Liver enzymes and evaluation
1/98
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
99 Terms
Which species have ALT as a fairly liver-specific enzyme?
Cats, dogs, rabbits, rats, and primates.
Besides the liver, where else can ALT be found in the body?
Skeletal muscle (5% of liver activity) and cardiac muscle (25% of liver activity).
What does a marked elevation of ALT (>10x reference) typically indicate?
Toxic, hypoxic, or traumatic conditions, or acute severe liver injury/necrosis.
Why is ALT not a useful diagnostic enzyme in large animals?
It is present only in very low concentrations in the hepatocytes of large animals.
What enzymes are preferred over ALT for diagnosing liver disease in large animals?
GLDH (Glutamate Dehydrogenase) or SDH (Sorbitol Dehydrogenase).
What is the primary diagnostic requirement when interpreting elevated AST levels?
Concurrent estimation of CK (Creatine Kinase) is required to exclude muscle damage.
How does the half-life of AST compare to CK in serum?
AST has a longer half-life in serum than CK.
What is the primary clinical use of GLDH (GDH/GMD)?
It is primarily used in large animals as an indicator of acute hepatocellular necrosis.
What is a major practical limitation of using SDH (ID) for diagnosis?
It is very unstable, with a half-life of up to 5 hours at 21 °C, making it impractical outside of research or teaching hospitals.
What are the three main sources of ALP isoforms?
Liver (L-ALP), bone (B-ALP), and corticosteroid-induced (C-ALP in dogs).
What does an increase in ALP generally indicate in a clinical setting?
Cholestasis (intra- or post-hepatic).
How does the diagnostic sensitivity of ALP for cholestasis differ between dogs and cats?
Dogs have high diagnostic sensitivity (usually elevated before icterus), while cats have very low sensitivity (usually icteric before elevation).
Why is any increase in ALP significant in cats?
L-ALP has a very short half-life in cats (< 8 hours).
What is the significance of Scottish Terriers having high ALP levels?
Adult Scottish Terriers can have high ALP (up to 4-5x) without corresponding abnormal liver histopathology, though some may have vacuolar hepatopathy.

What is the effect of corticosteroids on ALP in dogs?
They induce the synthesis of a specific isoenzyme (Ci-ALP), which can lead to steroid/vacuolar hepatopathy.

What is the mechanism of cholestasis in steroid hepatopathy?
Hepatocyte swelling (due to glycogen/hydropic change) impairs bile flow in the canaliculi.
What drug is known to induce liver disease and elevate ALP/ALT?
Phenobarbital.
What are the two main types of changes seen in vacuolar hepatopathy?
Glycogen deposition and hydropic change.
What is the relationship between thyroid hormone and the liver in cats?
High thyroid hormone levels are hepatotoxic and can cause mild to moderate ALT elevations.
What is the clinical significance of AST increasing with exercise?
AST can increase by 30% with exercise, and resting levels in horses in training are often 50-100% higher than those not in training due to a muscle component.

What enzyme can be used as an alternative or adjunct to ALP in small animals?
GGT (Gamma-glutamyl transferase).
What causes mild to moderate ALT elevations in aged animals?
Various factors including diabetes mellitus, passive congestion from heart disease, GI disease, and certain medications.
How does bilirubin relate to hepatobiliary disease?
It is an excretory product; its accumulation (hyperbilirubinemia) is often associated with cholestasis.
What is the primary cause of increased ALP in dogs?
Induction by glucocorticoids, bile acids, or phenobarbital, or cholestasis.
What is the primary source of Bone Alkaline Phosphatase (B-ALP)?
It is produced by osteoblasts.
Why is B-ALP elevated in young, growing animals?
It is 2-3 times higher than normal due to active bone growth, typically returning to adult ranges by 15 months of age.
What clinical conditions can cause an increase in B-ALP?
Bone disease, including tumors, infections, and fractures.
Why might ALP levels be significantly elevated in post-suckling pups?
Due to the ingestion of colostral ALP; these values typically decrease within 10 days.
How does pancreatitis affect ALP levels in dogs?
It can cause mild increases in ALP due to swelling or fibrosis around the bile duct.
Why is ALP not a useful diagnostic marker in ruminants?
The reference intervals for ALP in ruminants are too wide to be clinically useful.
What are the two primary origins of increased ALP in horses?
Liver origin (cholestasis) or osteoblast production (bone).
What causes steroid-induced ALP isoenzymes in chronically ill or stressed dogs?
The release of endogenous glucocorticoids.
What is the clinical significance of GGT as a liver marker?
It is a marker of cholestasis or biliary hyperplasia, indicating impaired bile flow.
How does the diagnostic sensitivity of GGT compare to ALP in dogs and cats?
In dogs, sensitivity is similar to ALP but the magnitude of increase is less; in cats, sensitivity is similar to ALP for most liver diseases except hepatic lipidosis.
In which species is GGT a better diagnostic indicator of cholestasis than ALP?
Large animals (horses and cattle).
What non-hepatic factors can induce GGT elevation in dogs?
Corticosteroids and anticonvulsants (e.g., barbiturates).
If GGT is elevated significantly more than ALP, what does this suggest?
Biliary tree disease, as GGT is located in the biliary epithelium, whereas ALP is found in hepatocellular membranes along bile canaliculi.
How does chronic fascioliasis affect GGT levels in animals?
It causes bile duct damage leading to GGT increases, though these are not primarily cholestatic in nature.
How can GGT be used to assess passive transfer in calves?
GGT is excreted in bovine colostrum; measuring GGT levels in calves under 2 weeks of age indicates successful colostrum intake.
How do GGT levels in donkeys compare to those in horses?
Donkeys typically have 2-3 times higher GGT levels than horses.
Why is ALP considered the most clinically significant liver enzyme in cats?
It has a very short half-life, so even small increases are highly significant.
What is the classic enzyme pattern for steroid-induced liver changes in dogs?
High ALP with relatively low or normal ALT.
What does an increase in AST without an increase in CK suggest?
Liver injury (as opposed to muscle injury, where both would be elevated).
Which enzymes should be evaluated when assessing liver disease in horses?
GGT, SDH, and GLDH.
What are the three main mechanisms of increased liver enzyme activity?
Hepatocellular damage, biliary stasis (cholestasis), and enzyme induction.
What does the presence of ammonium biurate crystalluria suggest in a hepatic context?
It is often associated with hepatic disorders, such as portosystemic shunts.
What is the effect of daylight exposure on serum bilirubin samples?
It causes a false decrease in bilirubin concentration.
How do lipemia and hemolysis affect bile acid concentration results?
Lipemia can cause false elevations, while hemolyzed blood samples can cause false decreases.
What are the components of Total Bilirubin?
Total Bilirubin = Direct (conjugated) bilirubin + Indirect (unconjugated) bilirubin + Delta bilirubin.
What is the origin of bilirubin?
It originates from the breakdown of hemoglobin in old red blood cells by macrophages.
What is the difference between unconjugated and conjugated bilirubin regarding solubility?
Unconjugated bilirubin is insoluble (indirect), while conjugated bilirubin is water-soluble (direct).
What is delta bilirubin?
It is a fraction of conjugated bilirubin that is covalently bound to albumin, which is not filtered by the kidneys and has a half-life of 8-20 days.
What are the three main classifications of icterus (jaundice)?
Pre-hepatic (hemolytic), Hepatic (intra-hepatic), and Post-hepatic (obstructive).
Why does bilirubinuria often occur before jaundice?
Because conjugated bilirubin is water-soluble and can be filtered by the kidneys as soon as levels rise in the blood, whereas jaundice requires higher tissue accumulation.
What are common causes of pre-hepatic (hemolytic) hyperbilirubinemia?
IMHA, parasites, zinc or copper toxicity, RBC enzyme deficiencies, and hypophosphatemia.
How does severe hemolysis lead to cholestasis?
Hemolysis causes anemia and hypoxia, which damages hepatocytes and interferes with their ability to excrete bilirubin.
Why does fasting/anorexia cause hyperbilirubinemia in horses and cats?
It causes decreased uptake of unconjugated bilirubin by hepatocytes, often due to competition from free fatty acids mobilized from adipose tissue.
What is sepsis-associated cholestasis?
A functional hyperbilirubinemia where inflammatory mediators like TNF-alpha inhibit bile acid transport to the canaliculi, even without physical obstruction.
What are the primary causes of obstructive (cholestatic) hyperbilirubinemia?
Intrahepatic causes (hepatocyte swelling, lipidosis, lymphoma) and extrahepatic causes (tumors, calculi, bile duct rupture).
Why might a clinician incorrectly assume cholestasis is persisting even when a patient improves?
Because delta bilirubin has a long half-life (8-20 days) and remains in the blood even after the underlying cause of cholestasis has resolved.
In which species is bilirubinuria considered clinically insignificant?
Dogs and ruminants, as they have a low renal threshold and may show bilirubinuria even in health.
In which species is bilirubinuria always considered significant?
Cats and horses, as they have a high renal threshold for bilirubin.
What are bile acids and what is their primary function?
They are cholesterol-derived anionic acids required for the digestion and emulsification of dietary lipids.
Are bile acid tests specific to the type of liver disease?
No, they are sensitive indicators of hepatobiliary function but are not specific to the underlying disease process.
What is the clinical significance of a serum bile acid level > 25 µmol/L?
It is considered abnormal, regardless of whether the sample was taken fasting or post-prandially.
How is a paired bile acid test performed?
A pre-prandial sample is taken, followed by a small meal, and a second sample is taken 2 hours post-prandially.
What is the SNAP-BAT test?
A highly accurate, semi-quantitative test for bile acids that provides immediate results with low intra-assay variability.
What happens to bilirubin in the gut?
It is oxidized to stercobilin, which provides the brown pigment to feces.
What is the clinical term for yellow pigment accumulation in tissues?
Icterus or jaundice.
What does bile peritonitis cytology reveal?
Leakage of bile into the abdomen due to rupture of the gall bladder or bile duct, which can lead to hyperbilirubinemia.
What is the role of Z protein in the liver?
It is a fatty acid binding protein that can be occupied by free fatty acids during fasting, thereby reducing the uptake of unconjugated bilirubin.
List four tests of hepatic function mentioned in the notes besides bilirubin and bile acids.
Ammonium (NH4), Urea N, Cholesterol, and Plasma proteins.
What are four non-pathological causes of increased fasting bile acids?
Inadequate fasting, excitement causing spontaneous gallbladder contraction, glucocorticoid (steroid) hepatopathy, and hyperadrenocorticism (Cushing's disease).
How does lipemia affect serum bile acid test results?
It causes a false increase in bile acid levels.
Why is bile acid testing considered not useful in patients with high bilirubin?
Because obstructive cholestasis (intrahepatic or post-hepatic) already impairs bile acid excretion, making the test redundant or misleading.
How does inflammation lead to increased serum bile acids?
Inflammation releases TNF-alpha, which decreases the transport/excretion of bile acids into the canaliculi, causing them to accumulate in hepatocytes and regurgitate into the blood.
Is the degree of bile acid elevation proportional to the severity of liver malfunction?
No, the degree of increase is not proportional to the degree of liver malfunction.
Define a portosystemic shunt (PSS).
A vascular anomaly that allows blood from the hepatic portal circulation to bypass the liver and be delivered directly into the systemic circulation.
Which type of portosystemic shunt is more common in small breed dogs?
Extrahepatic shunts.
What hematological finding is often seen in dogs with PSS and why?
Microcytic anemia with hypoferremia, caused by abnormal iron sequestration or transport rather than true iron deficiency.
What type of urinary crystals are associated with liver failure and why?
Ammonium biurate crystals; they form because the liver fails to convert uric acid to allantoin, and the excess uric acid combines with ammonium.
What is Hepatic Microvascular Dysplasia (HMD/MVD)?
A congenital defect characterized by the abnormal development or absence of the portal microvasculature in the liver.
How is Hepatic Microvascular Dysplasia (HMD) definitively diagnosed?
It can only be diagnosed via liver biopsy.
What is the threshold for bile acid levels suggesting liver malfunction in horses?
Greater than 11 µmol/L.
Why should ammonia samples not be frozen and why must they be transported on ice?
Ammonia is unstable; freezing or delayed processing can cause false increases due to hemolysis, while exposure to air causes false decreases as NH4+ converts to NH3 gas.
What are two primary reasons for low cholesterol levels in liver disease?
Decreased hepatic production (chronic liver disease or PSS) or loss (malabsorption, maldigestion, PLE, or EPI).
At what point in the loss of functional hepatic mass is hypoalbuminemia typically observed?
Not until 60-80% of the functional hepatic mass has been lost.
Why is a coagulation screen recommended prior to liver FNA or biopsy?
Because the liver synthesizes most coagulation factors (except FVIII), and these may be depleted in chronic liver disease, increasing bleeding risk.
How does hepatic failure affect glucose levels?
Glucose values can be decreased due to reduced hepatic function (gluconeogenesis/glycogenolysis) or increased due to decreased hepatic uptake.
What is the primary cause of feline hepatic lipidosis?
Massive accumulation of triglycerides (>50%) within hepatocytes, often multifactorial involving obesity and anorexia.
Why is GGT typically 'normal' in cases of feline hepatic lipidosis?
Because the lipidosis causes swelling of hepatocytes that compresses bile canaliculi, inducing ALP, but the biliary tree epithelial cells themselves are not primarily affected.
What are common clinical signs of feline hepatic lipidosis?
Anorexia, lethargy, weight loss, vomiting/diarrhea or constipation, jaundice, and a large non-painful liver.
Which blood cell abnormality is commonly seen in cats with hepatic lipidosis?
Acanthocytes, potentially due to alterations in cell membrane lipids.
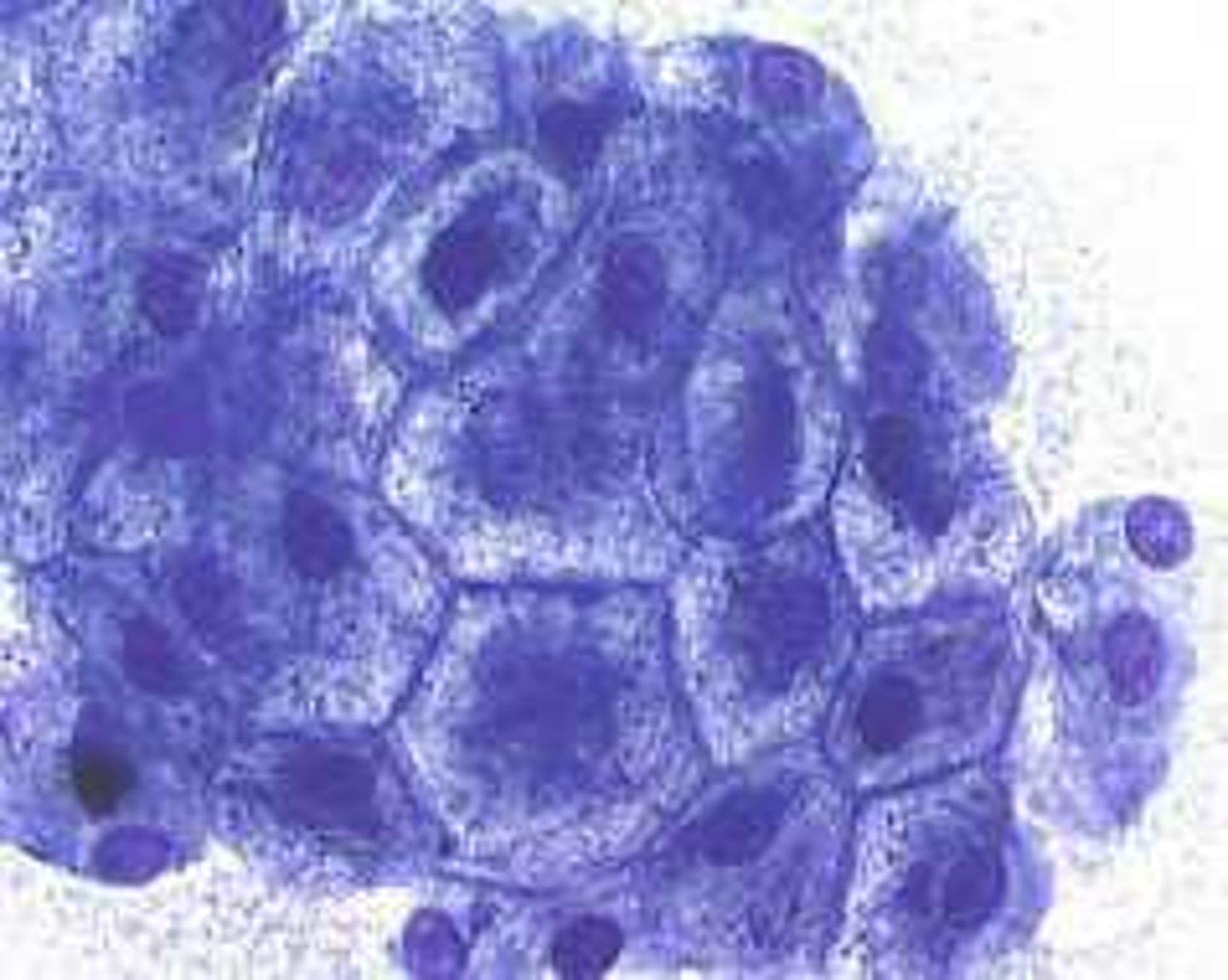
What are the key laboratory findings in a patient with chronic hepatitis leading to hepatic insufficiency?
Increased ALT/AST, marked increase in ALP, mild increase in GGT, marked increase in bile acids, and low albumin.
What is the urea cycle's role in hepatic function?
It converts ammonia (NH4+) into urea; failure of this cycle leads to hyperammonemia.
What is the clinical significance of hyperammonemia?
It may precipitate hepatic encephalopathy.
What are the common markers of hepatic dysfunction?
Hypoalbuminemia, hypocholesterolemia, hyperammonemia, hypercholemia, hyperbilirubinemia, decreased coagulation factors, and decreased urea nitrogen (UN).
What is the effect of hemolysis on serum bile acid measurements?
It can lead to artifactually decreased values.
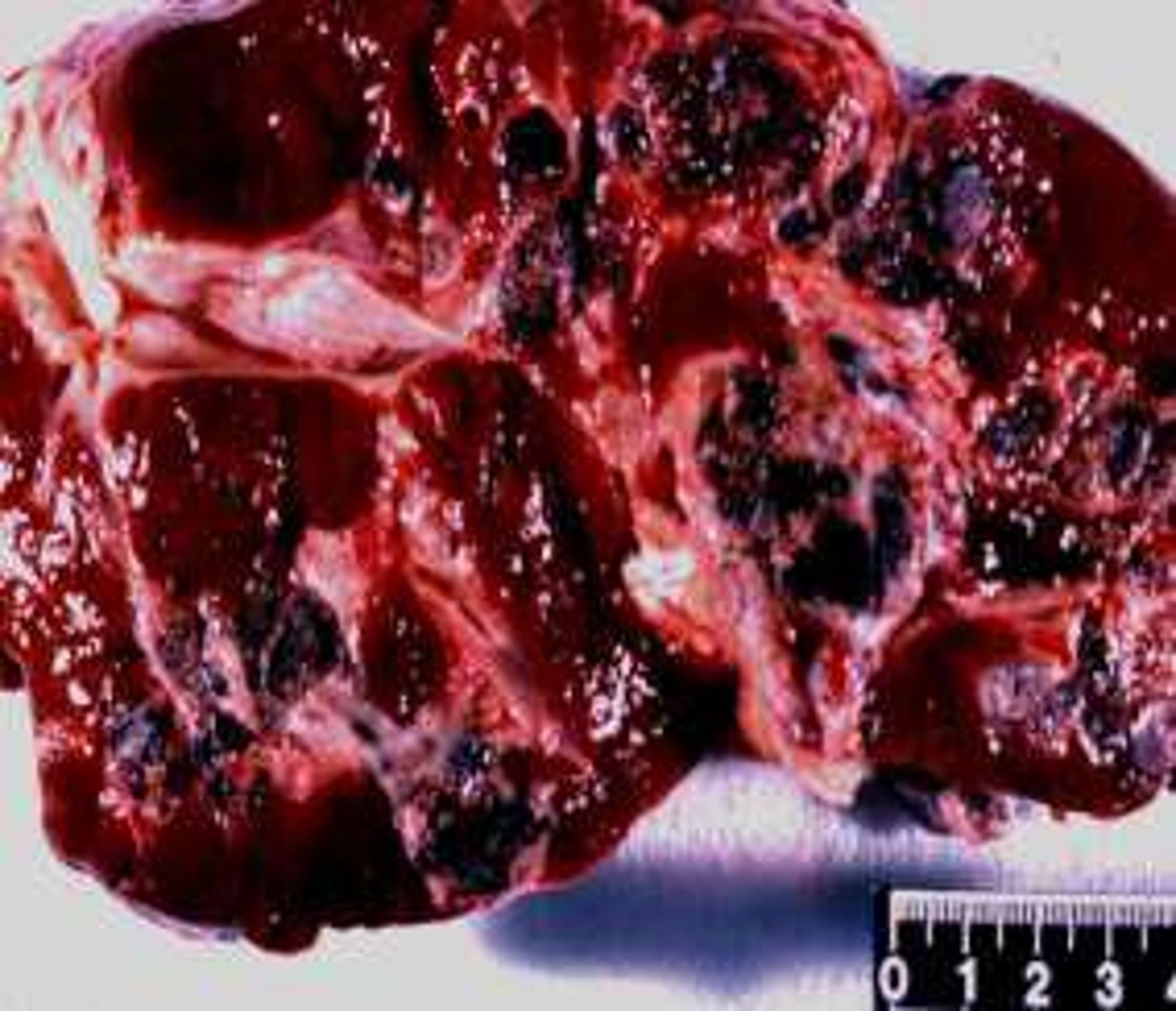
What is the primary reason for performing an ultrasound-guided FNA in a liver case?
To identify specific abnormal areas, such as tumor cells or hepatic lipidosis.