chem unit 4
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
What is green fuel
Fuel produces from renewable sources that have lower environmental impact (i.e. lower greenhouse gas emission and/or no harmful residue left after combustion)as compared to traditional fossil fuels
Give examples of green fuel
Hydrogen fuel, Biofuel, wind power, solar power, geothermal energy, synthetic fuel(ex synthetic methane produced from CO2 and H2)
Write environmental benefit of green fuel
no emission of greenhouse gases
no emission of pollutants
derived from renewable sources
limit global warming
write economic benefits of green fuel
new jobs created in field of production ,R&D and manufacturing of renewable energy(estimated 11 millions jobs in 2018, rising year by year)
development of green fuels demands technological innovation that benefits other fields, which improves overall economy and technological capabilities of a country
diversification of energy portfolio, eliminating dependency on one type of fuel
cleaner transportation options, reducing traffic congestion and improving air quality
what are types of hydrogen based on how it is produced
grey, brown, green, blue
define grey hydrogen and by-product during it’s manufacture
derived from hydrocarbon
CO2 is by-product
emission of CO2 makes it a significant greenhouse gas emitter
define brown hydrogen
also called black hydrogen
derived from coal
process cause high pollution
CO2 and CO produced as by-product
define blue hydrogen
derived from hydrocarbons(natural gas)
emissions generated can be captured and stored underground by industrial carbon capture and storage (CCS)
cleaner then grey hydrogen, but still relies on natural gas for it’s production
define green hydrogen
generated from renewable sources (wind and solar)
electricity splits water into H2 and O2
cleanest way to make the cleanest fuel
only byproducts is water, it is a zero carbon emission process
write two methods to split water
photo-catalytic water splitting and photo-electro-chemical water splitting
Write three fundamental steps of photo-catalytic water splitting
Photocatalyst absorbs photons having energy more than the bandgap, creating electron-hole pairs in the( e- in the conduction band and hole in the valence band)
these electron hole pairs migrate from the bulk to the active sites on the surface where redox reaction can occur
redox reaction occurs producing H2 and O2
Write reactions involved in photo-catalytic water splitting and give example of catalyst used

write the steps of photo-electro-chemical water splitting
photon of sufficient energy strikes the surface of semiconductor photo-electrode(anode), generating electron hole pair
electron migrates to cathode by means of external circuit(external circuit is biased)(anode connected to positive terminal and cathode connected to negative terminal)
oxidation of water molecule by hole at anode producing O2 and H+
H+ migrate to cathode thru electrolyte where it is reduced by electron to produce H2 gas.
Note- anode is made of semiconductor material, whereas cathode is made of inert material such as Pt
write reactions involved in photo-electro-chemical water splitting and type of semi conductor used

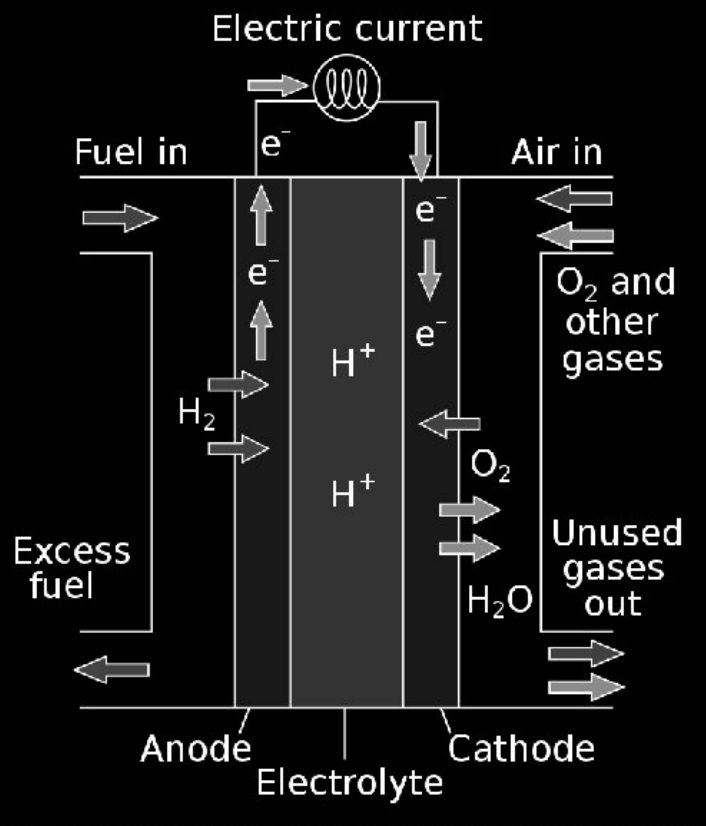
Draw diagram of hydrogen fuel cell and explain it’s working
hydrogen is fed to anode, and air is fed to cathode
at anode H2 gas breaks down to H+ and e-
H+ travels to cathode via electrolyte and e- travels to cathode via external circuit
at cathode, H+ and e- combine with oxygen to produce water and heat
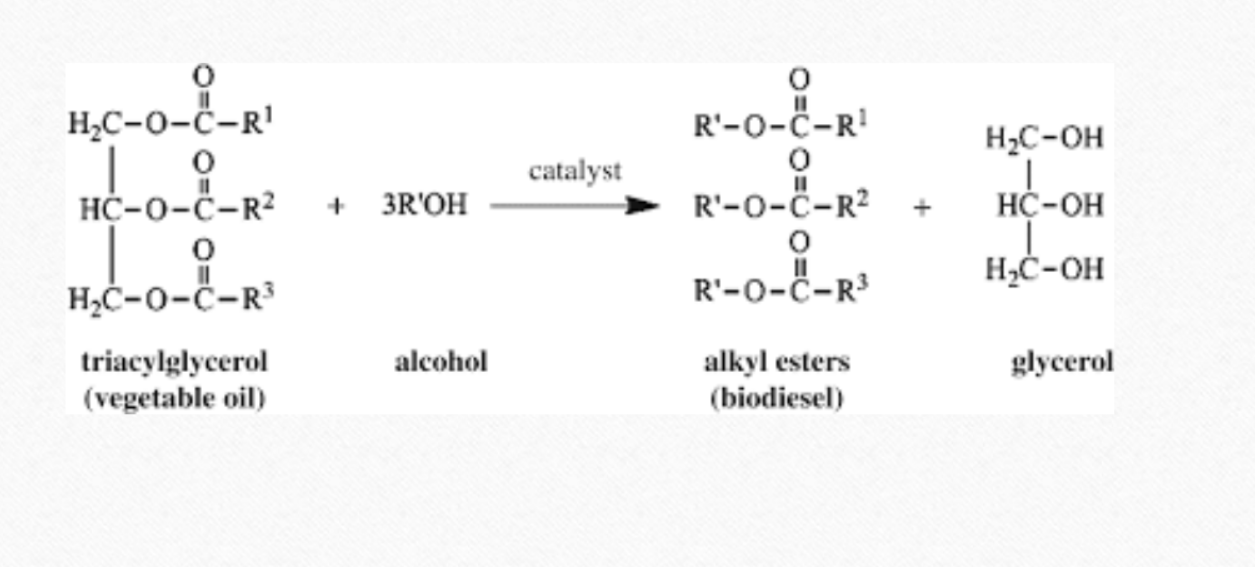
Write production of biodiesel from Jatropha
Name of reaction is transesterification
Mix an alcohol(usually methanol) with NaOH( or KOH), creating sodium methoxide(catalyst)
add this to jatropha oil(contains triglyceride), glycerin and biodiesel are obtained
glycerin settles to the bottom, biodiesel(methyl ester) floats on top
biodiesel is washed and filtered
what is biopolymer made from and what are it’s applications
derived from biobased materials
biodegradable
usage →biopolymers that degrade to harmless products in the body can be used in medical devices and drug delivery
greener than traditional plastics
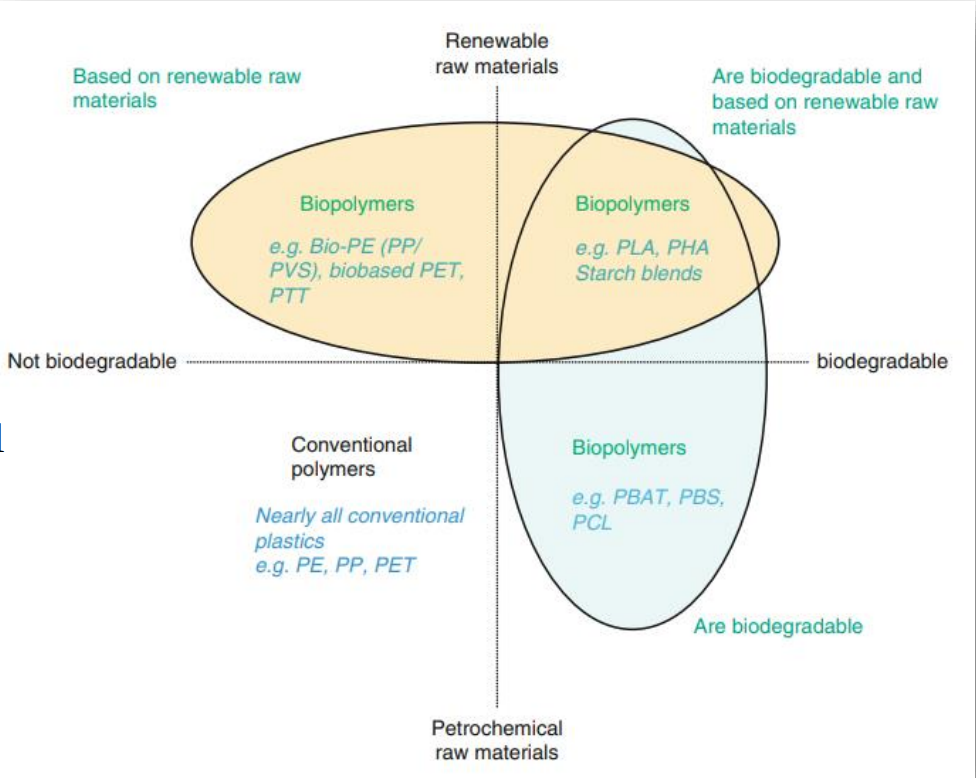
give examples of non/biodegradable x renewable/petrochemical materials
Name two biodegradable polymers(in syllabus)
Polylactic acid(PLA) and polyvinyl alcohol(PVA)
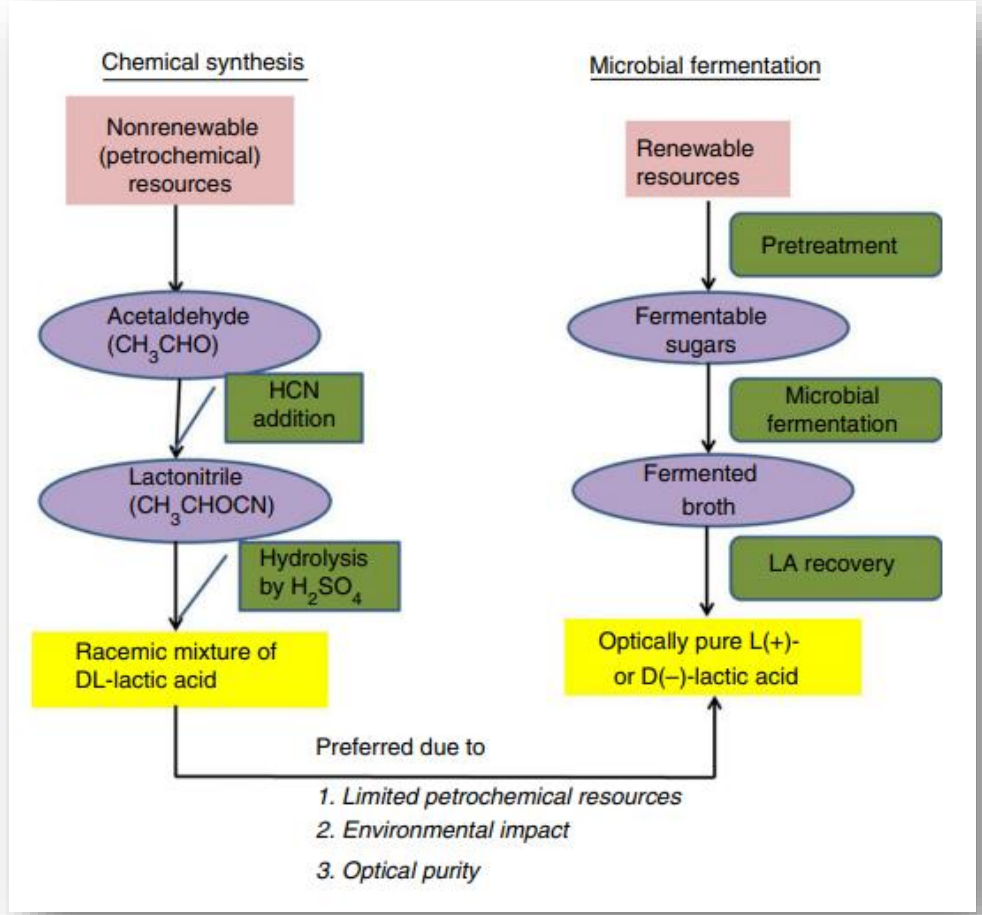
write the two methods of synthesis of lactic acid
write three methods for synthesis of PLA from Lactic acid
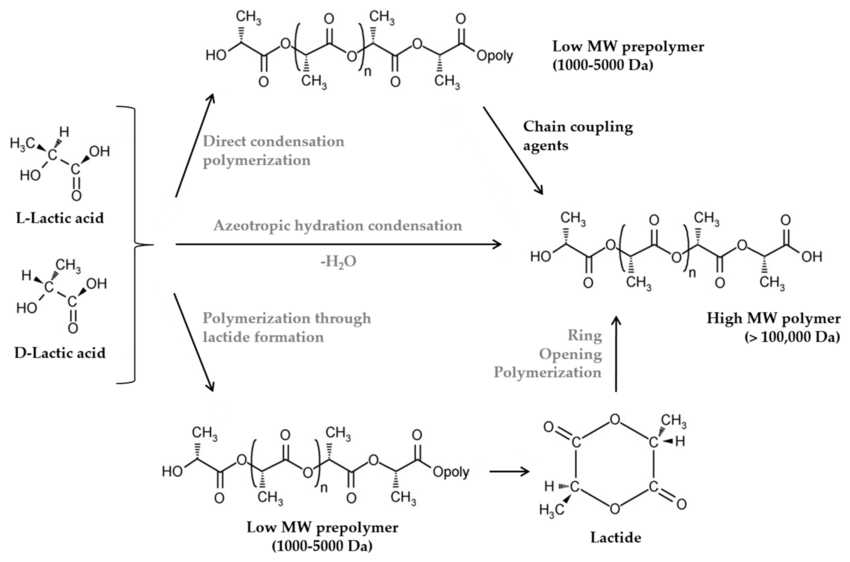
write properties of PLA
excellent shaping and molding properties, sheet extrusion, film blowing and fiber spinning
bio-compatible
bio-reabsorbable
transparency, high elastic modulus and high melting point
write applications of PLA
fixation of fractured bones in the forms of plates/pins/screws
drug delivery- microspheres, microcapsules, RNA/DNA delivery
3D electrospun fibrous scaffolds for bone regeneration
sutures in dermatology
what is polyvinyl alcohol produced from
polyvinyl acetate
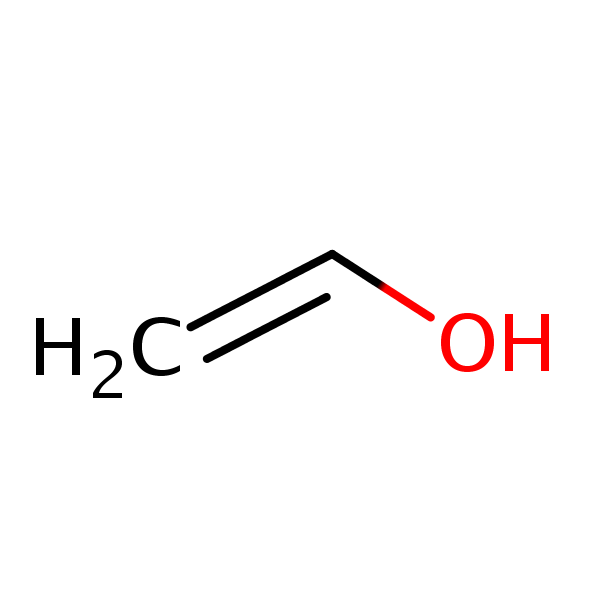
write structure of vinyl alcohol and why PVA is not produced by polymerizing vinyl alcohol
vinyl alcohol is thermodynamically unstable and tautomarized to aldehyde
write reaction for synthesis of polyvinyl alcohol
We polymerize vinyl acetate to get polyvinyl acetate
we convert PVAc into PVA through base-catalyzed transesterification
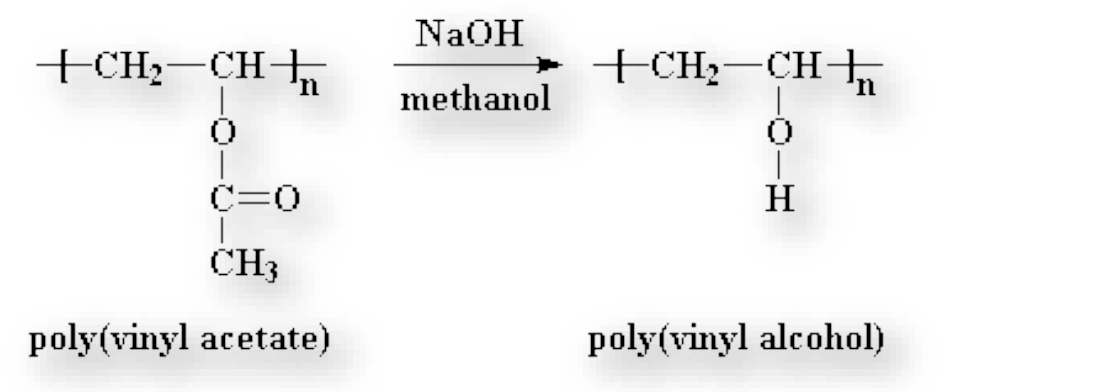
Properties of PVA
high flexibility and tensile strength, ductile
miscible in water
immiscible in organic solvents (except for slightly soluble in ethanol
Applications of PVA
Manufacturing of adhesives
food packaging materials
controlled release drug tablets/capsules
used as surface sizing agent to improve paper’s surface strength and printability