Exam 3 Self-Care (Bach)
1/69
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
70 Terms
Harmful Components
Nicotine (addiction)
Tar (lung damage, cancer)
Carbon monoxide (↓ oxygen delivery)
Nitrosamines (carcinogens)
Benzene (leukemia risk)
Formaldehyde (toxic irritant)
Absorption: buccal (gum/lozenge), dermal (patch), pulmonary (smoking, fastest).
Distribution: rapid CNS penetration; crosses placenta.
Metabolism: hepatic CYP2A6 → cotinine (half‑life ≈ 20 h).
Excretion: renal.
Dopamine reward pathway: nicotine stimulates mesolimbic dopamine release → reinforcement of smoking behavior → addiction
List four clinically significant drug interactions associated with tobacco smoking.
CYP 1A2 substrates
↓ levels of:
Theophylline
Clozapine
Olanzapine
When patient quits → levels increase → toxicity risk
Oral contraceptives
↑ thromboembolism risk
↓ effectiveness
Caffeine
Smoking ↑ metabolism → quitting causes jitteriness
May need caffeine reduction
Warfarin (and other vitamin K antagonists)
Smoking ↑ metabolism → ↓ INR
Monitor INR closely when quitting.
Withdrawal effects (≈ 6‑8 days peak)
Irritability
Anxiety
Poor concentration
Increased appetite
Insomnia
Depressed mood
Health benefits
↓ CV risk
Improved lung function
↓ cancer risk
Better overall health
Ask → Identify tobacco use
“Do you smoke or use any tobacco products?”
Identify use as a vital sign
Advise → Strong personalized recommendation
“Quitting is the single most important thing you can do for your health.”
Deliver strong, personalized recommendation
Assess → Readiness to quit
“Are you ready to quit in the next month?”
Gauge readiness
Assist → Medication + plan
Provide medication choice, set quit date, develop plan
Support quit attempt
Arrange → Follow-Up
“Let’s schedule a follow‑up in 2 weeks.”
Ensure ongoing support
Stage 1: Not Ready to Quit (Next Month)
Patient Profile:
Ambivalent about quitting
Pros of tobacco use outweigh cons
Low motivation
Goal: Start thinking about quitting
Counseling DOs:
Strongly advise to quit
Help patient find personal motivation by:
Providing information
Asking noninvasive questions about tobacco use
Raising awareness of health risks
Demonstrate empathy and foster communication
Stage 1: Not Ready to Quit (Next Month)
Techniques: The 5 R’s
Relevance – make quitting personally relevant
Risks – highlight health consequences
Rewards – identify benefits of quitting
Roadblocks – discuss barriers and strategies
Repetition – revisit motivation regularly
Stage 2: Ready to Quit (Next Month)
Patient Profile:
Aware of need and benefits of quitting
Preparing to take action
Goal: Achieve cessation
Key Counseling Elements:
Assess tobacco use history
Current use (type, amount)
Past use and quit attempts
Confidence in ability to quit
Triggers and routines associated with use
Discuss key issues
Weight gain concerns
Withdrawal symptoms (peak 1 week, resolve 2–4 weeks)
Living with smokers and social triggers
Alcohol and socializing triggers
Situational triggers (after meals, boredom, etc.)
Practical counseling
Set a quit date
Use Tobacco Use Log to track triggers and habits
Teach cognitive coping strategies (positive self-talk, mental rehearsal)
Teach behavioral coping strategies (environment control, substitutes like gum/water)
Promote social support and stress management
Discuss pharmacotherapy as part of plan
Follow-Up:
First week post-quit, first month, then as needed
Congratulate successes and address slips
Stage 3: Recent Quitter (≤6 Months)
Patient Profile:
Quit within past 6 months
Experiencing withdrawal
At risk for relapse
Goal: Maintain abstinence for at least 6 months
Counseling Focus:
Evaluate quit attempt and medication adherence
Identify triggers and temptations (stress, social situations, cravings)
Promote healthy replacement behaviors
Reinforce coping strategies
Discuss slips vs. relapse: “let a slip slide”
Stage 4: Former User (>6 Months)
Patient Profile:
Quit >6 months
Still vulnerable to relapse
Goal: Remain tobacco-free for life
Counseling Focus:
Assess quit status and congratulate success
Address any slips or relapse
Review relapse prevention strategies
Continue support and follow-up as needed
Moderate smoker → Patch daily OR Gum PRN cravings
Depressed patient → Bupropion + patch
Heavy smoker → Varenicline OR combination NRT

GUM + LOZENGES
Dose: 2 mg if first cigarette >30 min after waking; 4 mg if ≤30 min. Minimum 9 pieces/day for first 6 weeks.
Advantages: Oral substitute; titratable; inexpensive
Disadvantages: Requires proper chewing; oral irritation, hiccups; may interfere with dental work
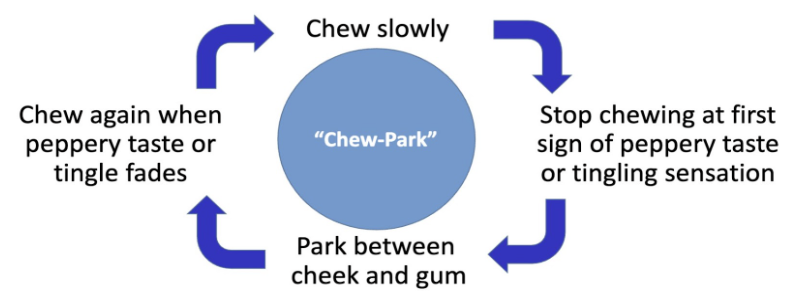
Nicotine polacrilex gum'
Gum provides a “busy‑hands” feel and lets you adjust the nicotine release by chewing more or less; it also offers a 6 mg option for those who need a higher dose.
Nicotine nasal spray
Dose: 0.5 mg per spray; 1 mg total dose = 2 sprays (one per nostril). 1–2 doses/hr, max 5 doses/hr
Advantages: Rapid symptom relief; titratable
Disadvantages: Nasal irritation; frequent dosing; contraindicated in chronic nasal disease
Nasal spray is the most expensive (Average daily cost of a cigarette pack ≈ $9.74)
Bupropion SR
Mechanism: atypical antidepressant; inhibits norepinephrine and dopamine reuptake.
Dosing: start 150 mg AM for 3 days → 150 mg BID for 7‑12 weeks (no taper).
C/Is: seizure disorder, bulimia/anorexia, abrupt alcohol/benzodiazepine cessation, MAO‑inhibitor use.
Key ADRs: insomnia, dry mouth, nausea, possible neuropsychiatric symptoms (monitor).
Generic bupropion SR is the cheapest cessation aid
Varenicline
Mechanism: partial α4β2 nicotinic receptor agonist; reduces withdrawal & blocks nicotine reward.
Dosing (Fixed Quit): start 0.5 mg daily Days 1‑3 → 0.5 mg BID Days 4‑7 → 1 mg BID Day 8‑end (12 weeks).
Highest efficacy
Varenicline
Day 1-3 = 0.5 mg qd
Day 4-7 = 0.5 mg bid
Day 8 to end of treatment = 1 mg bid
Up to 12 weeks
Shows stepwise increase from 0.5 mg to 1 mg twice daily.
Common ADRs: nausea, abnormal dreams, insomnia, headache
Combination therapy
Dose: Patch for baseline; gum/lozenge or spray for breakthrough cravings
Advantages: Highest efficacy (see meta‑analysis)
Disadvantages: More complex regimen; cost
Patch + short‑acting NRT
Strong (first‑line)
Moderate‑to‑heavy smokers; need rapid craving control
Varenicline + NRT
Moderate
Prior NRT failure or high dependence
Bupropion + NRT
Moderate
Depressed patients or those intolerant to varenicline
Varenicline + Bupropion
Limited
Considered in refractory cases
Nicotine Replacement Therapy (NRT)
All NRTs share precautions for pregnancy/breastfeeding, adolescents (<18), and recent (within 2 weeks) cardiovascular events (MI, arrhythmia, or worsening angina).
Gum / Lozenge
Based on time to first cigarette:
≤30 min: 4 mg
>30 min: 2 mg
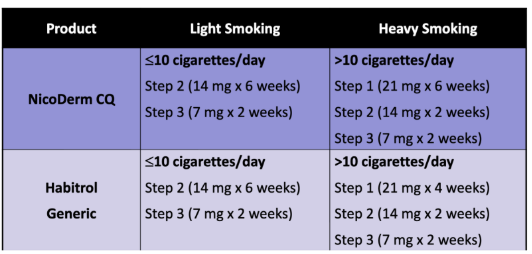
Patch (OTC)
Based on cigarettes per day:
>10 cigs: 21 mg starter
≤10 cigs: 14 mg starter
Rotate sites daily. Remove at bedtime if sleep disturbances/vivid dreams occur.
Nasal Spray (Rx)
1–2 doses/hour (max 40/day)
Avoid in patients with chronic nasal disorders or severe reactive airway disease
Oral Non-Nicotine Medications (Rx)
Bupropion SR (Generic)
Mechanism/Use: Often beneficial for patients with depression or those concerned about weight gain.
Dosing: 150 mg daily for 3 days, then 150 mg BID. Start 1–2 weeks before quit date.
Contraindications (Critical for Exams): * Seizure disorders.
History of Anorexia or Bulimia.
Abrupt withdrawal of alcohol or benzodiazepines.
MAO Inhibitor use within 14 days.
Varenicline (Generic)
Mechanism/Use: Most effective monotherapy; offers a different mechanism for those who failed NRT.
Dosing: Gradual titration (0.5 mg daily → 0.5 mg BID → 1 mg BID). Start 1 week before quit date.
Precautions: Requires dose adjustment for severe renal impairment.
Administration: Take after eating with a full glass of water to minimize nausea.
Important Safety Comparisons
Neuropsychiatric Symptoms
Both Bupropion SR and Varenicline carry warnings for treatment-emergent neuropsychiatric symptoms (e.g., agitation, depressed mood, suicidal ideation). Patients should stop the medication immediately if these occur.
Side Effect Profiles
Mouth/Throat Irritation: Common in gum, lozenge, and spray.
Insomnia: Common in Bupropion SR (avoid bedtime dosing) and Varenicline.
Vivid Dreams: Associated with the Patch and Varenicline.
GI Issues (Nausea): Most common with Varenicline.
Gum + Lozenges
To improve chances of quitting, when used a monotherapy, use at least nine pieces daily during the first 6 weeks
The gum/lozenge will not provide the same rapid satisfaction that smoking provides
The effectiveness of the nicotine gum/lozenge may be reduced by some foods and beverages: Coffee, Juices, Wine, Soft drinks
Do NOT eat or drink for 15 minutes BEFORE or while using the nicotine gum or lozenge.
Chewing the lozenge or using incorrect gum chewing technique can cause excessive and rapid release of nicotine, resulting in:
Lightheadedness/dizziness
Nausea and vomiting
Hiccups
Irritation of throat and mouth
ADEs of nicotine gum and lozenge:
Mouth and throat irritation
Hiccups
Gastrointestinal complaints (dyspepsia, nausea)
ADEs associated with nicotine gum:
Jaw muscle ache
May stick to dental work
ADVANTAGES of Gums + Lozenges
Oral substitute for tobacco
Might delay weight gain
Can be titrated to manage withdrawal symptoms
Can be used in combination with other agents to manage situational urges
Relatively inexpensive
DISADVANTAGES of Gums + Lozenges
Need for frequent dosing can compromise adherence
Gastrointestinal ADEs (nausea, hiccups, and dyspepsia) may be bothersome
Specific to nicotine gum:
Problematic with significant dental work
Proper chewing technique is necessary for effectiveness and to minimize ADEs
Might not be acceptable or desirable for some patients
Nicotine transdermal patch (NRT)
Continuous (24-hour) nicotine delivery system
Nicotine is well absorbed across the skin
Transdermal delivery to systemic circulation avoids hepatic first-pass metabolism
Plasma nicotine levels are lower and fluctuate less than with smoking
1 pack = around 20 cigarettes
PATIENT EDUCATION for Nicotine Patches
Water will not harm the nicotine patch if it is applied correctly; patients may bathe, swim, shower, or exercise while wearing the patch
Do not cut patches to adjust dose
Can unpredictably effect nicotine delivery
Patch may be less effective
Keep new and used patches out of the reach of children and pets
Remove patch before MRI procedures
Common ADEs include:
Irritation at patch application site (generally within the first hour)
Mild itching
Burning
Tingling
Sleep disturbances
Abnormal or vivid dreams
Insomnia
PATIENT EDUCATION for Nicotine Patches
After patch removal, skin may appear red for 24 hours
If skin stays red more than 4 days or if it swells or a rash appears, contact health care provider—do not apply new patch
Local skin reactions (redness, burning, itching)
Usually caused by adhesive
Up to 50% of patients experience this reaction
Fewer than 5% of patients discontinue therapy
Confirm patient is rotating patch application sites
Avoid use in patients with dermatologic conditions (e.g., psoriasis, eczema, atopic dermatitis)
ADVANTAGES for Nicotine Patches
Once-daily dosing associated with fewer adherence problems
Of all NRT products, its use is least obvious to others
Can be used in combination with other agents; delivers consistent nicotine levels over 24 hrs
Relatively inexpensive
DISADVANTAGES for Nicotine Patches
When used as monotherapy, cannot be titrated to acutely manage withdrawal symptoms
Not recommended for use by patients with dermatologic conditions (e.g., psoriasis, eczema, atopic dermatitis)
Nicotine nasal spray (Rx/NRT)
Aqueous solution of nicotine in a 10-ml spray bottle
Each metered dose actuation delivers 50 mcL spray
0.5 mg nicotine
~100 doses/bottle
Rapid absorption across nasal mucosa
DOSING for Nicotine Nasal Spray
One dose = 1 mg nicotine (2 sprays, one 0.5 mg spray in each nostril)
Start with 1–2 doses per hour
Increase as needed to maximum dosage of 5 doses per hour or 40 mg (80 sprays; ~½ bottle) daily
At least 8 doses daily for the first 6–8 weeks
Termination:
Gradual tapering over an additional 4–6 weeks
Recommended maximum duration of therapy is 3 months
PATIENT EDUCATION for Nicotine Nasal Spray
What to expect (first week):
Hot peppery feeling in back of throat or nose
Sneezing
Coughing
Watery eyes
Runny nose
ADEs should lessen over a few days
Regular use during the first week will help in development of tolerance to the irritant effects of the spray
If ADEs persist after a week, contact health care provider and consider alternative treatment
ADVANTAGES for Nicotine Nasal Spray
Can be titrated to rapidly manage withdrawal symptoms
Can be used in combination with other agents to manage situational urges
DISADVANTAGES for Nicotine Nasal Spray
Need for frequent dosing can compromise adherence
Nasal administration might not be acceptable/desirable for some patients; nasal irritation often problematic
Not recommended for use by patients with chronic nasal disorders or severe reactive airway disease
Cost of treatment
Bupropion SR (Rx)
Non-nicotine cessation aid
Mechanism of action: atypical antidepressant thought to affect levels of various brain neurotransmitters
Dopamine
Norepinephrine
Clinical effects
↓ craving for cigarettes
↓ symptoms of nicotine withdrawal
C/I for Bupropion
Seizure disorder
Current or prior diagnosis of bulimia or anorexia nervosa
Abrupt discontinuation of alcohol, benzodiazepines, barbiturates and antiepileptic drugs
Use of MAO inhibitors (within 14 days of initiating or discontinuing therapy)
WARNINGS and PRECAUTIONS for Bupropion
Use with caution in the following populations:
Patients with an elevated risk for seizures, including:
Severe head injury
Concomitant use of medications that lower the seizure threshold (e.g., other bupropion products, antipsychotics, tricyclic antidepressants, theophylline)
Severe hepatic impairment
Patients with underlying neuropsychiatric conditions
Neuropsychiatric symptoms and suicide risk
Changes in mood (including depression and mania)
Psychosis/hallucinations/paranoia/delusions
Homicidal ideation
Aggression/hostility/anxiety/panic
Suicidal ideation, suicide attempt, completed suicide
FDA boxed warning removed
Advise patients to stop taking bupropion SR and contact a health care provider immediately if symptoms such as agitation, depressed mood, or changes in behavior or thinking that are not typical are observed or if the patient develops suicidal ideation or suicidal behavior.
DOSING for Bupropion
To ensure therapeutic plasma levels of the drug are achieved, begin therapy 1 to 2 weeks PRIOR to the quit date.
Initial treatment
150 mg orally in the morning for 3 days
Then…
150 mg orally twice daily for 7–12 weeks
Doses must be administered at least 8 hours apart
Tapering not necessary when discontinuing therapy
ADEs for Bupropion
Common ADEs include the following:
Insomnia (avoid bedtime dosing)
Dry mouth
Nausea
Less common but reported effects:
Anxiety/difficulty concentrating
Constipation
Tremor
Skin rash
ADVANTAGES for Bupropion
Twice daily oral dosing is associated with better adherence
Might delay weight gain
Might be beneficial in patients with depression
Can be used in combination with
NRT agents
Relatively inexpensive
DISADVANTAGES for Bupropion
Seizure risk is increased
Several contraindications and precautions preclude use in some patients
Patients should be monitored for neuropsychiatric symptoms
Varenicline (Rx)
Non-nicotine cessation aid
Mechanism of action: partial nicotinic receptor agonist that binds with high affinity and selectivity at α4β2 neuronal nicotinic acetylcholine receptors
Stimulates low-level agonist activity
Competitively inhibits the binding of nicotine
Clinical effects
↓ symptoms of nicotine withdrawal
Blocks dopaminergic stimulation responsible for reinforcement & reward associated with smoking
WARNINGS and PRECAUTIONS for Varenicline
Neuropsychiatric symptoms and suicide risk
Changes in mood (including depression and mania)
Psychosis/hallucinations/paranoia/delusions
Homicidal ideation
Aggression/hostility/anxiety/panic
Suicidal ideation, suicide attempt, completed suicide
FDA boxed warning removed Dec 2016
Advise patients to stop taking varenicline and contact a health care provider immediately if symptoms such as agitation, depressed mood, or changes in behavior or thinking that are not typical are observed or if the patient develops suicidal ideation or suicidal behavior.
STANDARD DOSING for Varenicline
FIXED QUIT
Set quit date for 1 week after starting varenicline
Continue treatment for 12 weeks
FLEXIBLE QUIT
Start taking varenicline and pick a quit date between 8 to 35 days from treatment initiation
Continue treatment for 12 weeks
GRADUAL QUIT
Start taking varenicline and reduce smoking by 50% within the first 4 weeks, an additional 50% in the next 4 weeks, and continue until complete abstinence by 12 weeks
ADEs for Varenicline
Common ADEs include the following:
Nausea
Abnormal dreams
Insomnia
Headache
Less common ADEs:
Gastrointestinal (flatulence, constipation)
Taste alteration
PATIENT EDUCATION for Varenicline
Doses should be taken after eating, with a full glass of water
Nausea and insomnia are usually temporary side effects
If symptoms persist, notify your health care provider
May experience vivid, unusual or strange dreams during treatment
Use caution driving, drinking alcohol, and operating machinery until effects of quitting smoking with varenicline are known
ADVANTAGES for Varenicline
Twice daily oral dosing is associated with better adherence
Offers an alternative mechanism of action for individuals who have not tolerated or have not responded to other agents
Most effective agent for smoking cessation
DISADVANTAGES for Varenicline
Cost of treatment
Patients should be monitored for potential neuropsychiatric symptoms
Combination therapy
Combination NRT [first-line, recommended treatment approach]
Long-acting formulation (patch)
Produces relatively constant levels of nicotine
AND
Short-acting formulation (gum, lozenge, nasal spray)
Allows for acute dose titration as needed for nicotine withdrawal symptoms
Other combinations [evidence less compelling]
Bupropion + NRT
Varenicline + NRT
Varenicline + bupropion SR
Strong evidence that combination NRT and varenicline are more effective than bupropion SR or NRT monotherapy

KNOW IMAGE
Combination NRT [first-line, recommended treatment approach]
Long-acting formulation (patch)
Produces relatively constant levels of nicotine
AND
Short-acting formulation (gum, lozenge, nasal spray)
Allows for acute dose titration as needed for nicotine withdrawal symptoms
EAGLES STUDY (Varenicline, Bupropion SR, Nicotine patch, Placebo)
No significant differences in neuropsychiatric events by treatment arm
Highest efficacy with varenicline
ASK
Ask about tobacco use; with a tone that conveys sensitivity, concern and is non-judgmental
Consider tobacco use assessment to be as important as other vital signs
“Do you smoke or use other types of tobacco or nicotine, such as e-cigarettes?”
“We ask all of our patients, because tobacco smoke can affect how some medicines work.”
We care about your health, and we have resources to help our patients quit smoking.”
“Has there been any change in your smoking/tobacco use/vaping status?”
ADVISE
Advise tobacco users to quit (clear, strong, personalized)
“Quitting smoking is the most important thing you can do for your health.”
“Quitting is the most important thing you can do to...[control your asthma, reduce your chance for another heart attack, better manage your diabetes, etc.]”
“The best way to quit is to combine a tobacco cessation medication with a support program—we are trained to help our patients quit.”
“I can help you select medications to increase your chances of quitting for good.”
ASSESS
Assess readiness to make a quit attempt
ASSIST
Assist with the quit attempt
Not ready to quit: enhance motivation
Ready to quit: design a treatment plan
Recently quit: relapse prevention
ARRANGE
Arrange follow-up care
Provide assistance throughout the quit attempt.
Ask → about tobacco USE
Advise → tobacco users to QUIT
Assess → READINESS to make quit attempt
Assist → with the QUIT ATTEMPT
Arrange → FOLLOW-UP care
STAGE 1: Not ready to quit in the next month
Some patients are aware of the need to quit.
Patients struggle with ambivalence about change.
Patients are not ready to change, yet.
Pros of continued tobacco use outweigh the cons.
Patients have inadequate motivation to quit.
GOAL: Start thinking about quitting
Counseling DO’S
Strongly advise to quit
Guide patient to find a strong personal motivation to quit by:
Providing information
Asking noninvasive questions; identify reasons for tobacco use
Raising awareness of health consequences/concerns
Demonstrate empathy, foster communication
Counseling DON’TS
Persuade
Leave decision up to patient
“Cheerlead”
Tell patient how bad tobacco is, in a judgmental manner
Provide a treatment plan
The 5 R’s—Methods for enhancing motivation:
Relevance
Risks
Rewards
Roadblocks
Repetition
STAGE 2: Ready to quit in the next month
Patients are aware of the need to, and the benefits of, making the behavioral change.
Patients are getting ready to take action
GOAL: Achieve cessation.
Assess tobacco use history
Current use: type(s) of tobacco, amount
Past use: duration, recent changes
Past quit attempts:
Number, date, length
Methods/medications used, adherence, duration
Reasons for relapse
STAGE 2: Ready to quit in the next month
When fear of weight gain is a barrier to quitting
Consider pharmacotherapy with evidence of delaying weight gain (bupropion SR or 4-mg nicotine gum or lozenge)
Assist patient with weight maintenance or refer patient to specialist or program
W/D Sxs
Most pass within 2–4 weeks after quitting
Cravings can last longer, up to several months or years
Often can be ameliorated with cognitive or behavioral coping strategies
Refer to Withdrawal Symptoms Information Sheet
Symptom, cause, duration, relief
Most symptoms manifest within the first 1–2 days, peak within the first week, and subside within 2–4 weeks.
Tobacco Use Log: Instructions for use
Continue regular tobacco use for 3 or more days
Each time any form of tobacco is used, log the following information:
Time of day
Activity or situation during use
“Importance” rating (scale of 1–3)
Review log to identify situational triggers for tobacco use; develop patient-specific coping strategies
Cognitive coping strategies
Thinking about cigarettes doesn’t mean you have to smoke one:
“Just because you think about something doesn’t mean you have to do it!”
Tell yourself, “It’s just a thought,” or “I am in control.”
◼ As soon as you get up in the morning, look in the mirror and say to yourself:
“I am proud that I made it through another day without tobacco.”
Reframe how you think about yourself:
Begin thinking of yourself as a non-smoker, instead of as a struggling quitter
STEP 1
Ask:
“What could you do differently in this situation so you won’t be prompted to want a cigarette?”
“How could you think differently in this situation, so that you aren’t triggered to want to smoke?”
STEP 2
If they provide a reasonable alternative, be supportive
If they say “I don’t know” or “I can’t think of anything”
Suggest a coping technique (or two)
Make suggestions appropriate to their lifestyle
Offer to assist throughout quit attempt
Follow-up contact #1: first week after quitting
Follow-up contact #2: in the first month
Additional follow-up contacts as needed
STAGE 3: Recent quitter, quit within past 6 months, Actively trying to do good
Patients have quit using tobacco sometime in the past 6 months and are taking steps to increase their success.
Withdrawal symptoms occur.
Patients are at risk for relapse
GOAL: Remain tobacco-free for at least 6 months.
STAGE 3: Recent quitter, quit within past 6 months, Actively trying to do good
Evaluate Quit Attempt
Tailor interventions to match each patient’s needs
Status of attempt
Ask about social support
Identify ongoing temptations and triggers for relapse (negative affect, others who smoke, eating, alcohol, cravings, stress)
Encourage healthy behaviors to replace tobacco use
Slips and relapse
Has the patient used tobacco/inhaled nicotine at all—even a puff?
Medication adherence, plans for termination
Is the regimen being followed?
Are withdrawal symptoms being alleviated?
How and when should pharmacotherapy be terminated?
STAGE 3: Recent quitter, quit within past 6 months, Actively trying to do good
Relapse Prevention
Congratulations on your success!
Encourage continued abstinence
Discuss benefits of quitting, problems encountered, successes achieved, and potential barriers to continued abstinence
Ask about strong or prolonged withdrawal symptoms (change dose, combine or extend use of medications)
Promote smoke-free environments
Schedule additional follow-up as needed
STAGE 4: Former tobacco user, quit > 6 months ago
Patients differ in their readiness to quit.
Assessing a patient’s readiness to quit enables clinicians to deliver relevant, appropriate counseling messages.
For most patients, quitting is a cyclical process, and their readiness to quit (or stay quit) will change over time.
Patients remain vulnerable to relapse.
Ongoing relapse prevention is needed.
GOAL: Remain tobacco-free for life.
Former Tobacco Users
Assess status of quit attempt
Congratulate continued success
Inquire about and address slips and relapse
Plans for termination of pharmacotherapy
Review tips for relapse prevention
Continue to assist throughout the quit attempt.
WHAT ARE “TOBACCO QUITLINES”?
Tobacco cessation counseling, provided at no cost via telephone to all Americans
Staffed by highly trained specialists
Up to 4–6 personalized sessions (varies by state)
Some state quitlines offer pharmacotherapy at no cost (or reduced cost)
Nearly 30% success rate for patients who use the quitline and a medication for cessation (versus 13% for quitline use alone)
Quitlines have broad reach and are recommended as an effective strategy in the Clinical Practice Guideline.
WHEN a PATIENT CALLS the QUITLINE
Contact and demographic information
Smoking behavior
Choice of services
Individualized telephone counseling
Quitting literature mailed within 24 hrs
Referral to local programs, as appropriate
The Tobacco Quitline is a formal cessation program, with multiple sessions. It is NOT a crisis hotline.