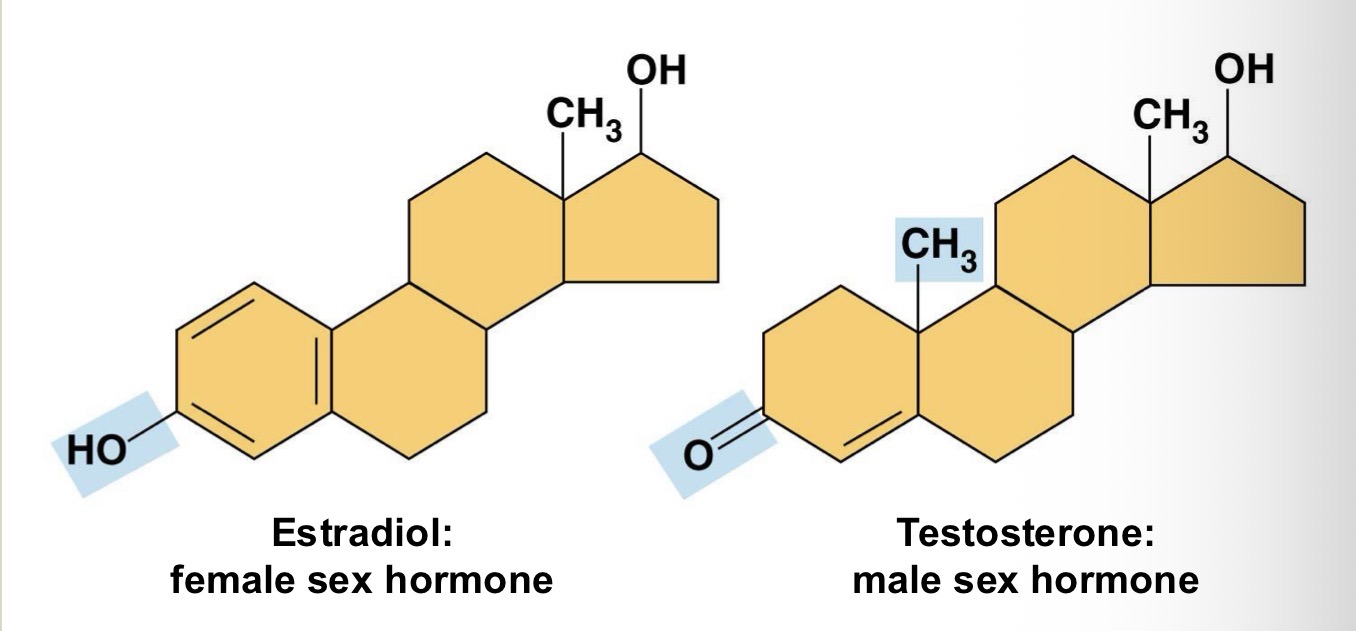
Fill in the blank: Both estradiol and testosterone have a common “steroid nucleus” bu differ in _________ _____ that are attached to the nucleus.
Chemical groups
What are the 7 chemical groups most important in the chemistry of life?
Hydroxyl
Carbonyl
Carboxyl
Amino
Sulfhydryl
Phosphate
Methyl
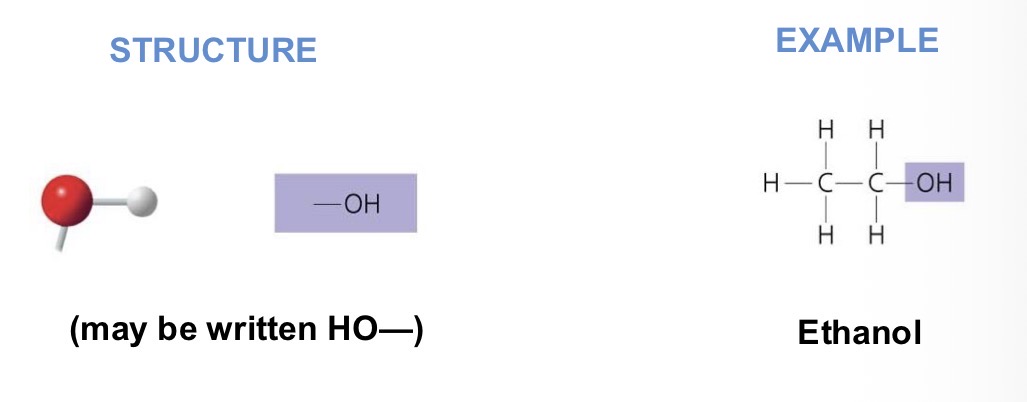
What chemical group is this?
Functional properties:
polar
Attracts water molecules, helping dissolve organic compounds (i.e. sugars)
Alcohols (their specific names usually end in -ol)
Hydroxyl
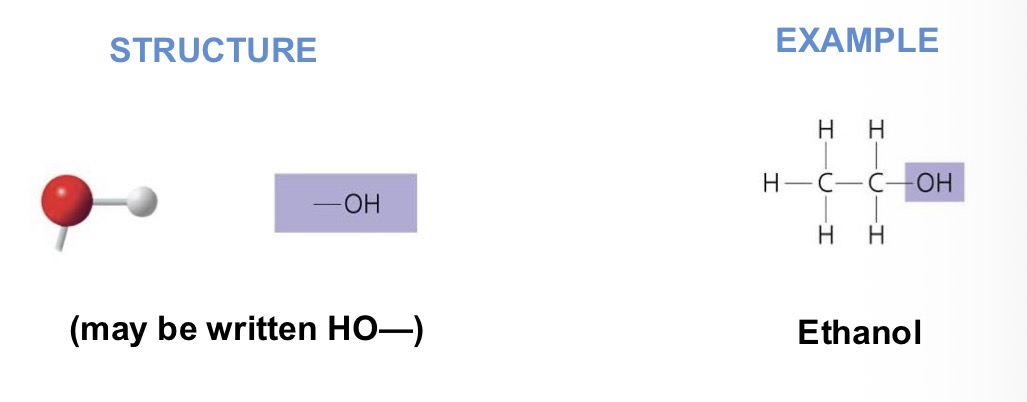
Why does Hydroxyl attract water molecules?
It’s polar

What chemical group is this?
Functional properties:
polar
Found in sugars
Carbonyl
What is the difference between ketones and aldehydes?
hint: where does a key go?
Ketones if the carbonyl group is within a carbon skeleton.
Aldehydes if the carbonyl group is at the end of the carbon skeleton


What chemical group is this?
Functional Properties:
polar
Acidic properties
Carboxyl

What chemical group is this?
Functional properties:
acts as a base
Polar
Amine
What chemical group is this?
Functional properties:
2 sulfhydryl groups can interact to help stabilize protein structure (disulfide bond)
Sulfhydryl


What chemical group is this?
Functional Properties:
polar
Contribute negative charge
Transfer energy
Phosphate
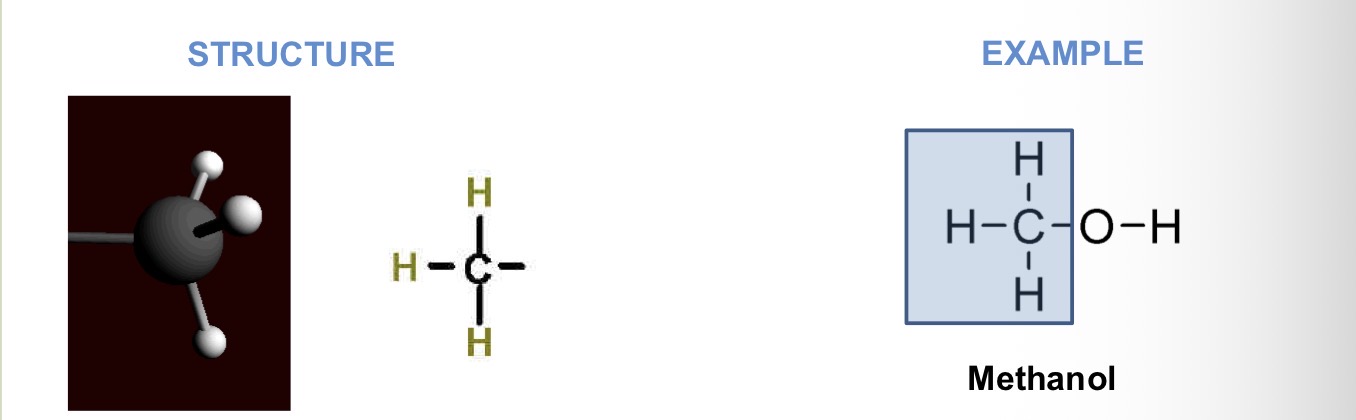
What chemical group is this?
Functional properties:
affects gene expression (help turn gene off)
Affects molecular shape and function
Hydrophobic
Sort of nonpolar
Methyl