3.1.4.4 bond enthalpies
1/12
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
13 Terms
the enthalpy needed to break a particular covalent bond is called what?
the bond dissociation energy
define bond dissociation energy
enthalpy change to break the bond in 1 mol of gaseous molecules, to form gaseous atoms
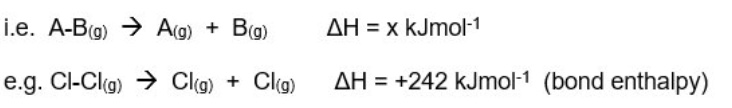
explain what bond enthalpies are
some bonds only appear in one molecule eg Cl-Cl only appears in Cl₂
the values for these bonds would therefore be simply named ‘bond enthalpies’
the values for bond enthalpies would be ______
accurate
explain why the values for some bonds are called mean bond enthalpies
other bonds appear in many molecules eg C-H appears in methane, ethanol, propene etc
the bond enthalpy for these types of bonds would be slightly different in different molecules
the values provided for these bonds would therefore be named ‘mean bond enthalpies’
why would the values for mean bond enthalpies not be 100% accurate?
as they are average values from a range of compounds
define mean bond enthalpy
enthalpy required to break 1 mol of covalent bonds averaged over a range of compounds
in what molecules is the term mean bond enthalpy used?
polyatomic molecules
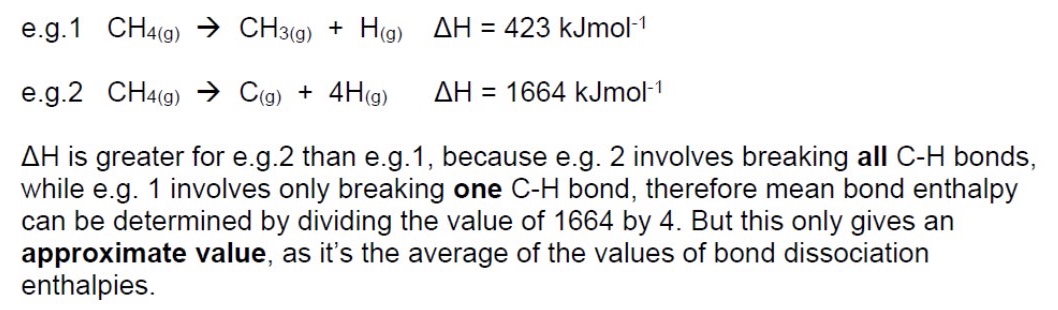
what can mean enthalpy data be used to calculate?
the enthalpy change of a reaction
give the equation of calculating enthalpy change reaction using mean enthalpy data

what can be helpful to do when calculating enthalpy change?
drawing out the displayed formulae of the substances in the question

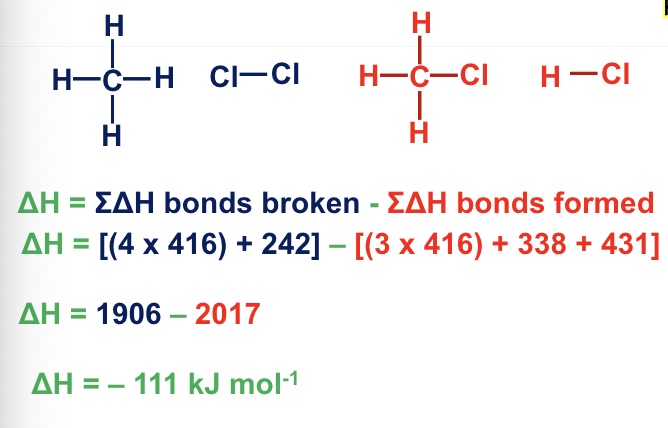

rearranging bond enthalpy calculations
