Past papers
1/28
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
29 Terms
Can bromine displace bromine
No
What color is silver iodide precipitate?
Yellow

Slower rate of reaction
C-Br bond stronger than C-I bond
Line for bromoutane below iodophor butane
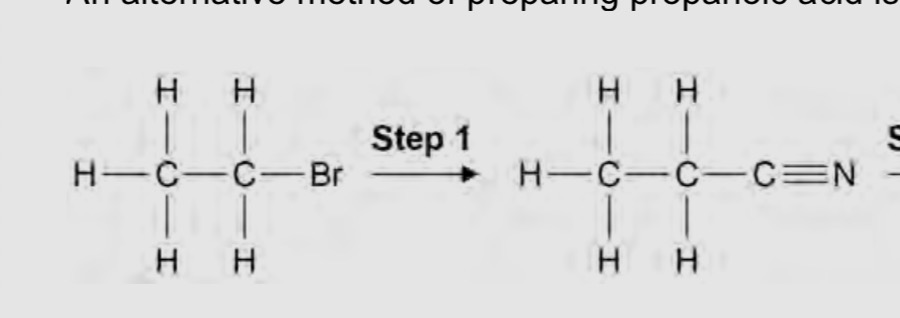
Why would you not use water for step 1?
It would form an alcohol
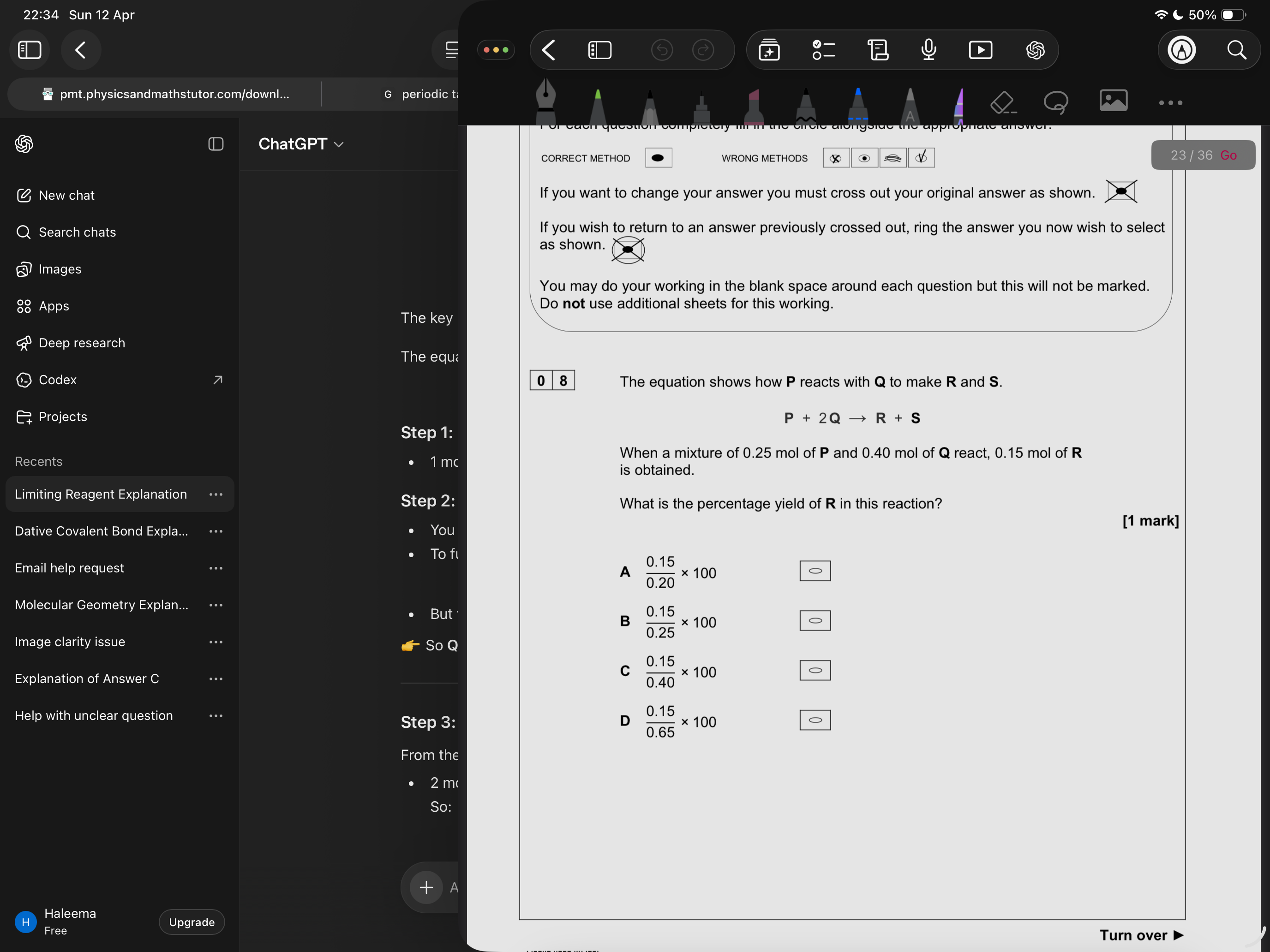
A
Use limiting reagent

Are these values for formation or normal or bond enthalpy
And why
Formation
1 mole of substance is formed
Not combustion as not burned in oxygen

How do you find the number of molecules here
3.0111×10²4
Divided by 6 as 6 carbon atoms in cyclohexane
Divide by avogadros
Multiply by mr
Moles formula
Number of particles divided by avogadros
Explain why the C-Cl bond is polar
Chlorine is more electronegative than carbon
So chlorine becomes slightly negative and carbon becomes slightly positive
There are van der waa;s forces between non polar molecules
Explain what causes these forces
Random movement of electrons in one molecule
Induces a dipole in another
Dipoles in different molecules attract
State how the detector enable the relative abundance of each ion to be determined
Abundance is proportional to size of current
Trigonal pyramidal bond angle
107
Test for ammonium nitrate
Add NaOH
Hold damp red litmus paper at mouth of tube
turns blu
What is the observation for magnesium nitrate with sodium hydroxide
White precipitate
Standard enthalpy of formation definition
Enthalpy change when 1 mole of a substance is formed from its elements a
All substances in their standard states


Formula for percentage atom economy
Mr of Desired product
Divided by
Mr of total reactant
AlLWAYS REACTANT

D
B is ionic bond in and A and C have a full outer shell now
Is hydrogen a non metal
Yes

C
Describe trend in first ionisation energy inperiod 3
Increases
Atomic radius in period 3 describe the trend
Decreases
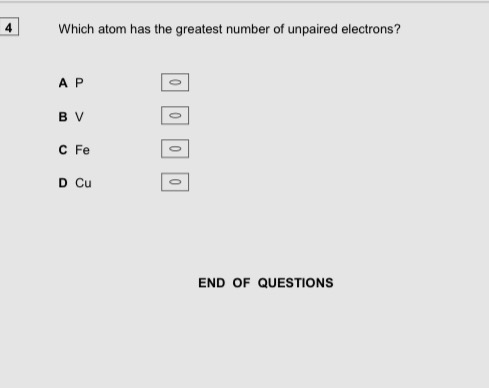
C

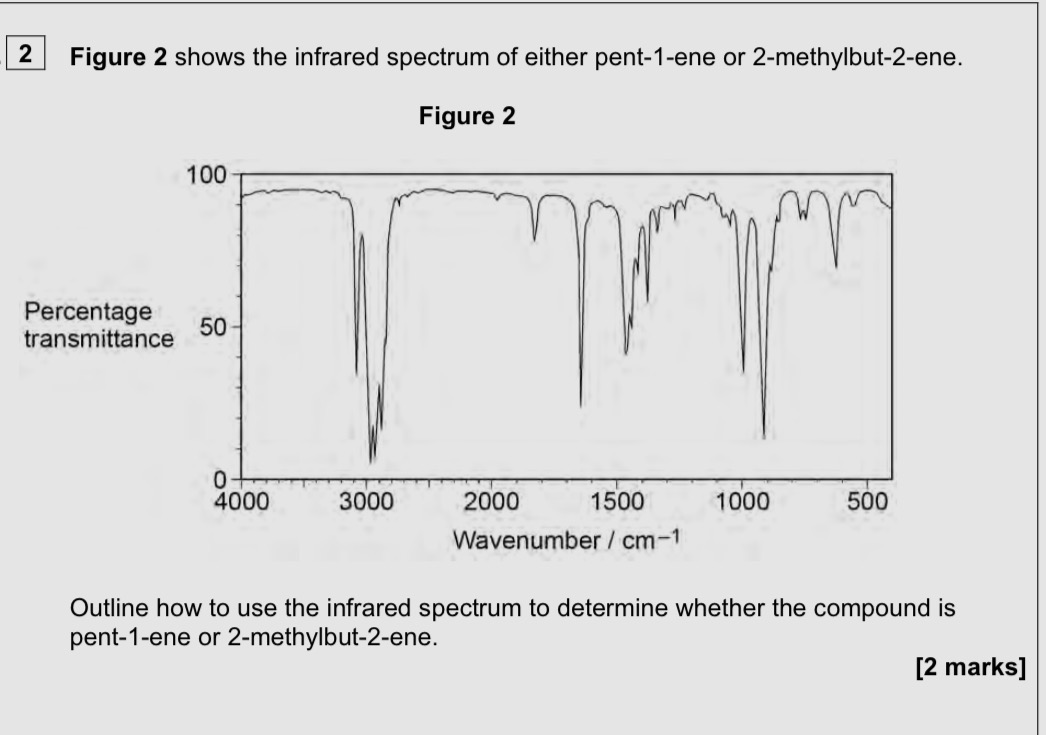
Look for finger print region and look for match to known spectrum
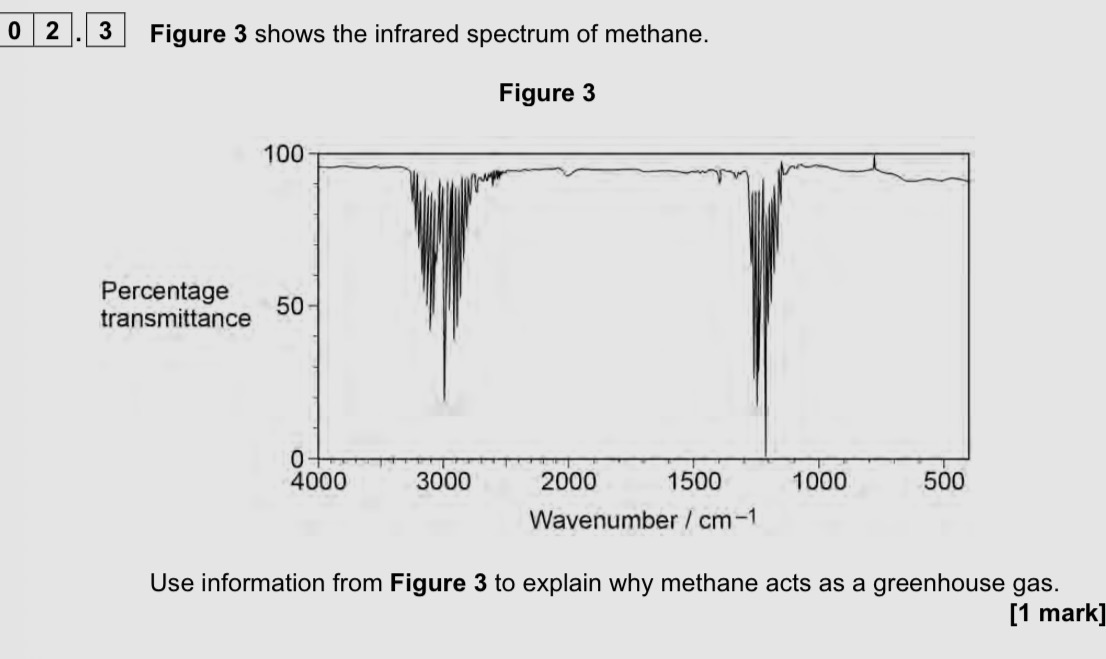
Absorbs infrared radiation
Absorbs around 3000 and 1200 cm-1

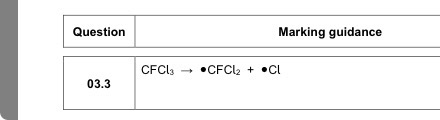

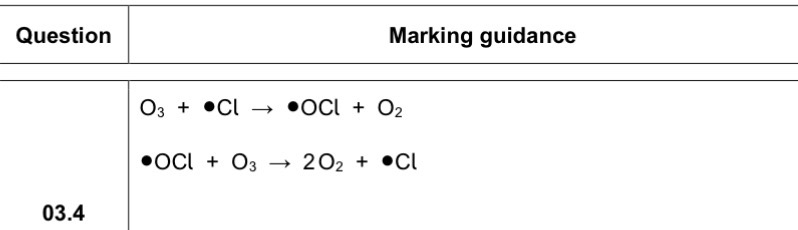

ABSORBs UV radiation
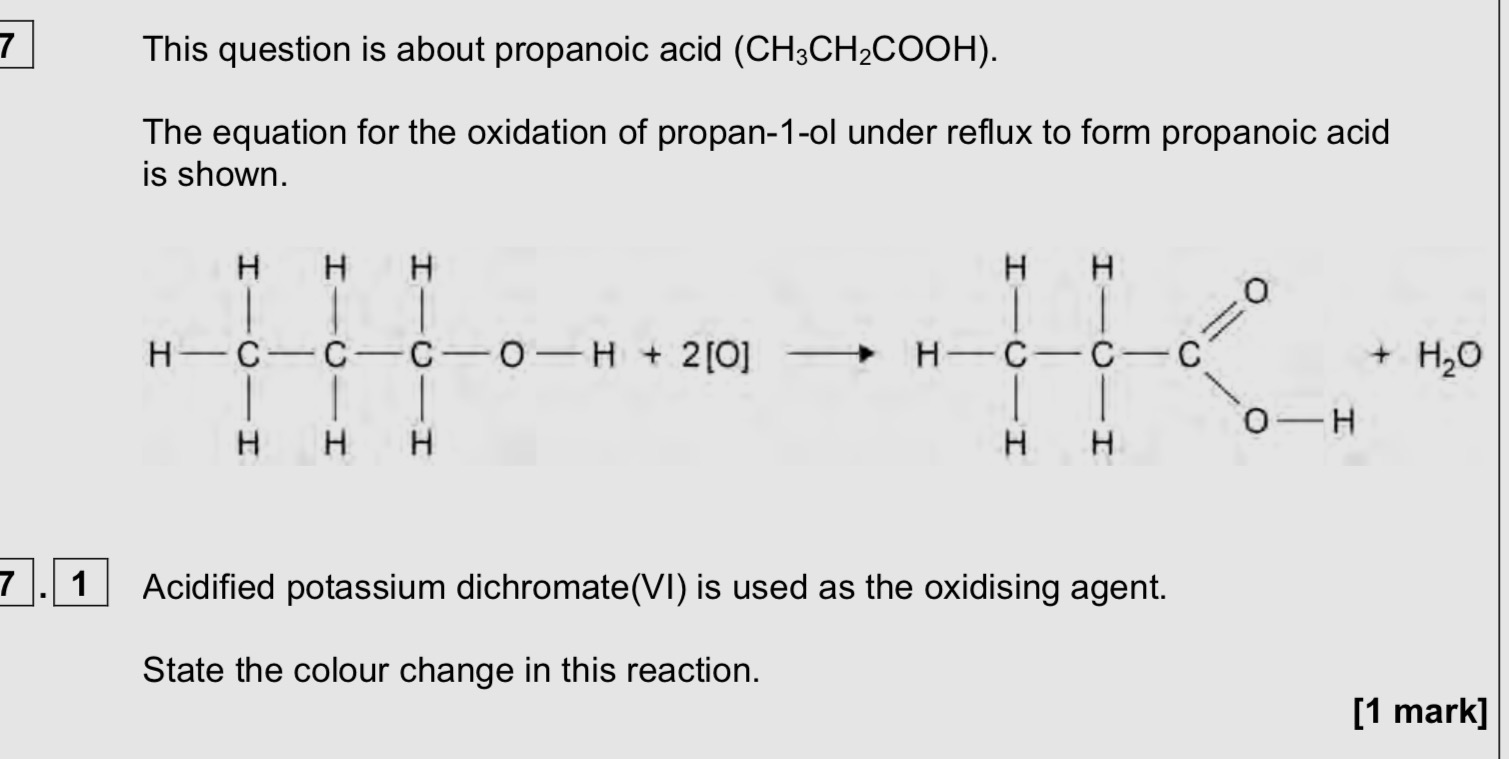
Orange to green