Organic chemisty exam 3 ffs god help me
1/237
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
238 Terms
Q: What is the α-carbon?
A: The carbon directly next to a carbonyl (C=O)
Q: What is the α-carbon in simple terms?
A: “The carbon touching the carbonyl carbon” (the carbon thats double bonded to the oxygen)
Q: Why are α-carbons important?
A: Reactions happen there instead of the carbonyl
Q: What usually gets replaced on the α-carbon?
A: A hydrogen (H)
Q: What replaces that hydrogen?
A: An electrophile (E⁺)
Q: What are keto and enol forms called together?
A: Tautomers
Q: Which form is most common?
A: Keto form (>99%)
Q: Why is keto favored?
A: C=O is stronger than C=C
Q: Why are enols reactive?
A: They have extra electron density (they’re “electron rich”)
Q: What do enols react with?
A: Electrophiles
Q: What is an enolate?
A: A negatively charged form of a carbonyl compound
Q: How is an enolate formed?
A: A base removes an α-hydrogen
Q: Why can α-hydrogens be removed?
A: They are acidic (pKa ~20)
Q: Why are α-hydrogens acidic?
A: The negative charge is spread out (resonance)
Q: What happens if you use a strong base?
A: You get more enolate
Q: What happens if you use a weak base?
A: You get very little enolate
Q: What is LDA?
A: A very strong, bulky base
Q: What does LDA do?
A: Forms enolate 100%
Q: Why doesn’t LDA attack molecules?
A: It’s too bulky → not nucleophilic
Q: Where does an enolate attack?
A: The α-carbon
Q: Does an enolate attack at oxygen or carbon?
A: Carbon (α-carbon)
Q: When is enol NOT tiny (<1%)?
A: In β-dicarbonyl compounds
Q: Why are enols stable in β-dicarbonyls?
A: resonance hydrogen bonding
Q: What is the geometry of an enolate?
A: Trigonal planar (flat)
Q: What happens if the α-carbon is chiral and you add base?
A: Racemization (mix of both stereoisomers)
Q: When enolates react, what bond is formed?
A: New C–C bond at the α-carbon
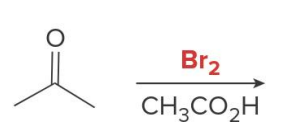
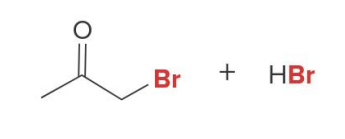
Q: Halogenation in base gives what?
A: MULTIPLE halogens (polysubstitution)
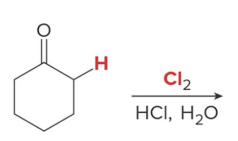
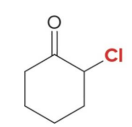
Q: Halogenation in acid gives what?
A: ONE halogen added (monosubstitution)
Q: Why does base give multiple halogens?
A: Each halogen makes the next H easier to remove
Q: What functional group is required for haloform?
A: Methyl ketone (R–CO–CH₃)
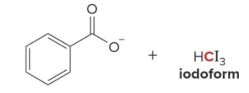
Q: What confirms a haloform reaction occurred?
A: Yellow solid (CHI₃)
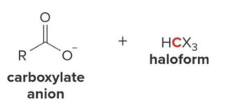
Q: What happens in haloform reaction?
A: CH₃ group is removed becomes carboxylate + CHX₃
Q: What is the kinetic enolate?
A: Less substituted, forms faster
Q: Conditions for kinetic enolate?
A: LDA, low temp (−78°C), bulky base
Q: What is the thermodynamic enolate?
A: More substituted, more stable
Q: Conditions for thermodynamic enolate?
A: weaker base, room temp, protic solvent
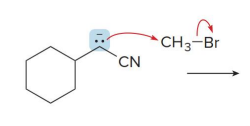
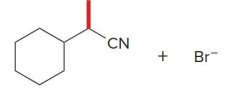
Q: What is enolate alkylation?
A: Adding an R group to the α-carbon
Q: What type of reaction is enolate alkylation?
A: SN2
Q: What type of alkyl halides work best?
A: Primary (NOT tertiary)
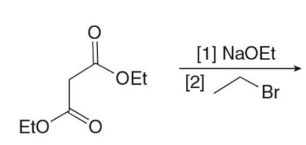
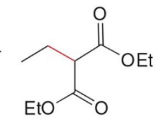
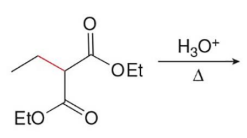
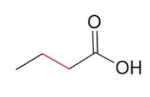
Q: What does malonic ester synthesis make?
A: Carboxylic acids
Q: Steps of malonic ester synthesis?
A:
Deprotonate
Add R group
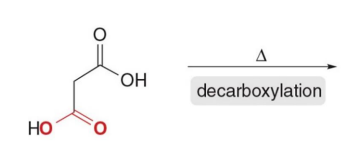
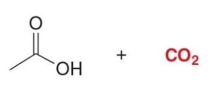
Hydrolysis + heat → CO₂ leaves
Q: What leaves during decarboxylation?
A: CO₂
Q: What does acetoacetic ester synthesis make?
A: Ketones
Q: Malonic vs acetoacetic difference?
A:
malonic → acid
acetoacetic → ketone
Q: If you see LDA, what should you think?
A: Enolate formation
Q: If you see Br₂ + acid → what happens?
A: Single α-halogenation
Q: If you see Br₂ + base → what happens?
A: Multiple halogens
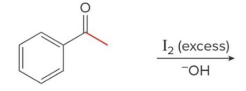
Q: If you see I₂ + base + CH₃ ketone → what happens?
A: Haloform (yellow CHI₃)
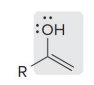
name?
enol
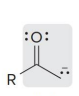
name
enolate
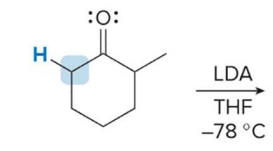
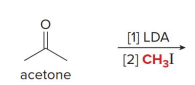
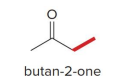
kenetic or thermo
kenetic
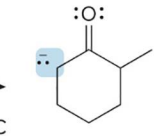
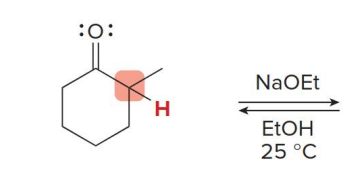
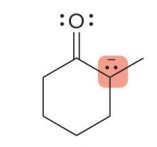
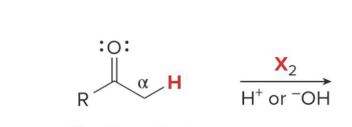
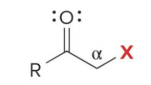
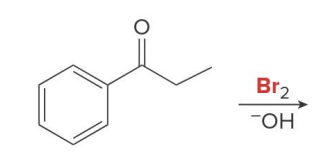
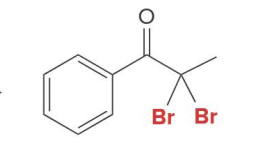
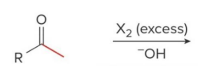
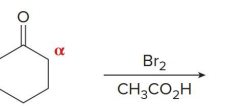
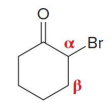
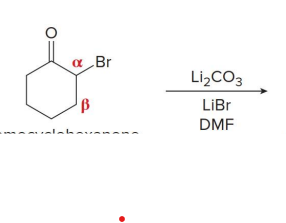
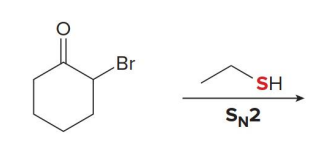
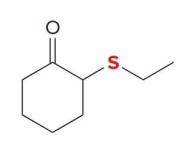
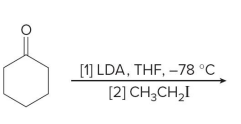
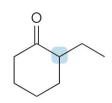
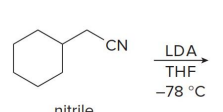
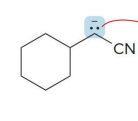
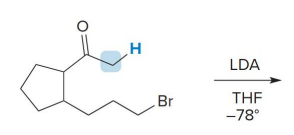
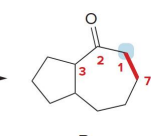
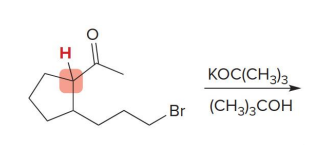
what kind of reaction is this
thermo so its slower and doesnt choose the easier alpha carbon
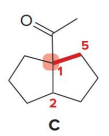
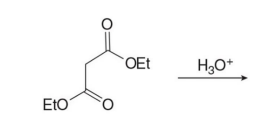
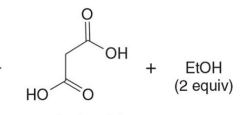
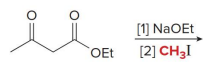
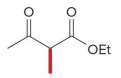
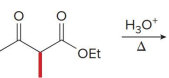
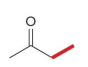
Q: What do condensation reactions between carbonyls do?
A: Form new C–C bonds
Q: In these reactions, what are the two roles?
A:
one = enolate (nucleophile)
one = carbonyl (electrophile)
Q: What is an aldol reaction?
A: Two carbonyls react → form β-hydroxy carbonyl
Q: What do you ALWAYS form first in aldol?
A: β-hydroxy carbonyl (OH on β-carbon)
Q: What conditions cause aldol reaction?
A: Base (OH⁻, OR⁻)
Q: Where does the new C–C bond form in aldol?
A: α-carbon → attacks carbonyl carbon
Q: What happens to the aldol product under basic conditions?
A: Loses H₂O (dehydration)
Q: What is formed after dehydration?
A: α,β-unsaturated carbonyl (alkene + C=O)
Q: Aldol reaction overall pattern?
A: carbonyl → β-OH → loses H₂O → alkene
Q: What is an aldol condensation?
A: Aldol reaction + loss of H₂O
Q: What mechanism removes H₂O in aldol condensation?
A: E1cB
Q: What is special about E1cB?
A:
2 steps
intermediate = carbanion (NOT carbocation)
Q: When given an aldol product, what bond do you break?
A: Between α and β carbons
Q: After breaking α–β bond, what do you get?
A: Two carbonyl compounds
Q: How do you identify the β-carbon?
A: The carbon two away from the carbonyl (the carbon nect to the alpha carbon)
Q: What is a crossed aldol reaction?
A: Two different carbonyls react
Q: What happens if both have α-hydrogens?
A: Multiple products (messy)
Q: When does crossed aldol give ONE product?
A: When only one compound has α-hydrogens
Q: Why do β-dicarbonyls react easily?
A: α-hydrogens are very acidic
Q: What are β-dicarbonyls called?
A: Active methylene compounds
Q: What is a directed aldol reaction?
A: You choose which enolate forms using LDA
Q: What does LDA give in aldol?
A: Kinetic enolate
Q: If you see β-OH + carbonyl → what reaction?
A: Aldol
Q: If you see alkene + carbonyl (α,β-unsaturated)?
A: Aldol condensation
Q: If asked to “break apart product”?
A: Break α–β bond
Q: In an aldol reaction, which molecule becomes the enolate?
A: The one with α-hydrogens
Q: In an aldol reaction, which molecule is the electrophile?
A: The one whose carbonyl gets attacked