PHYSL 410 First topics
1/28
Earn XP
Description and Tags
Endometriosis, Eating disorders, Substance misuse disorder, Epilepsy, IBD and Cushings syndrome
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
29 Terms
Endometriosis characteristics
Chronic inflammatory state, where endometrial like tissues (cells, glands or stroma = connective tissues) develop outside of the uterus = ectopic. A hyperestrogenic state. Ectopic cells continue to menstruate/respond to hormone levels.
Typical presentation: Pain (dysmenorrhea, dyspareunia, dysuria, dyschezia, lower back or abdominal discomfort, non-cyclic chronic pelvic pain) is the hallmark symptom, along with fatigue and infertility.
Atypical presentation: Cyclic leg pain following menstrual cycle (sciatica involvement), rectal bleeding or hematuria (invasion of endometrial tissue in bowel or bladder), cyclic dypsnea secondary to catemenail pneumothorax (invasion of endometrial tissue into the lungs).
Endometriosis risk group/population
10% of women worldwide and over 77% of infertile women. Most common in those ages 18-49, especially in ages 18-29.
Poor historical management of women’s health
Thought that the normal state of a women was to be sick. Psychological perspective further hampered this, assuming that hysteria, pain and discomfort in relation to menstruation was all just psychological instead of physiological.
Why is endometriosis hard to diagnosis
Partially due to women hiding or delaying seeking treatment for pain due to not wanting to be embarrased or just thinking it is normal (largely due to mistreatment of women in the past) = 2-3 year delay.
Contains many general symptoms, disease is poorly defined, all women’s bodies are different and so there is not one model/guideline for what endometriosis looks like from one person to the next
Relevant reproductive questions relating to endometriosis
Age at menarche, cycle frequency and regularity, past births, use of oral contraceptives/hormones, med/sx history, fam history
Factors that increase the liklihood of endometriosis
Nuliparity, subfertility, regular cycles and first degree relatives with endometrosis.
Factors that decrease the liklihood of endometriosis
increased parity and irregular cycles.
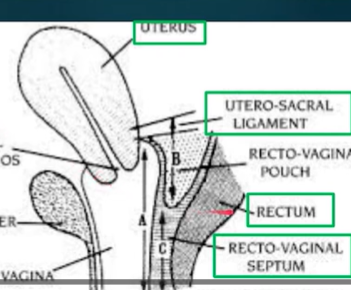
Physical examination for endometriosis
Beginning with position and orientation of the uterus (ensuring it is anteverted and not retroverted = associated with severe disease states). Then rectovaginal examination, palpating uterosacral ligaments and recto-vaginal septum; tender nodes here indicate deeply infiltrating endometriosis. Adnexal masses may also be detected (growths along reproductive tract), such as endometriomas = specific adnexal masses growing on ovaries. Can be done at any time, but liklihood of detection is increased during the menstruation phase.
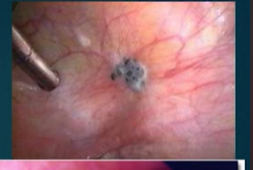
Gold standard for confirming endometriosis
Laparoscopic surgery excising lesions, which is then followed by histological verification. Not necessary for treatment, can be risky.
Medical management of endometriosis
Chronic pain management (as no cure), with the goal of suppressing estrogen/ovulation and create a hypoestrogenic state via
Oral contraceptives (continuous administration of estrogen and progestin) or
Progestin only through oral or depot (intramuscular injection) methods or
Intrauterine (IUD) device releasing progestin locally or
Gonadotropin suppression (weak androgen administration, can produce unfavourable side effects = weight gain, acne, breast atrophy) or
GnRH agonists (potential irreversible side effects with bone mineral density).
All treatments take at least one cycle to start impacts, so opiods/NSAIDs may be required to supplement.
Surgical management of endometriosis
Secondary to medical therapy, if it is ineffective or contraindicated. This can help to relieve pain in ~50% of patients. Further complications as a result of endometriosis (ruptured ovarian cyst, torsion of the fallopian tube) or severe invasive disease may necessitate surgery. Two methods:
Conservative: Restores normal autonomy for those that may want to reproduce in the future or do not want to begin menopause. Laparoscopic approach, in order to lyse/excised/ablated characterstic gunshot/powder burn lesions.
Definitive: Bilateral oophorectomy (Just ovary removal, to induce early menopause) or the removal of the ovaries, uterus and the fallopian tubes = hysterctomy
Despite surgeries, lesions may return and/or px symptoms may not improve.
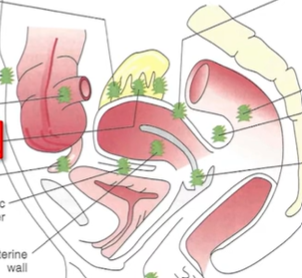
Sampson’s theory - Retrograde menstruation theory (1920’s)
Eutopic endometrium during menstruation travels retrograde up the fallopian tubes and into the pelvic cavity/peritoneal fluid, rather than out through the vagina. This is supported by lesion dispersion. Limited because it does not tell us how implantation occurs, or why all women don’t get endometriosis (as retrograde menstruation is universal)
Where are endometriosis lesions most common
In the cul de sac (in between uterus and colon posteriorly, due to gravity) and the left side of the pelvis, due to sigmoidal colon blocking the ectopic cells from travelling any further.
Metaplastic theory
Ectopic endometrial tissue is non-uterine. Instead, coelomic metaplasia tissue (peritoneal cells) somehow differentiate into ectopic endometrial cells (trigger unknown). Plausible as both originate from coelomic epithelium. Supports rare cases of endometriosis in males.
Induction theory
Hybrid of metaplastic and Samson’s theory. Begins with retrograde menstruation introducing endometrial cells into abdomen and then releasing a factor which allows for implantation and differentiation of other tissues to become endometrial cells/full fledged legions
Endometriosis cellular and molecular abnormalities
High in estrogen, cytokines, prostaglandins (rabbit research) and metalloproteinases. Normal endometrial stroma does not exhibit these characteristics, and could explain how cellular implantation and resistance to the immune system occurs in endometriosis.
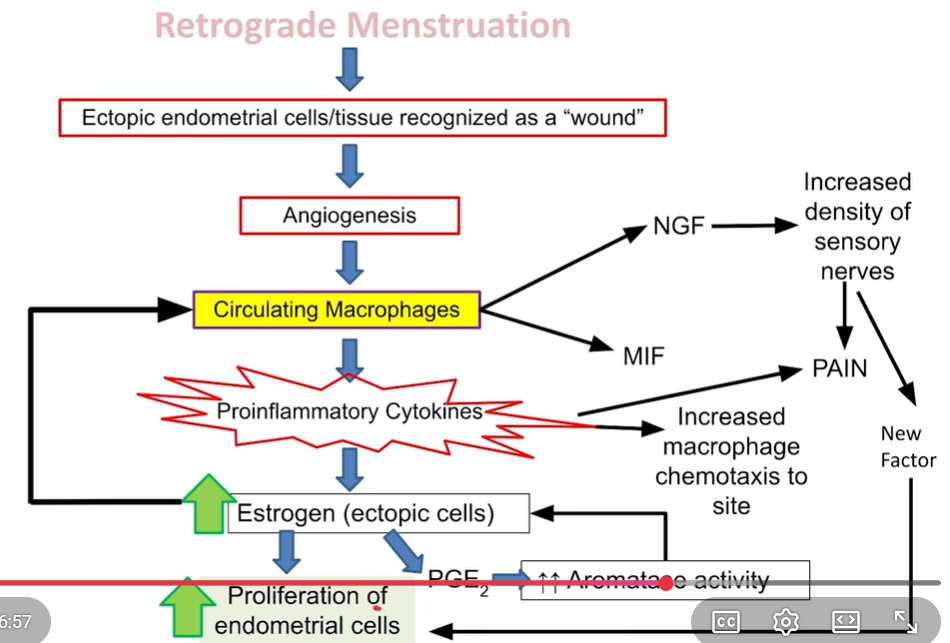
Estrogen driven inflammation
Critical feature of endometriosis, causes the pain and chronic nature associated with this condition. Enhances the survival of ectopic endometrial lesions, and is released from the follicles, at ovulation and via adipose tissue (aromatases = testosterone to estrogen). Furthermore, endometrial legions themselves create estrogen. Estrogen stimulates prostaglandins which create more estrogen (positive feedback); estrogen stimulates cytokines: IntraLeukin 17-A for angiogenesis (implantation and sustaining ectopic tissue) and IL1-B, IL-6 and TNFa (causes inflammation)
Endometriotic lesions impact on progesterone
Exhibit progesterone inhibitors, which impairs the aMnti-estrogen impact of progesterone. Due to this, retinoic acid production is also impaired (normally converts estrodiol to its less potent form = estrone)
Macrophage endometriosis impacts
Increases proinflamatory cytokines. Increases Macrophage migration inhib factor (MIF) = keeps macrophages at the lesion and recruits more. Increases Nerve Growth Factor (NGF) = increased density of sensory nerves, causing more intense pain.
Difficulty in creating in vivo endometriosis research models
Very few species undergo spontaneous menstruation (bats, some primates, humans and small mammals in Australlia). Even rodents don’t undergo spontaneous menstruation (covert instead = reabsorption rather than shedding of endometrium). Furthermore, animals do not correlate to humans in this field at all (~80% failure rate), and the approach with animals (to reduce lesion sizes) is not the same approach as what is used with px’s (to help with pain management and underlying causes).
Mouse endometriosis models (chimeric)
Human endometrial tissue implanted in immunocompromised rodent abdomen (allows the human tissue to grow without being attacked). Not fully translatable due to no inflammatory response
Syngeneic mouse model
Removal of mouse uterus, chopped into pieces (mimic ectopic lesions) and then reinsterted into abdomen of another mouse. Allows for genetic mutations if wanted and mimics retrograde menstruation
Limitations of rodent models (endometriosis)
Only superficial lesions appear (not super clinically relevant, as we are more concerned with deep set lesions). ex) antioxidant (resveratrol) therapy was effective for rodents but not in humans
In vitro endometriosis models
Endometriotic cell lines = we created (feature more prostoglandins, cytokines, estrogen, vascular growth and steroid synthesis = as expected, helps to match what is seen in vivo). Used to create 3d models
Human primary endometrial cells
Isolated from eutopic or ectopic human endometrial tissue samples. Used to create 3d models. Can be co-cultured with other cells to study interactions (epithelial, stromal and immune). Then exposed to human peritoneal fluid to conduct studies
Aromatase inhibitors (endometriosis trial treatment)
Target the excessive estrogen produced locally by the ectopic lesions (rather than the ovaries estrogen produciton). Reduce pelvic pain and lesion size. Can be paired with progestin (which suppresses ovarian follicle development). So far well tolerated, only mild side effects
COX inhibition (endometriosis trial treatment)
Prevents prostoglandin production, reducing pelvic pain. Unfortunatley, if not selective can target GI prostoglandin production, having negative impacts and if selective, can have cardiovascular risks.
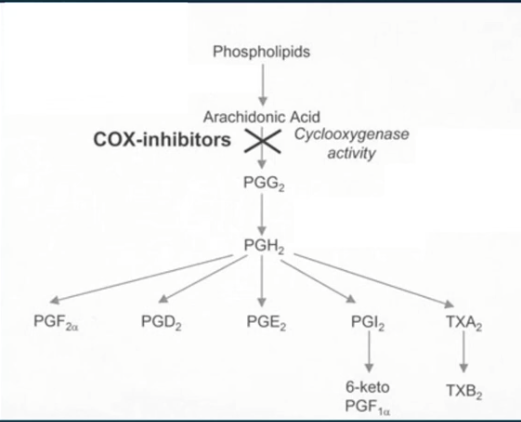
Prostoglandin receptor antagonist (endometriosis trial treatment)
Effective in mouse models (decreases growth of lesions, angiogenesis and presence of nerve fibers in the lesions. Also suppresses inflammation and restores endometrial lining).
Estrogen receptor beta (endometriosis trial treatment)
Master regulation of endometrial tissue. Estrogen binding here prevents TNF cell death and enhances IL-1B = cell adhesion and proliferation. Selective antagonists of this receptor could be a new approach to break up lesions