FINALS PMOC Antipsychotic
1/70
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
71 Terms
Reserpine and chlorpromazine
first drugs found to be useful to reduce symptoms of schizophrenia
Psychosis
denotes a variety of mental disorders
SCHIZOPHRENIА
Characterized by clear sensorium but a marked thinking disturbance.
Excess dopamine in mesolimbic system (involved in emotions)
POSITIVE SCHIZOPHRENIА
NOT present in the NORMAL individual but are added to the schizophrenic person
POSITIVE SCHIZOPHRENIА
DELUSIONS (False beliefs) HALLUCINATIONS (Perceptional distortion
NEGATIVE SCHIZOPHRENIА
PRESENT in NORMAL person but MISSING in the schizophrenic individual
NEGATIVE SCHIZOPHRENIА
Flat affect
Alogia
Avolition
Asociality
Anhedonia
TYPICAL NEUROLEPTICS
Blockade of dopamine 2 receptors in the CNS especially in mesolimbic system
Chlorpromazine, Haloperidol
TYPICAL NEUROLEPTICS examples
ATYPICAL NEUROLEPTICS
Blockade of dopamine 2 and serotonin 2a receptors in the brain.
Clozapine, Olanzapine
ATYPICAL NEUROLEPTICS examples
Phenothiazine derivatives
Butyrophenones
Thioxanthenes
Diphenylbutylpiperidine
Pimozide
TYPICAL ANTIPSYCHOTICS
Loxapine - succinate
Dibenzoxazepines
Clozapine
Dibenzodiazepines
Olanzapine
THIOBENZODIAZEPINE
Sertindole
FLUOROPHENYLINDOLE
MOLINDONE
beta - Aminoketone
Extrapyramidal symptoms (EPS)
-First-generation antipsychotics
- dystonia, parkinsonism, and akathisia
Weight gain
Drowsiness
NEUROLEPTICS Side effects:
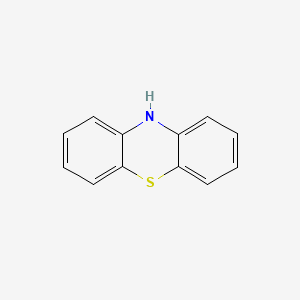
SAR
Phenothiazine
Phenothiazine
a three carbon side chain =
Inc. potency
Increase D2 blocking effect
Modifications of alkyl side chain at R10:
nitrogen of phenothiazine and basic amino group is connected by:
Chlorpromazine (Thorazine®)
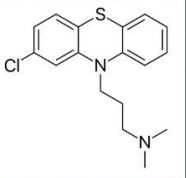
Chlorpromazine (Thorazine®)
First phenothiazine compound introduced
antihistaminic and sedative effect only
Modifications of alkyl side chain at R10:
the ring and side-chain nitrogens if separated by the two-carbon chain exhibits only
stable, highly lipophilic esters
fluphenazine and long-chain alcohols form
Promethazine
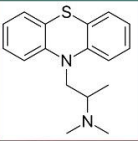
Ethopropazine
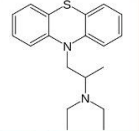
eplaced with piperidyl or Piperazine groups.
The activity is retained or increased if the amino group is
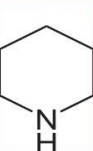
Piperidine
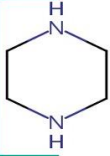
Piperazine
Introduction of hydroxyl, methyl, hydroxy ethyl groups of piperidine and Piperazine moieties
what would be added to increase the potency.
Phenothiazine ring substituents at R2:
substitution produces greatest antipsychotic property
Chlorpromazine
Promazine
triflupromazine
Propyl dialkyl side chain
Thioridazine
Mesoridazine
Alkyl piperidyl side chain
Prochlorperazine
Trifluoperazine
Perphenazine
Fluphenazine
Propyl piperazine side chain
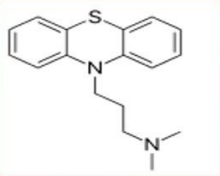
Promazine (Sparine®)
2 Cl: ↓ antipsychotic
Promazine (Sparine®)
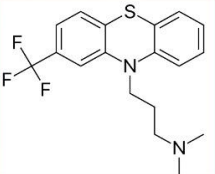
Triflupromazine (Vesprin®)
2 CF3: ↑ antipsychotic
Triflupromazine (Vesprin®)
Thioridazine (Mellaril)
Low EPS
High anticholinergic
At high doses, pigmentary retinopathy
Mesoridazine (Serentil)
No pigmentary retinopathy
Thioridazine (Mellaril)
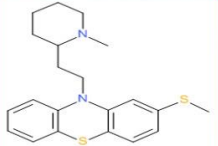
Mesoridazine (Serentil)
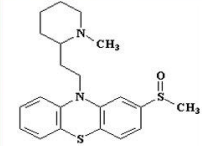
Prochlorperazine (Compazine)
High EPS but low sedative and autonomics effects
Perphenazine (Trilafon)
Effective antipsychotic and anti-emetic
Prochlorperazine (Compazine)
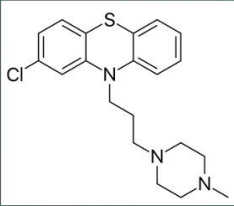
Perphenazine (Trilafon)
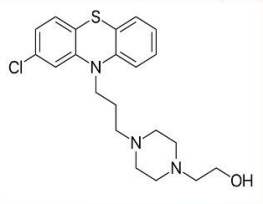
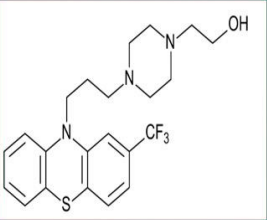
Fluphenazine (Permitil)
2-CF3 • Most potent
Fluphenazine (Permitil)
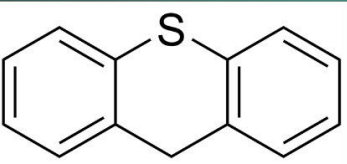
Thioxanthenes
lacks the ring nitrogen of phenothiazines and have side chain attached to the double bond
Thioxanthenes
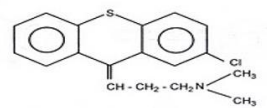
Chlorprothixene
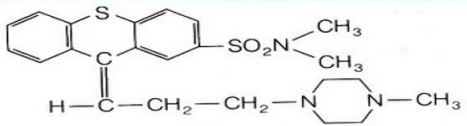
Thiothexene
Clozapine (Clozaril®)
Effective against both positive and negative symptoms
Low EPS
agranulocytosis
Clozapine
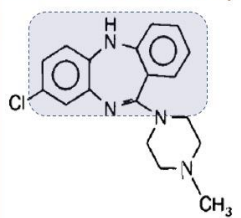
Olanzapine (Zyprexa®)
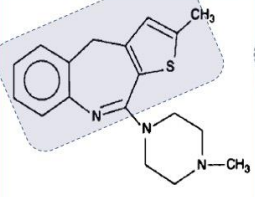
Quetiapine (Seroquel®)
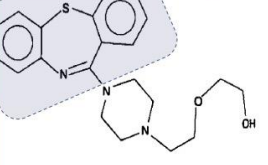
Quetiapine (Seroquel®)
Used in depression and bipolar
Off-label use: for patients with PTSD
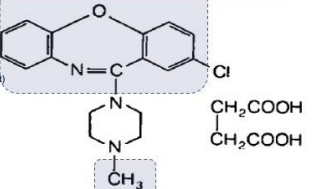
LOXAPINE (Daxolin®)
EWG at pos 2 .
M: Aromatic hydroxylation
Metabolite; amoxapine (antidepressant)
LOXAPINE (Daxolin®)
Fluorobutyrophenones
Chemically unrelated to phenothizines but have similar activity
Fluorobutyrophenones
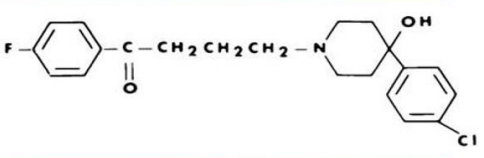
HALOPERIDOL (Haldol)
Chemically unrelated to phenothiazines but have similar activity
HIGH EPS
Most widely used antipsychotic
Used for intractable hiccups
Droperidol
Short acting, highly sedating
Droperidol + Fentanyl
InnovarⓇ
HALOPERIDOL
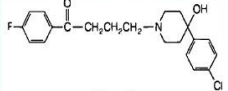
Droperidol
CH2CH2CH2 - important in its activity
3o amine - neuroleptic acitivity
Fluorobutyrophenones SAR
Risperidone (Risperdal)
Low risk of tardive dyskinesia
Decrease EPS
Risperidone (Risperdal)
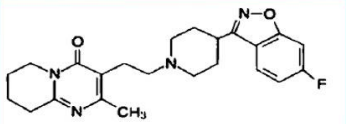
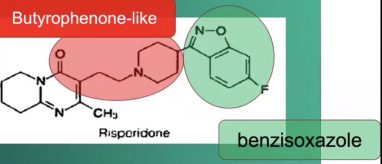
Risperidone (Risperdal) parts