AP Biology: Unit 1 - Chemistry of Life (Majoros)
1/90
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
91 Terms
organic chemistry
The study of carbon & hydrogen compounds (organic compounds).
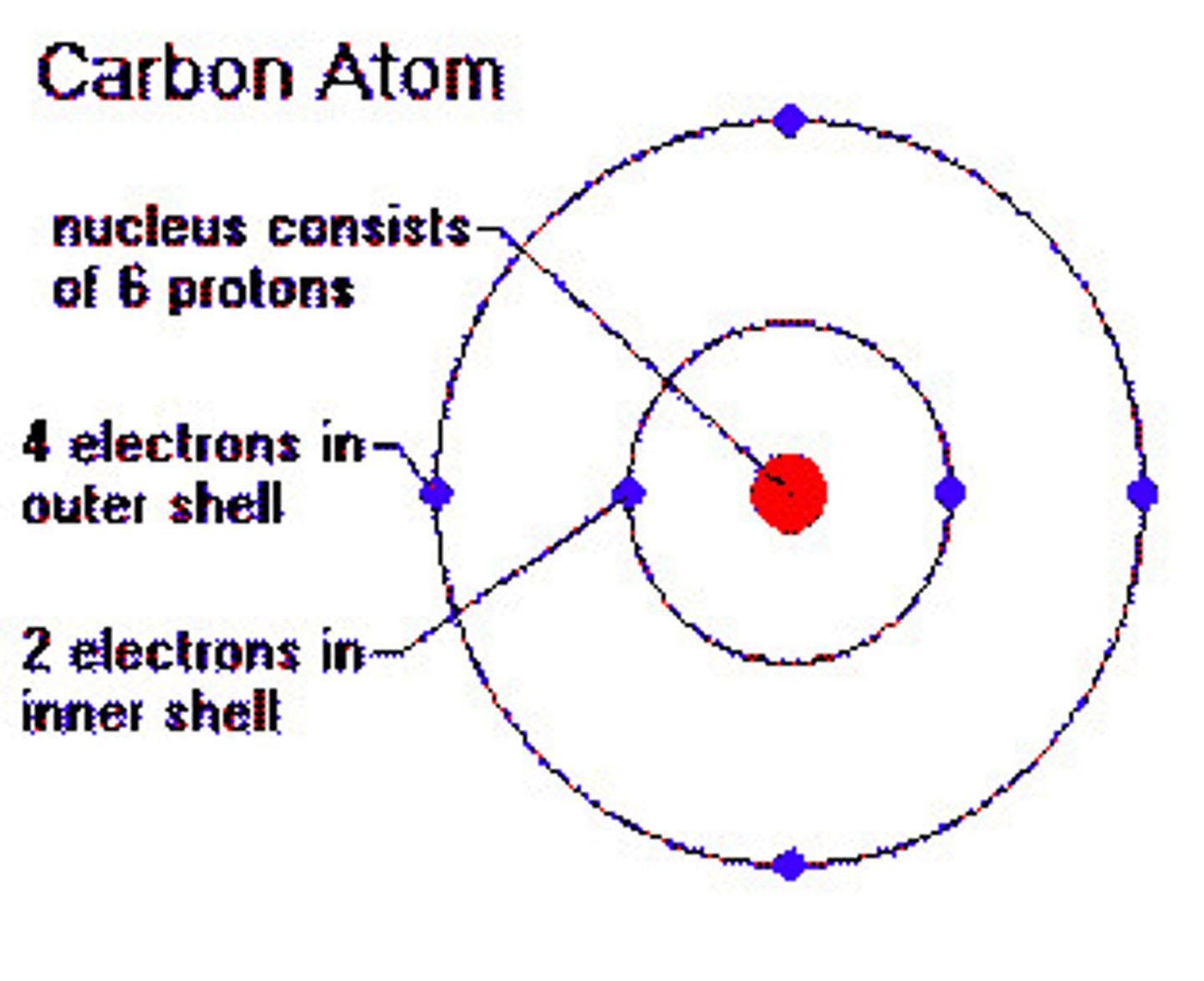
carbon
this atom has 4 electrons in valence shell and can form 4 covalent bonds with other atoms
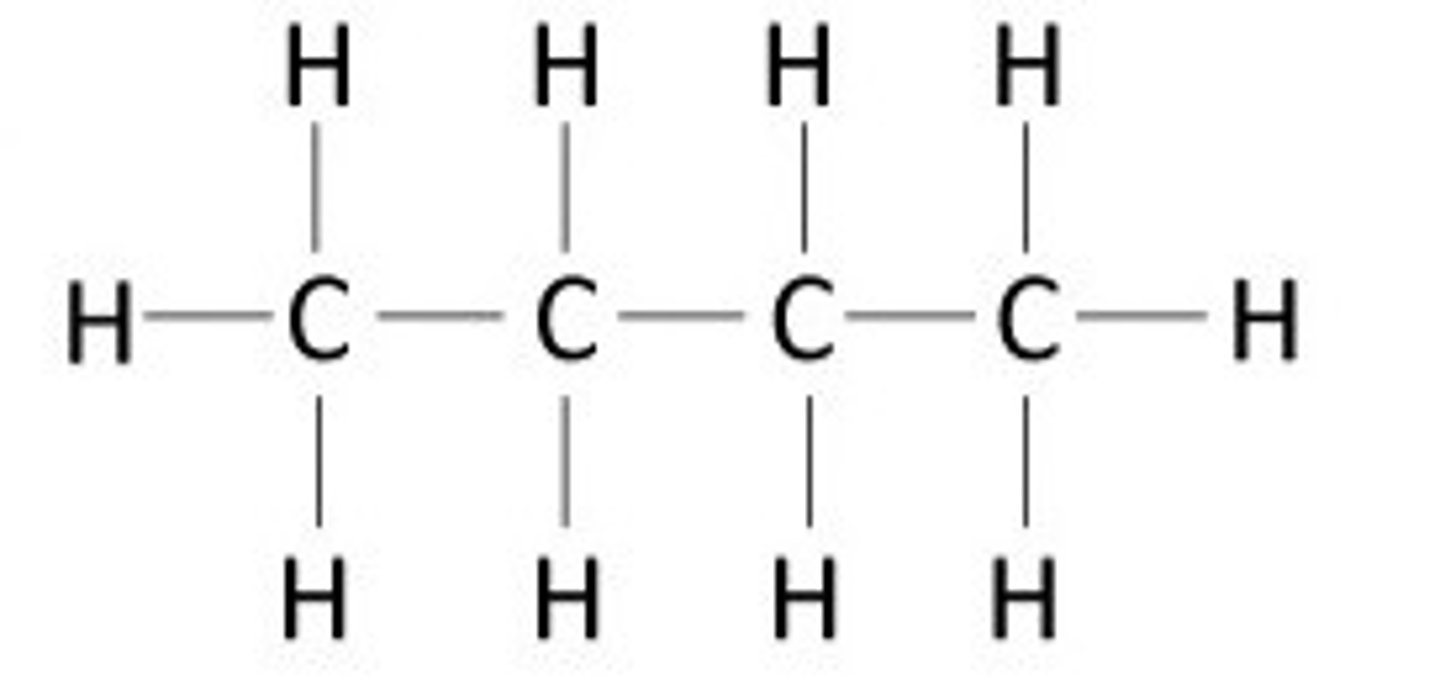
hydrocarbon
An organic molecule consisting only of carbon and hydrogen.
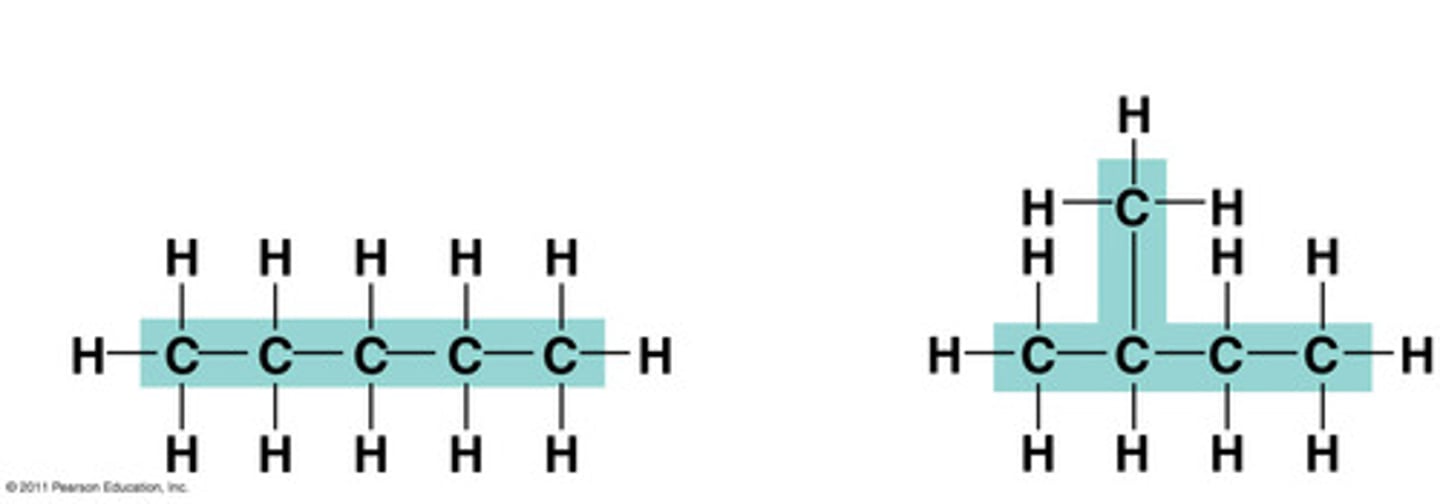
isomer
One of several compounds with the same molecular formula but different structures and therefore different properties. The three types are structural, geometric, and enantiomers.
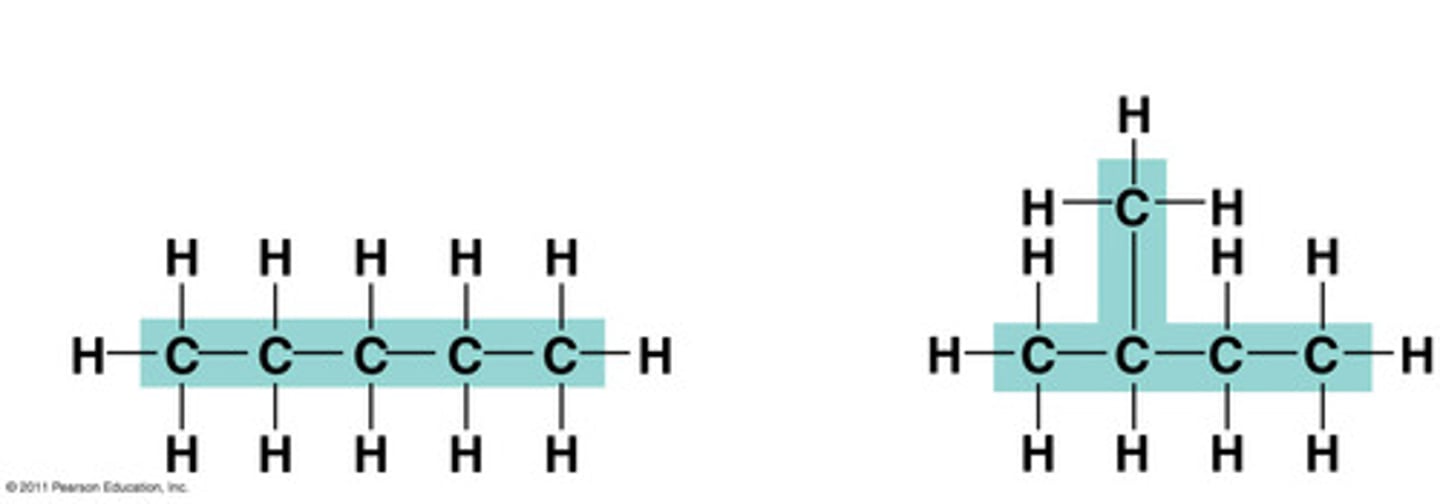
structural isomer
One or several compounds that have the same molecular formula but differ in the covalent arrangement of their atoms. (DIFFERENT SHAPE)
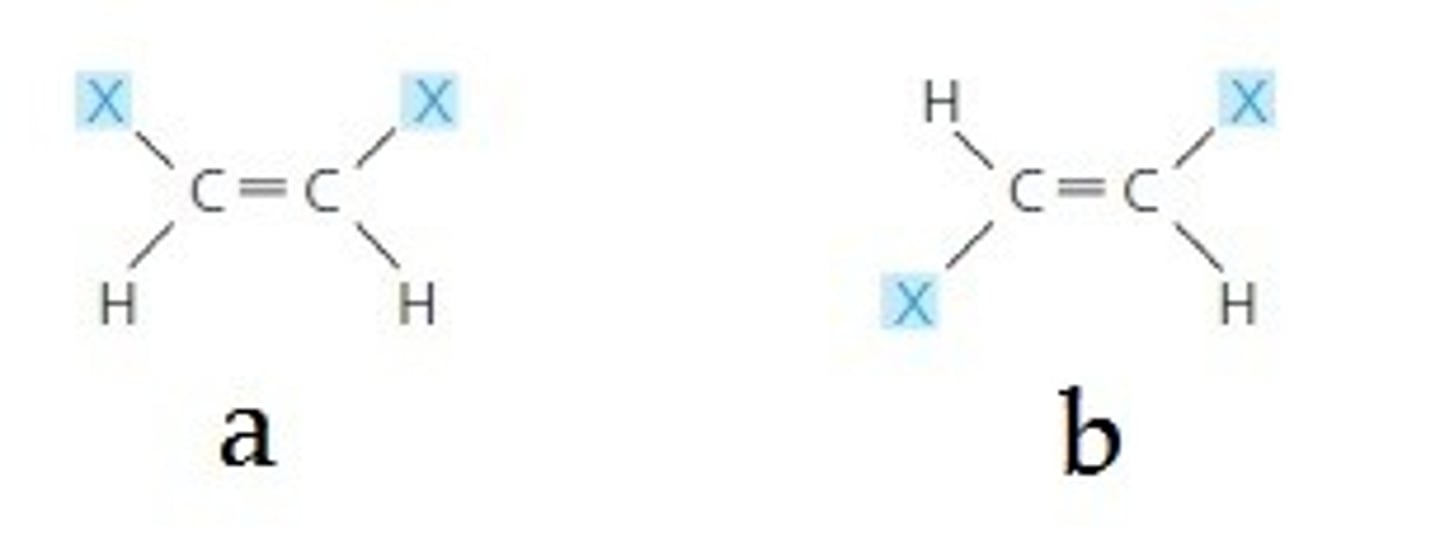
geometric isomer (cis/trans)
One of several compounds that have the same molecular formula and covalent bonds between atoms but differ in the spatial arrangements of their atoms owing to the inflexibility of double bonds (DIFFERENT ARRANGEMENT)
enantiomer
One or two compounds that are mirror images of each other and that differ in shape due to the presence of an asymmetric carbon. (MIRROR IMAGE)

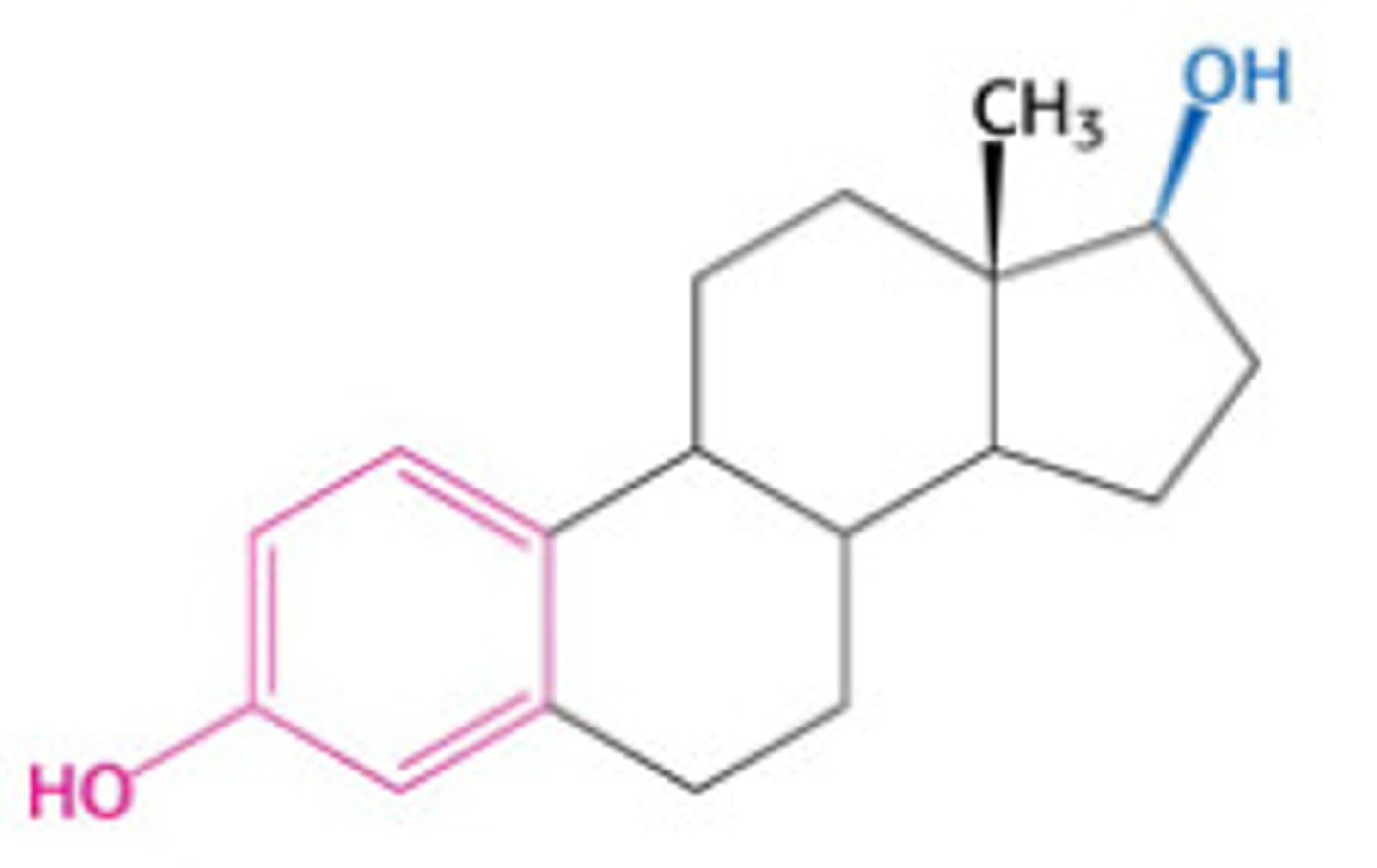
estradiol
A steroid hormone that stimulates the development and maintenance of the female reproductive system and secondary sex characteristics; the major estrogen in mammals.
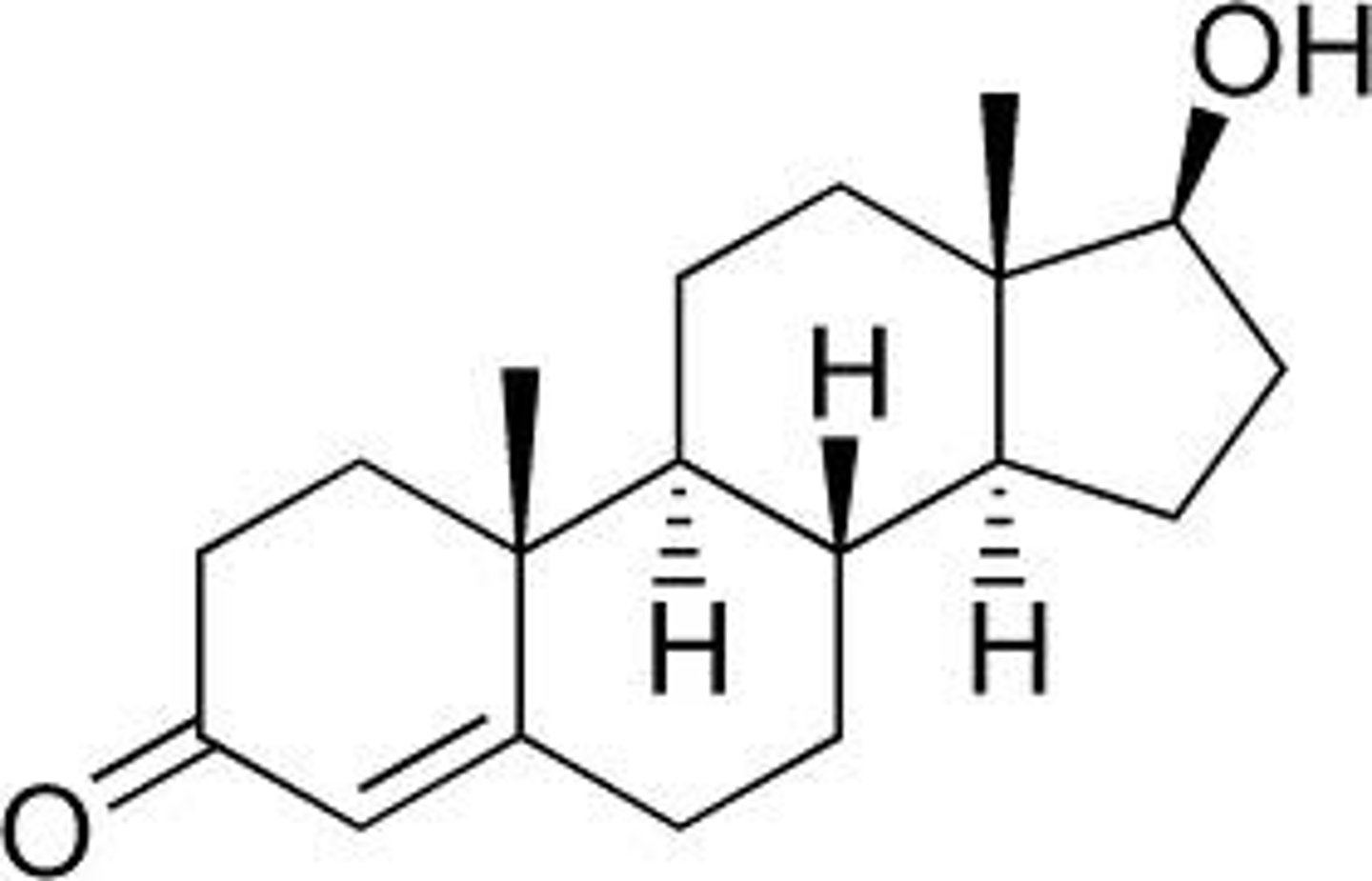
testosterone
Asteroid hormone required for development of the male reproductive system, spermatogenesis, and male secondary sex characteristics; the major androgen in mammals.
functional groups
A specific configuration of atoms commonly attached to the carbon skeletons of organic molecules and involved in chemical reactions.
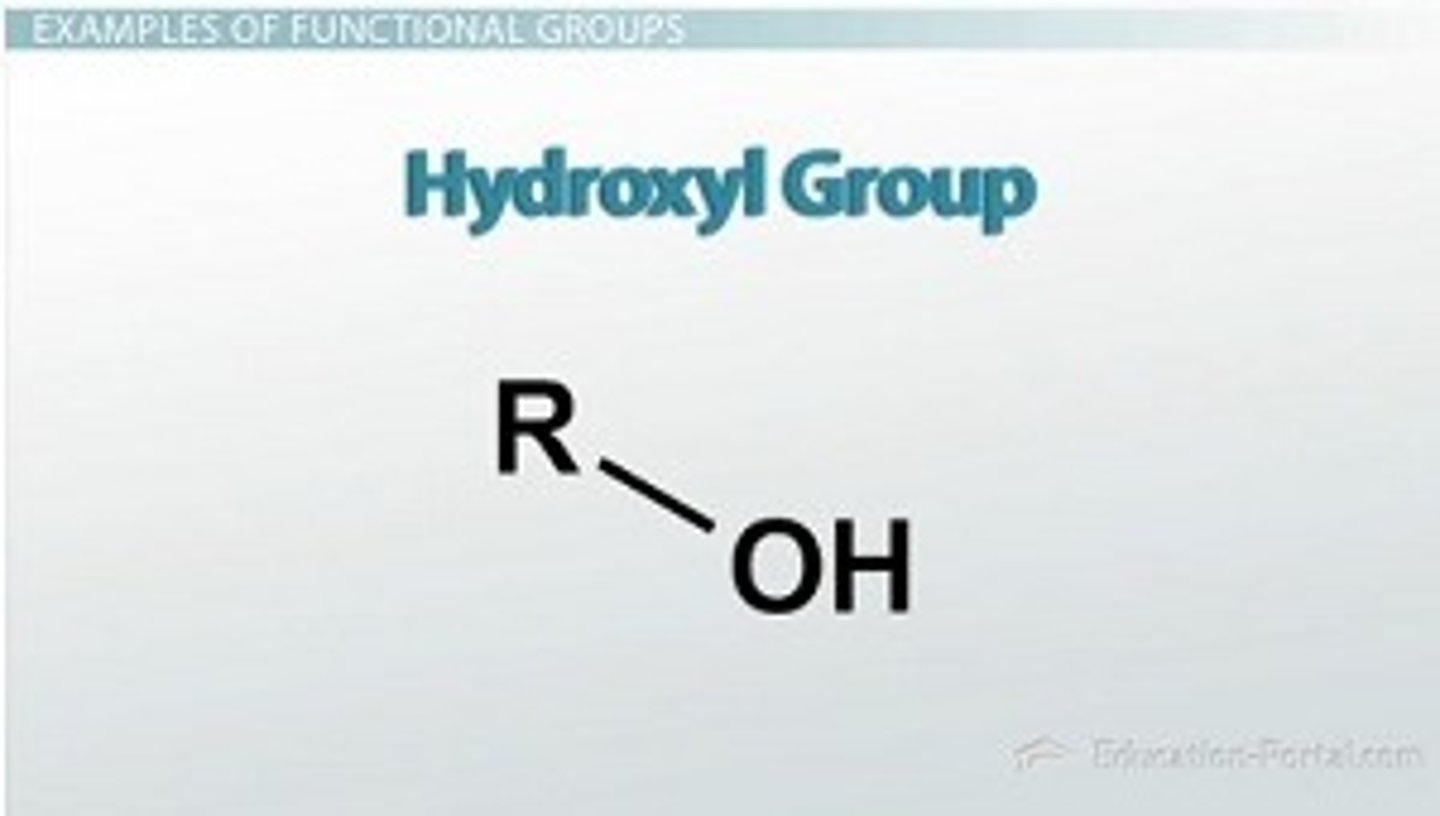
hydroxyl group
A chemical group consisting of an oxygen atom joined to a hydrogen atom. Molecules possessing this group are soluble in water and are called alcohols.
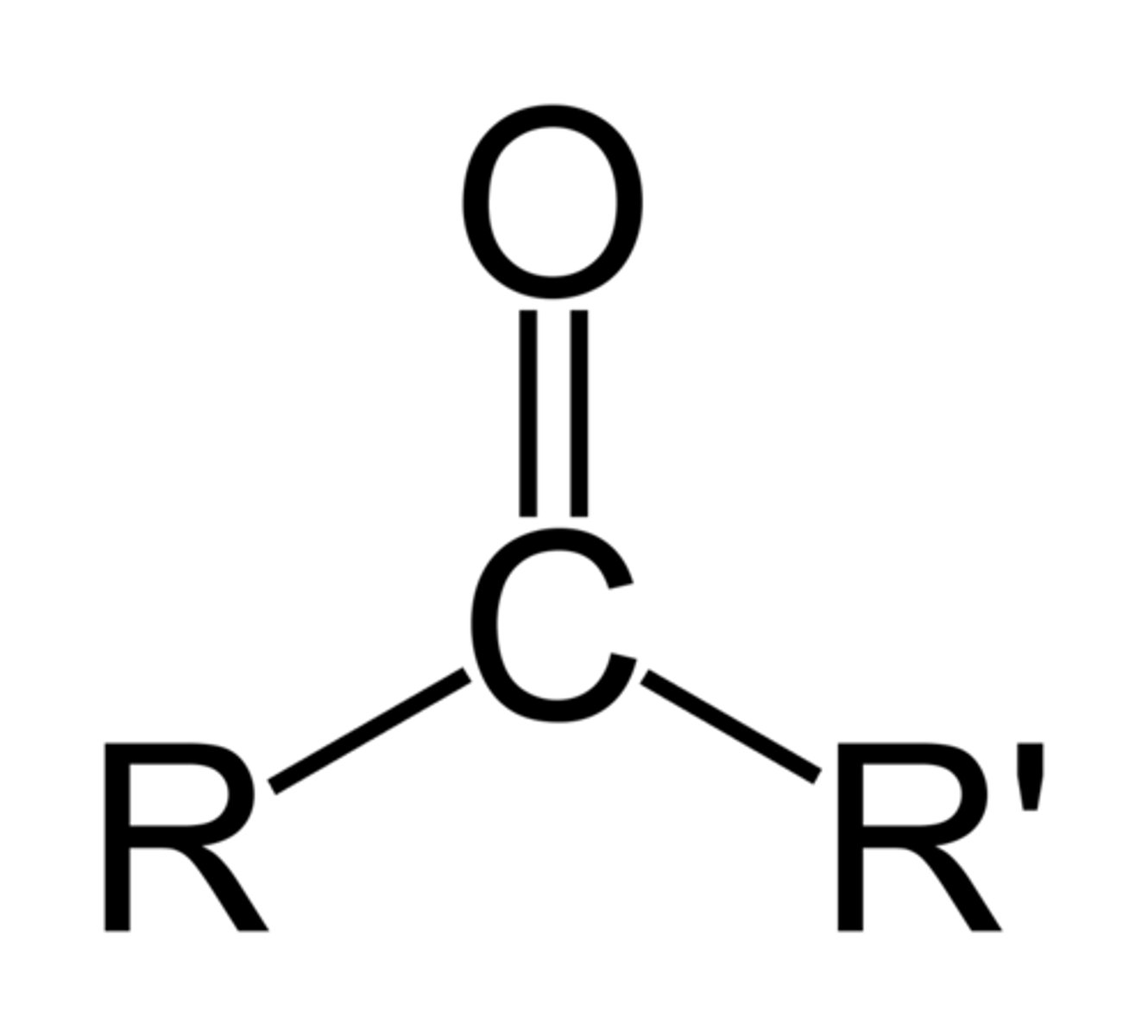
ketone group
A chemical group consisting of a carbon double bonded to oxygen (must be in middle of chain)
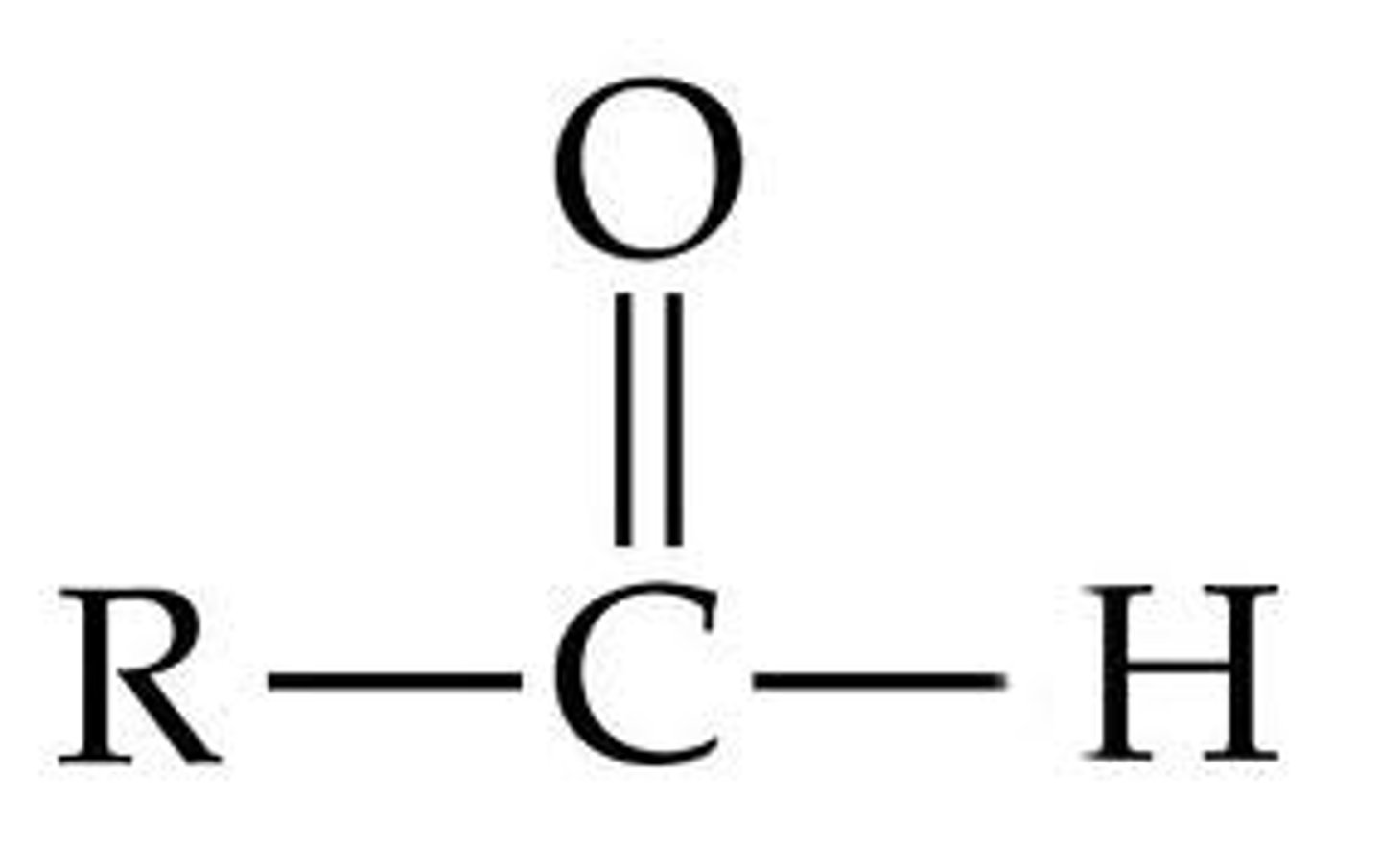
aldehyde group
A chemical group consisting of a carbon double bonded to oxygen and single bonded to hydrogen (must be at end of chain)
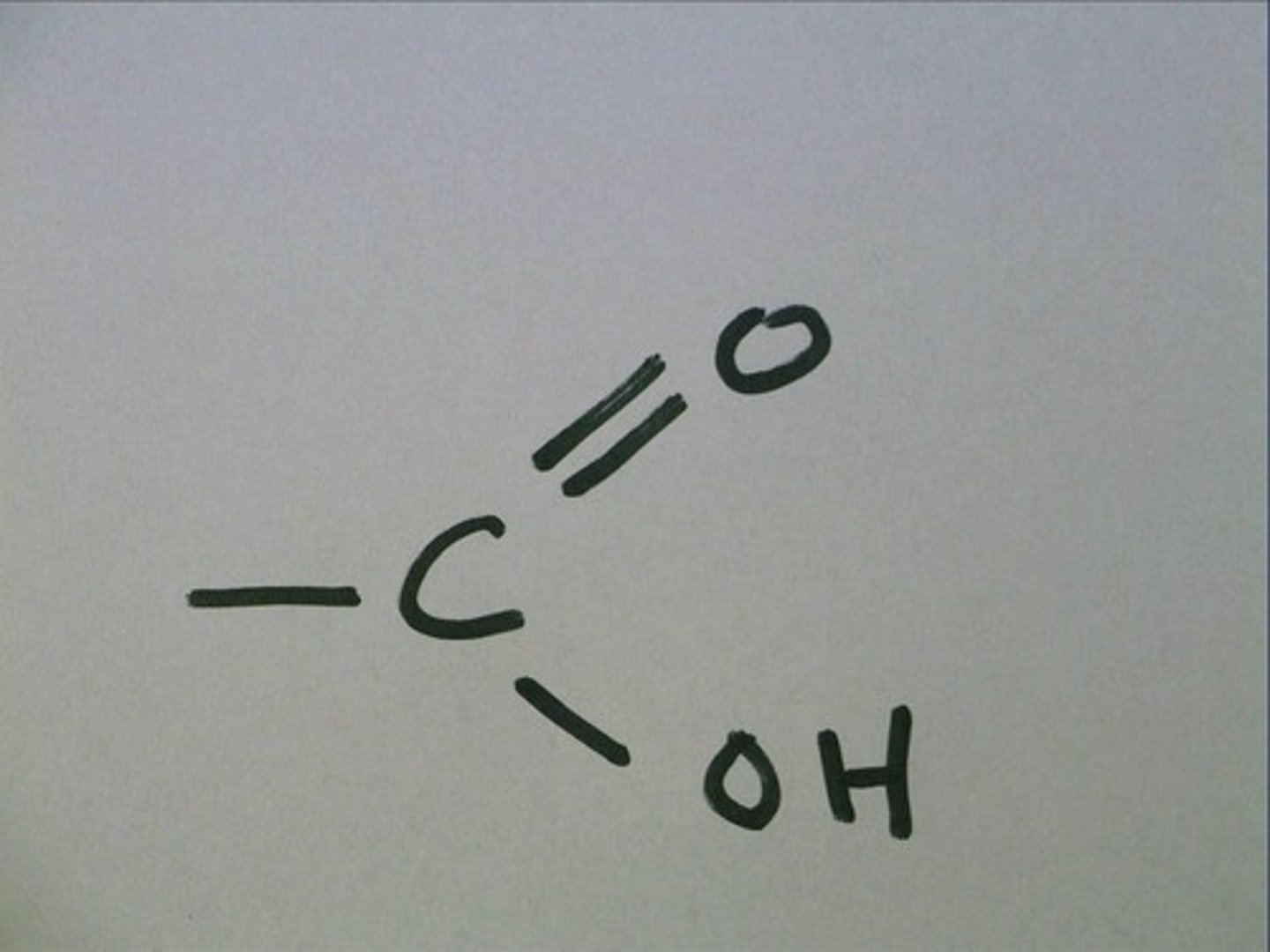
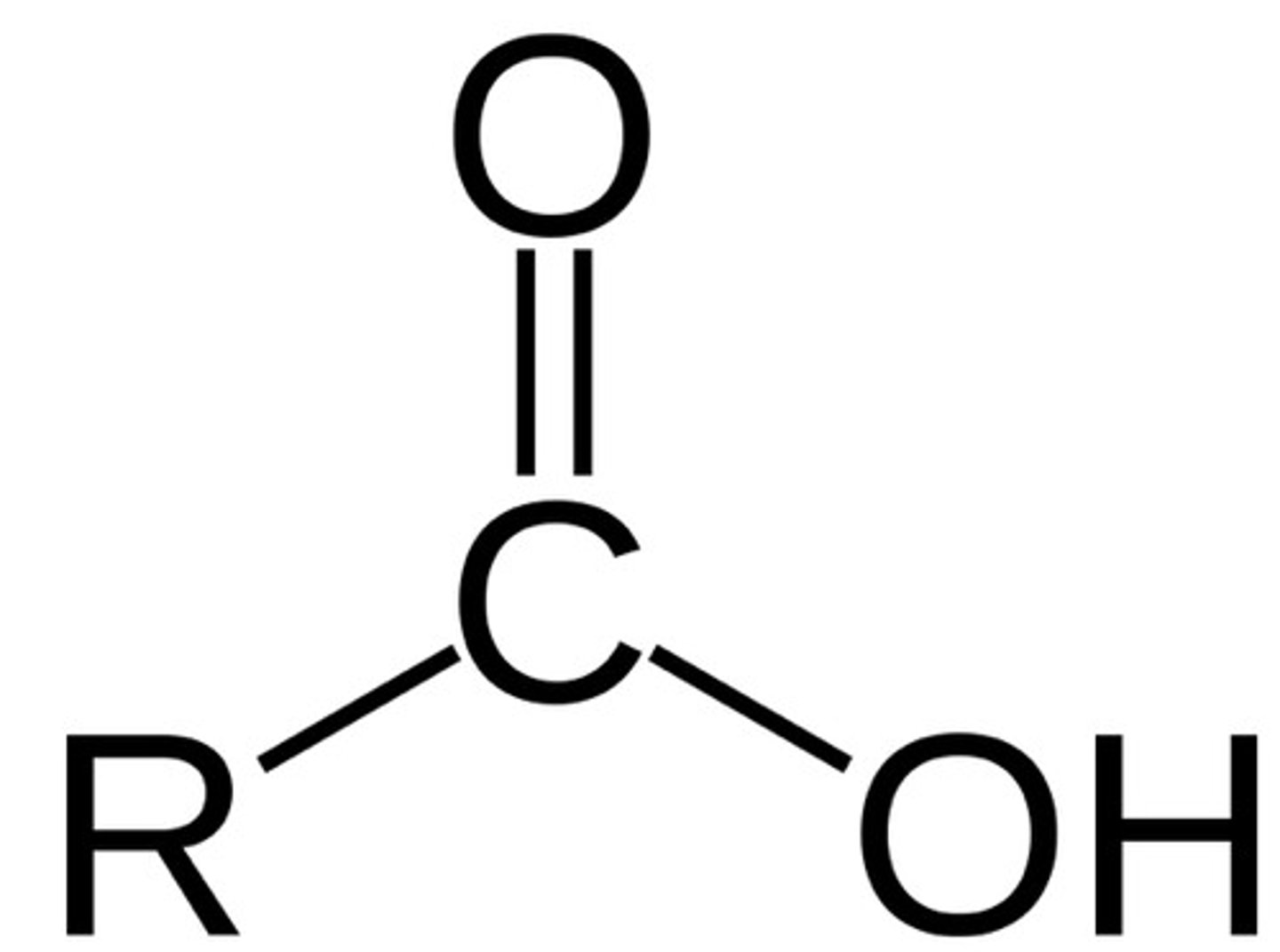
carboxyl group
A chemical group consisting of a single carbon atom double-bonded to an oxygen atom and also bonded to a hydroxyl group
ionized carboxyl group
A chemical group consisting of a single carbon atom double-bonded to an oxygen atom and also bonded to a negatively charged oxygen
amino group
A chemical group consisting of a nitrogen atom bonded to two hydrogen atoms can act as a base in solution, accepting a hydrogen ion and acquiring a charge of 1⁺
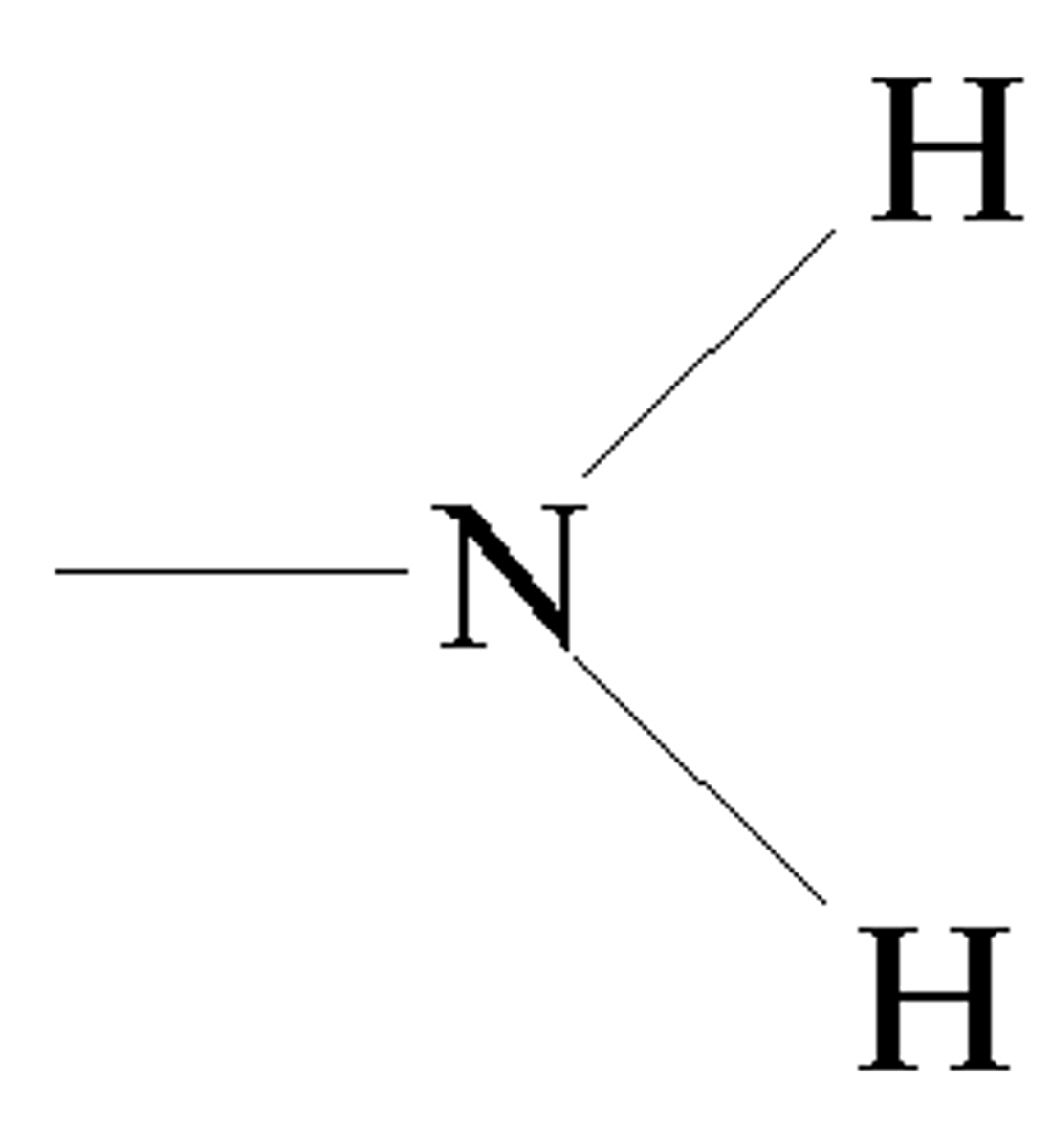
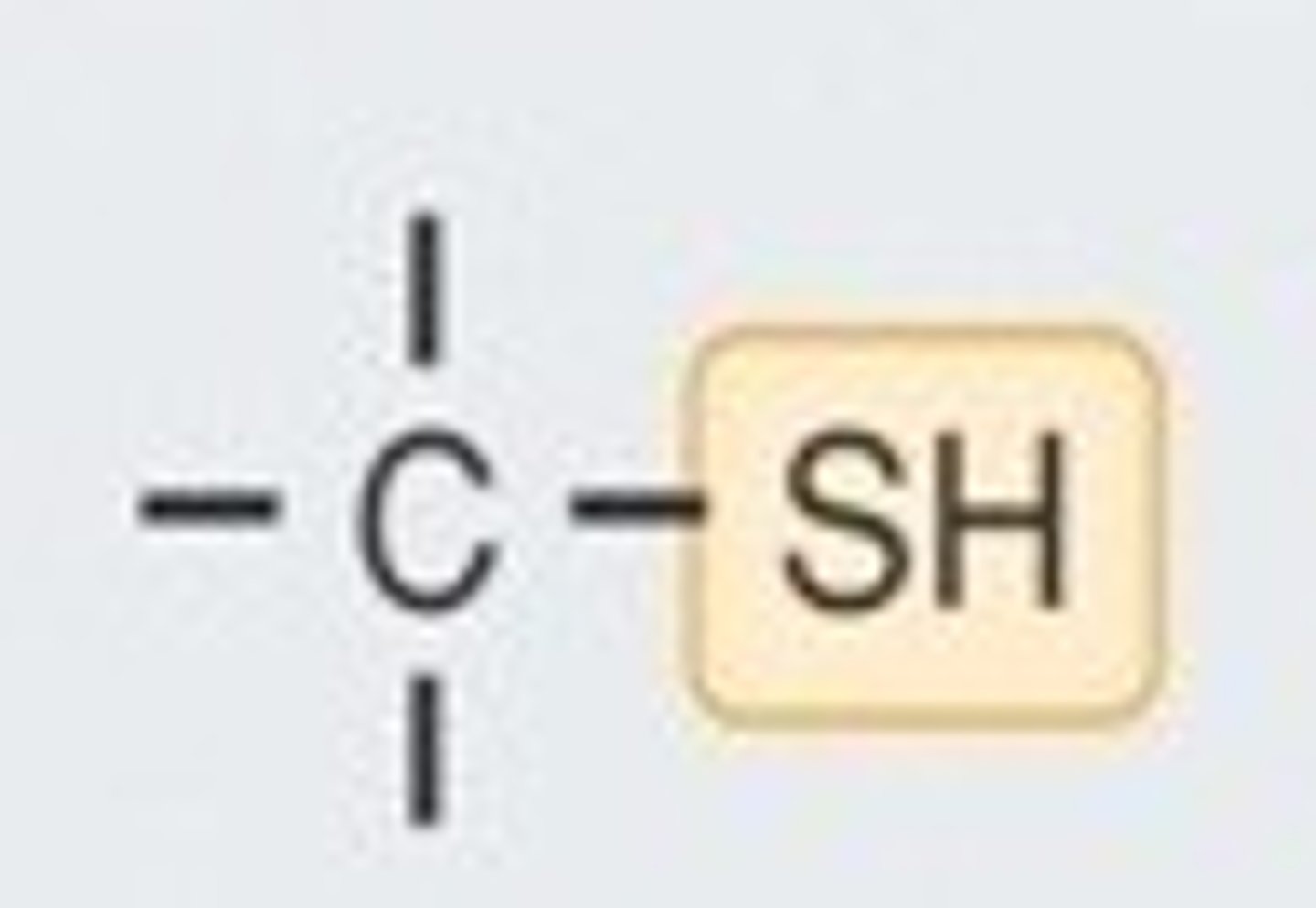
sulfhydryl group
A chemical group consisting of a sulfur atom bonded to a hydrogen atom.
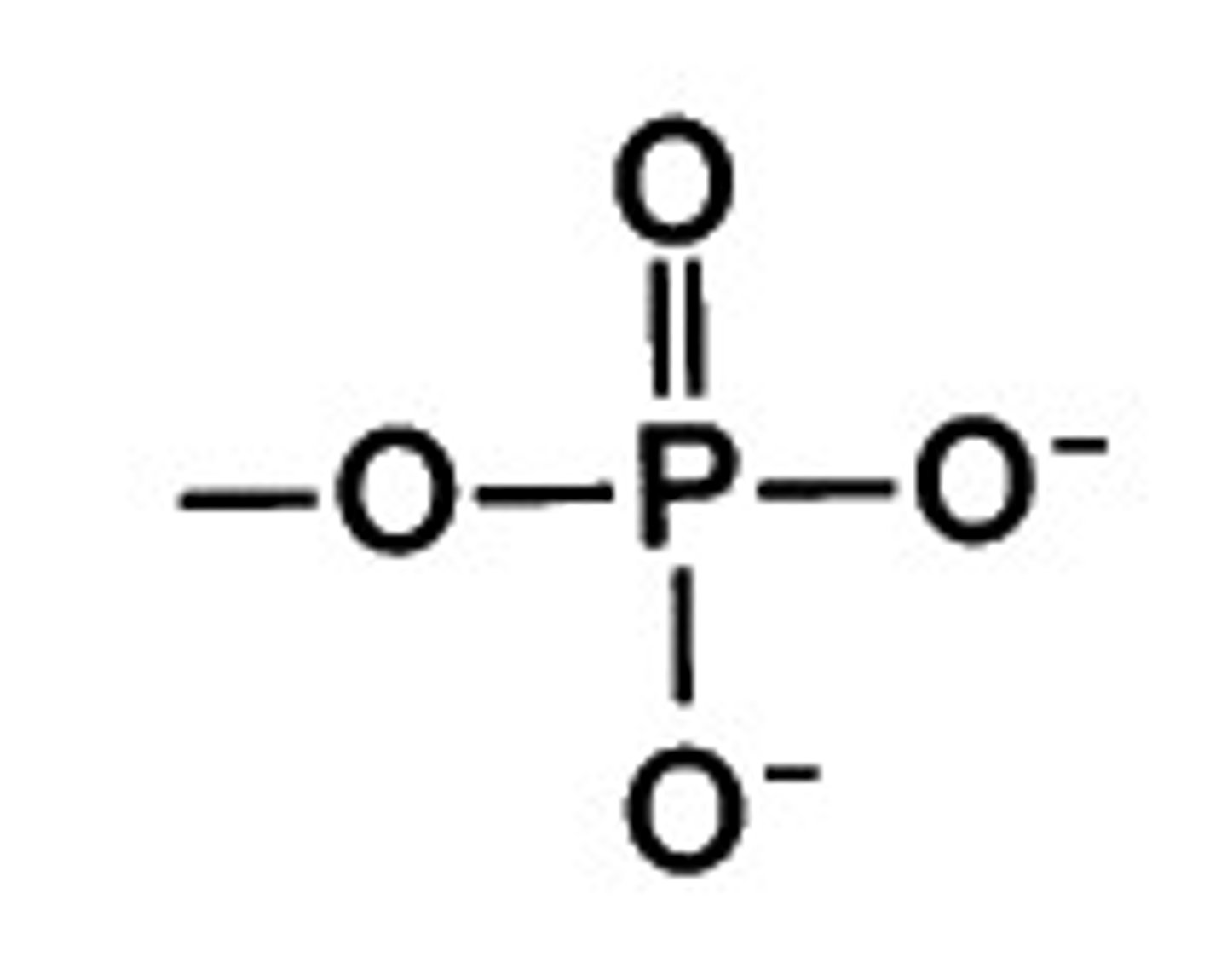
phosphate group
A chemical group consisting of a phosphate atom bonded to four oxygen atoms; important in energy transfer.
Carbohydrates, proteins, lipids, and nucleic acids
Name the 4 types of macromolecules

Metabolism
All the chemical conversions that occur within a cell
Metabolism = anabolism + catabolism
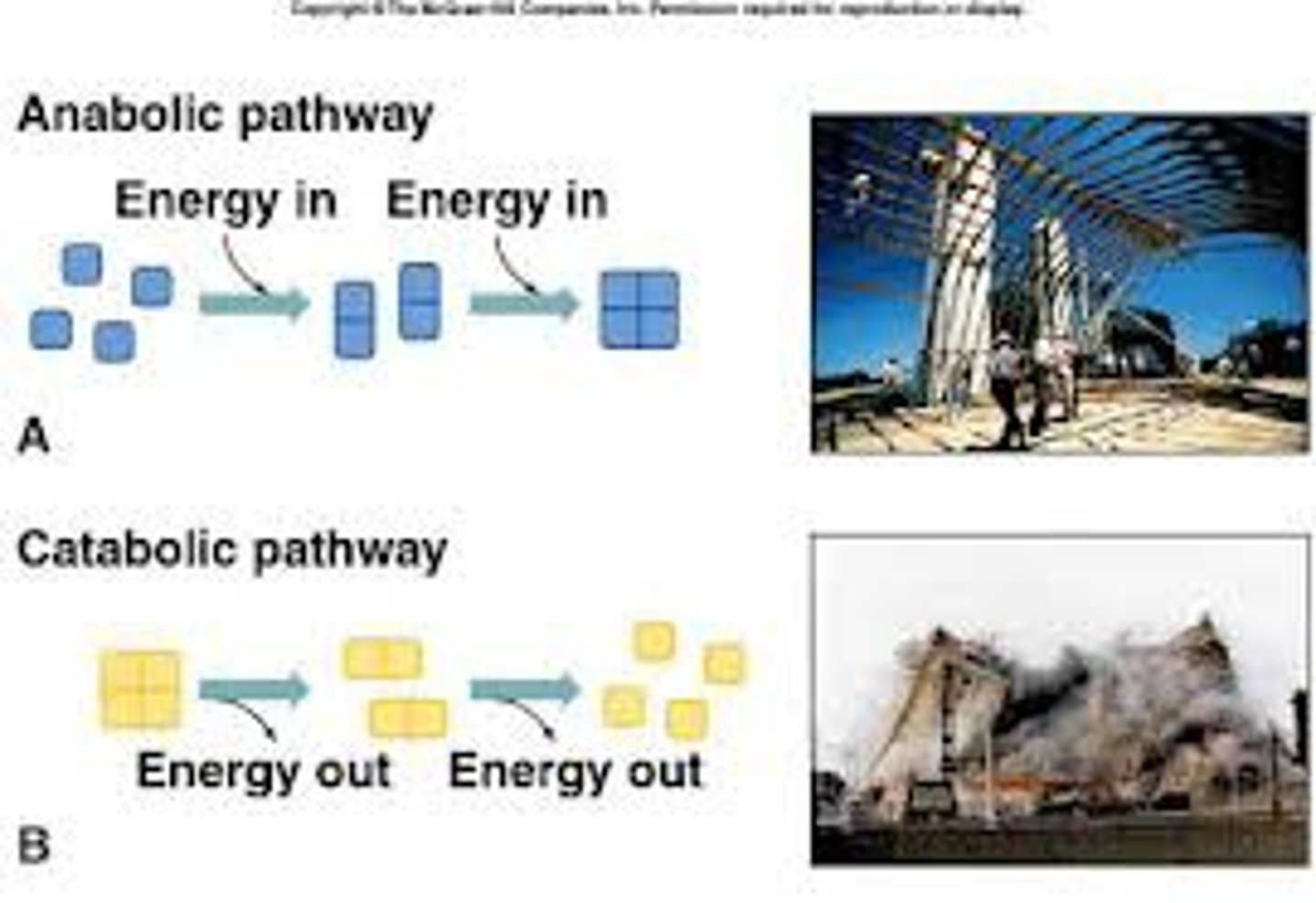
Catabolism
Conversion of complex organic molecules into smaller molecules by breaking chemical bonds

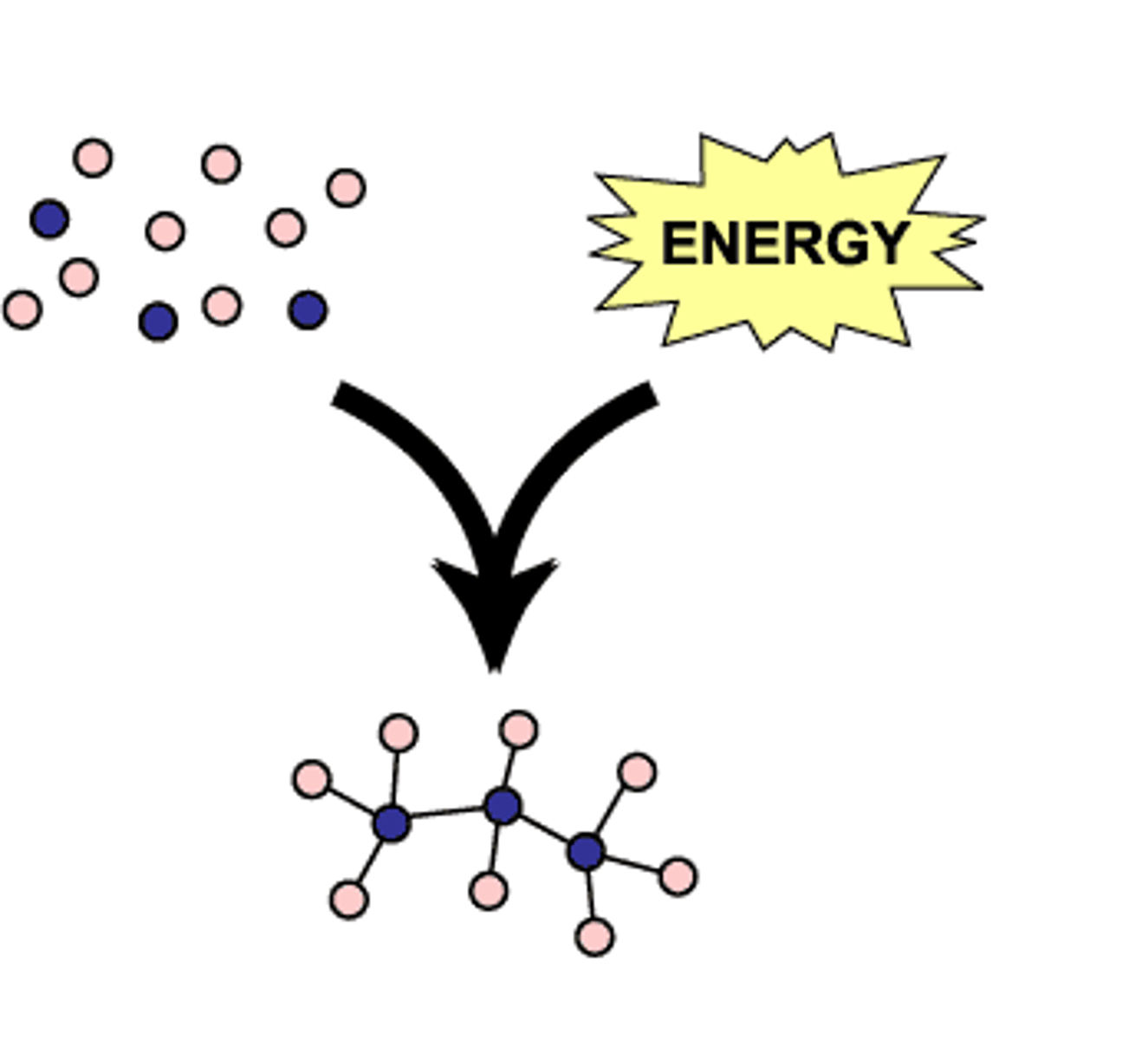
Anabolism
Conversion of small organic molecules by forming chemical bonds between smaller molecules
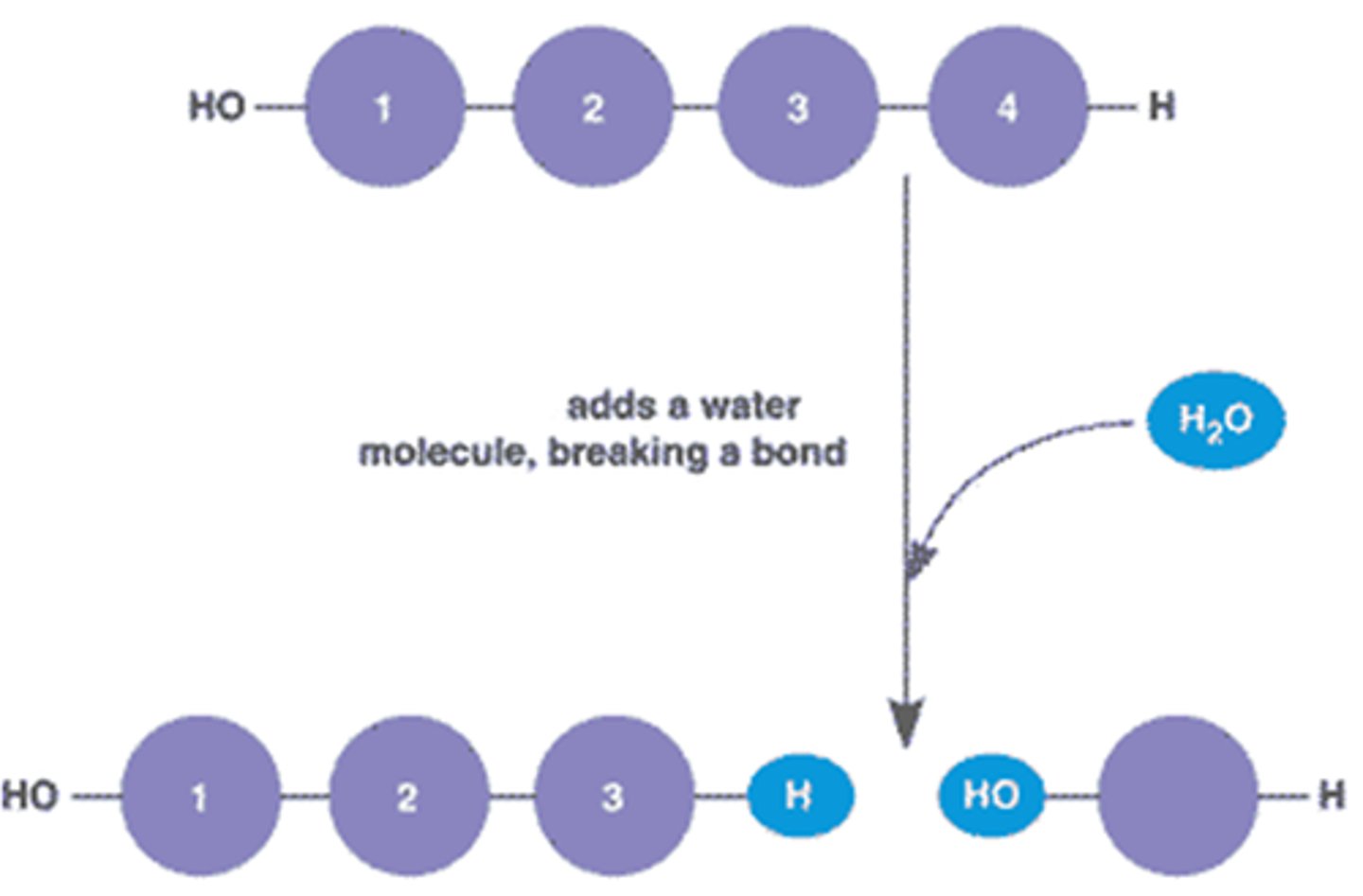
Hydrolysis
Pocess that is the reverse of dehydration synthesis. In __________, or water breakage, the bond between monomers is broken by the addition of a water molecule.
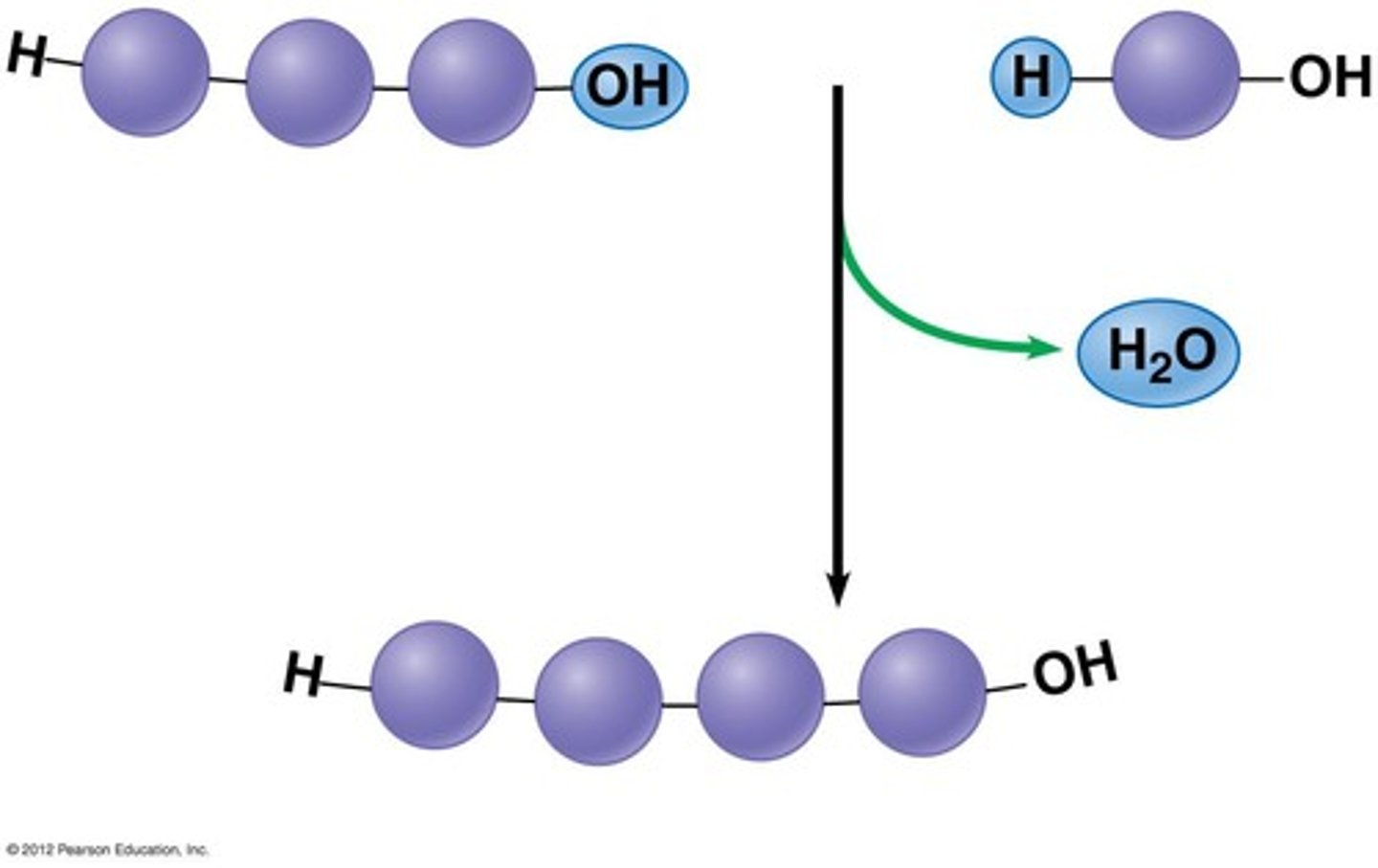
Dehydration synthesis
Reaction in which two molecules are covalently bonded, with the loss of a water molecule
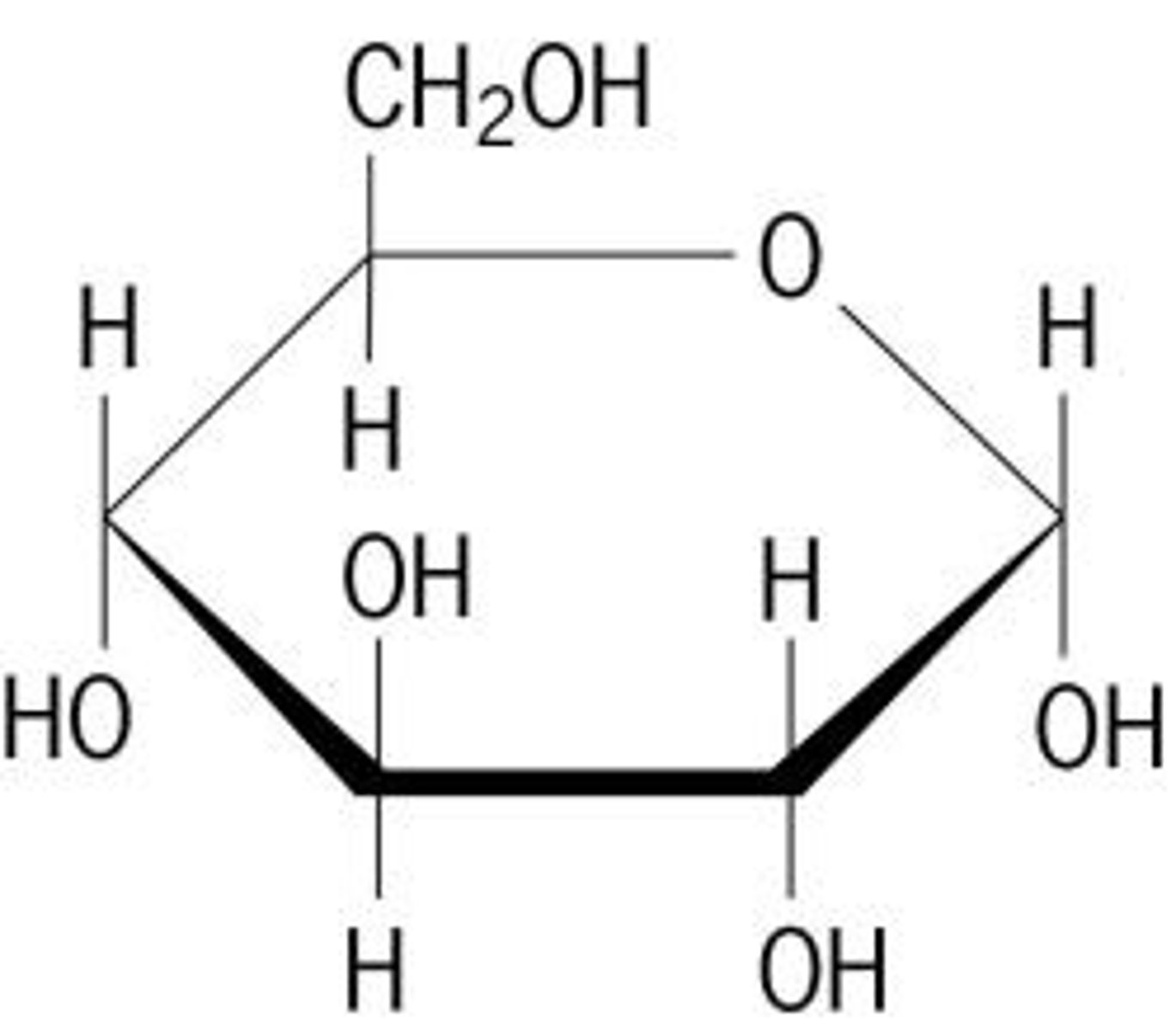
Monosaccharides
Carbohydrates are made up of _______________.
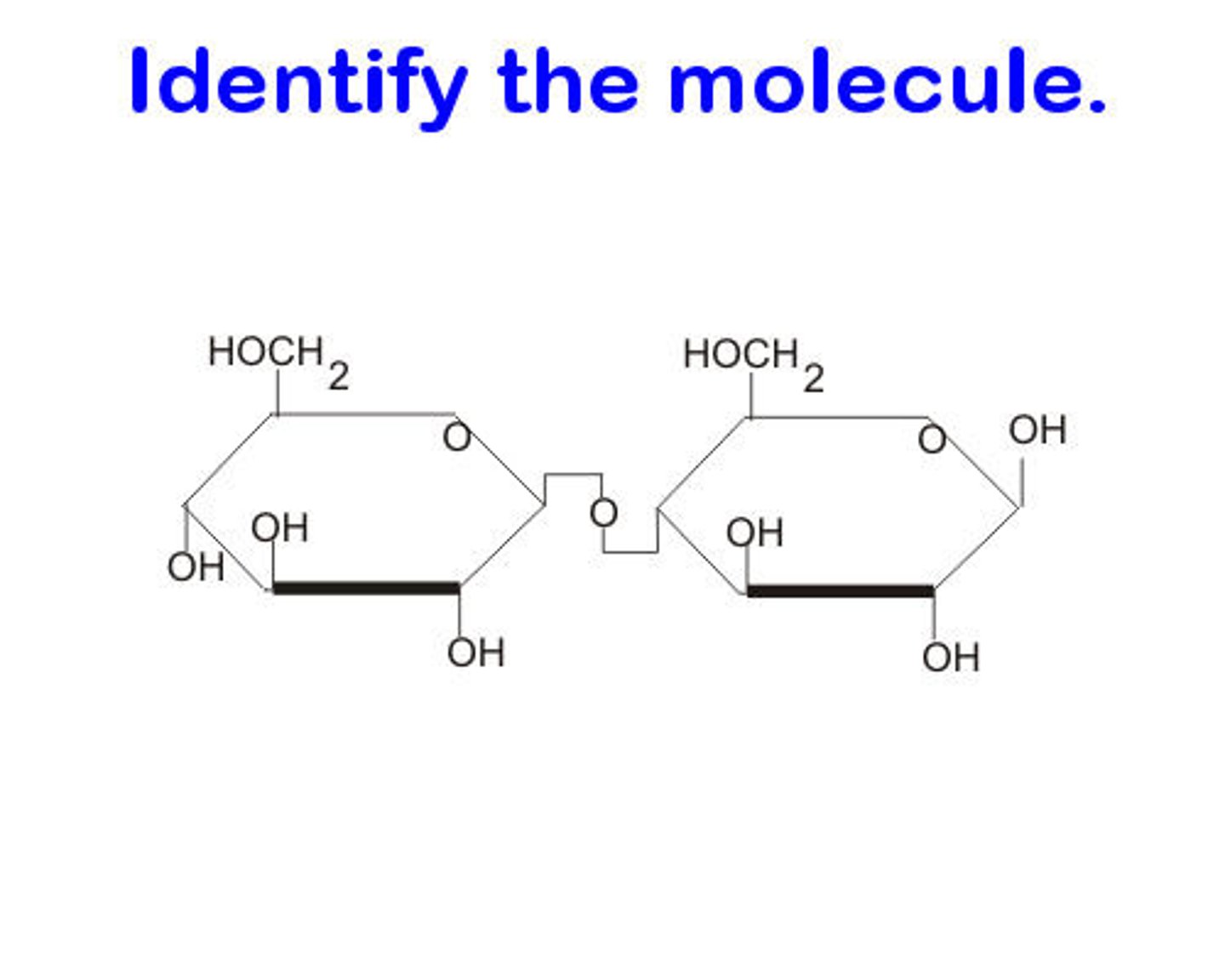
Disaccharide
Two monosaccharides covalently bonded form a ____________.
EX: Sucrose
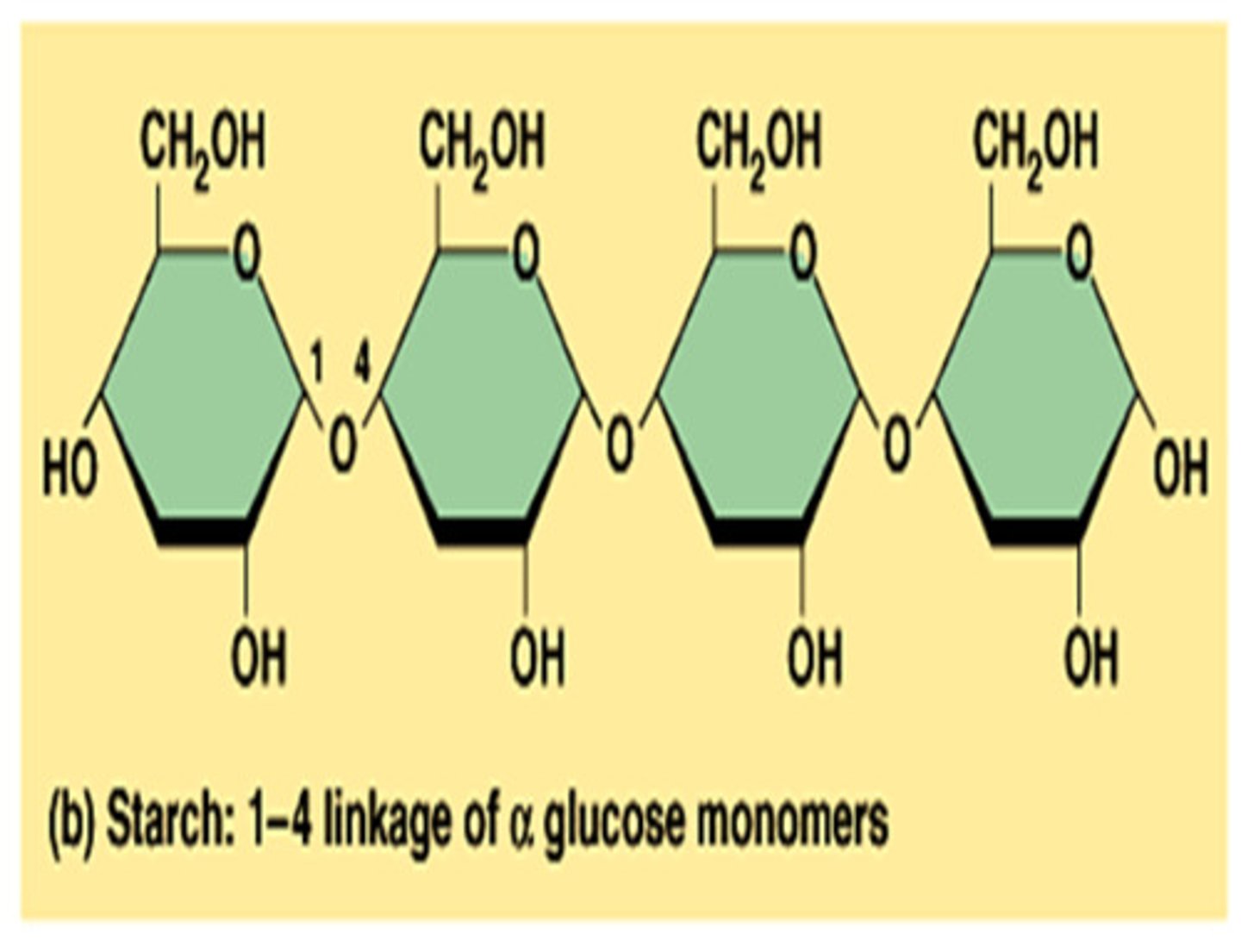
Polysaccharide
Three or more monosaccharides linked together form a ______________.
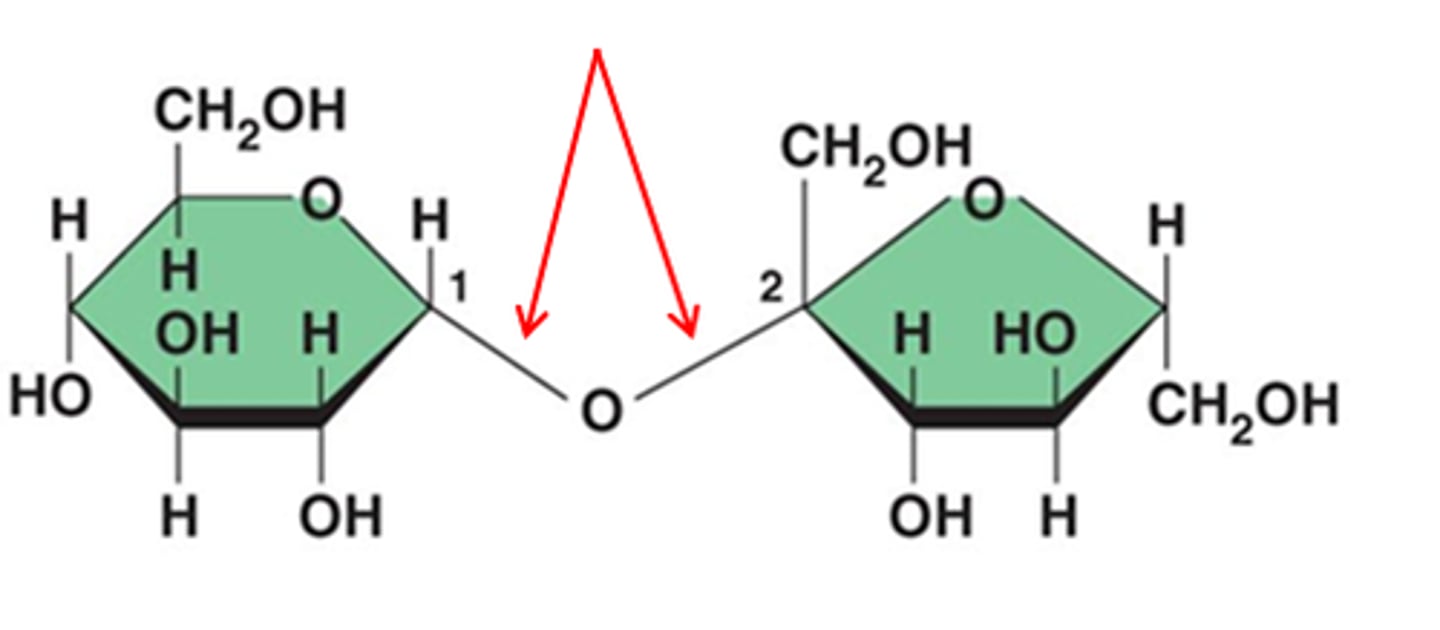
Glycosidic linkage
The bond in a carbohydrate is a(n) __________ _______.
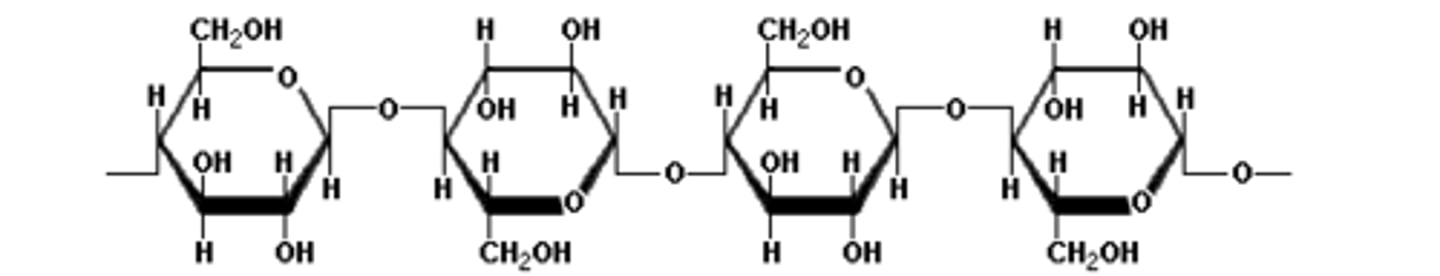
Chitin
Polysaccharide that makes up the exoskeleton pictured is called ___.

Cellulose
Polymer of glucose monomers that is found in plant cell walls
Glucose
Starch and glycogen are polymers of ________ molecules.
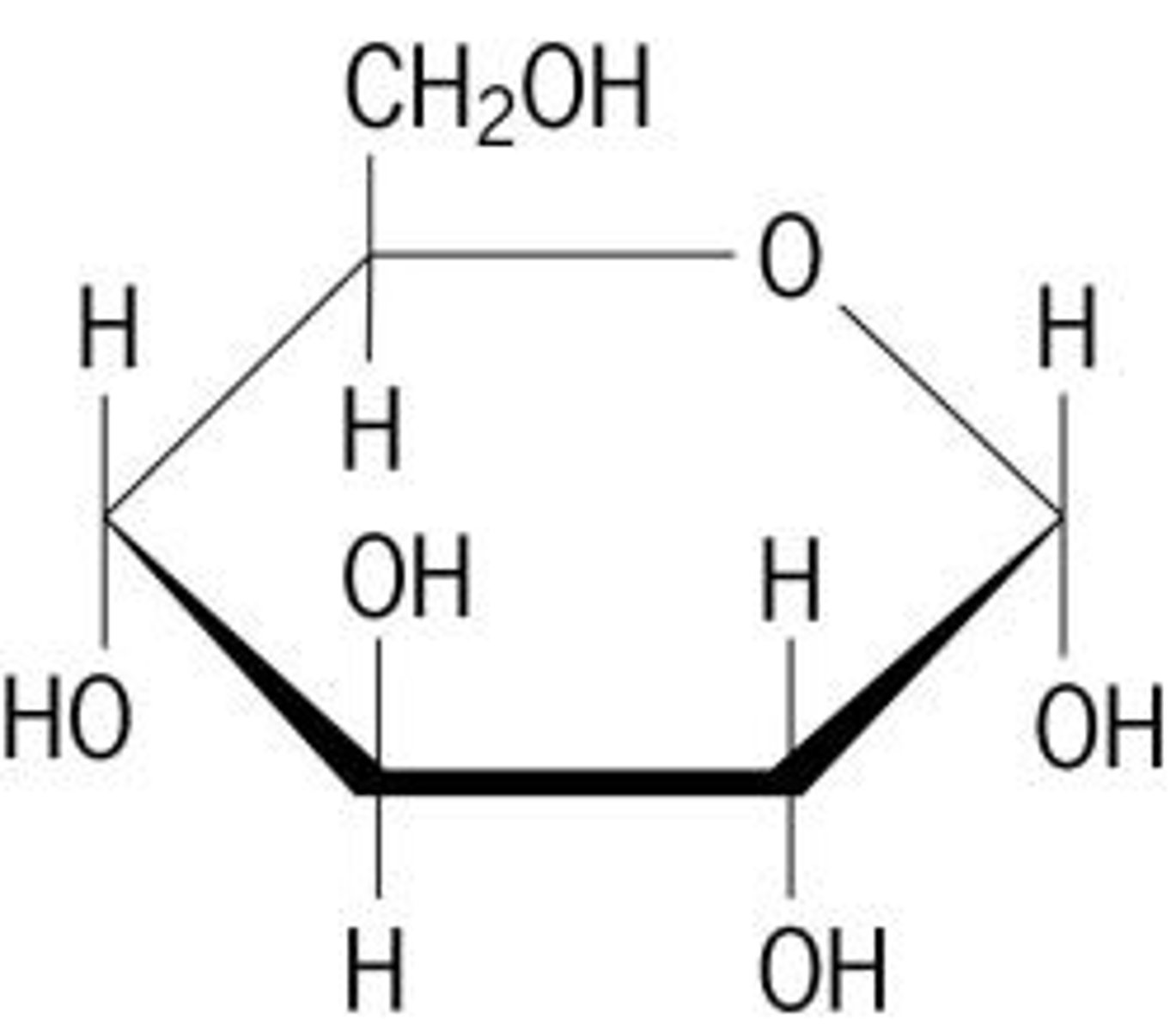
Lipids
Cushioning of organs, insulation, and energy storage are three functions of ___________.
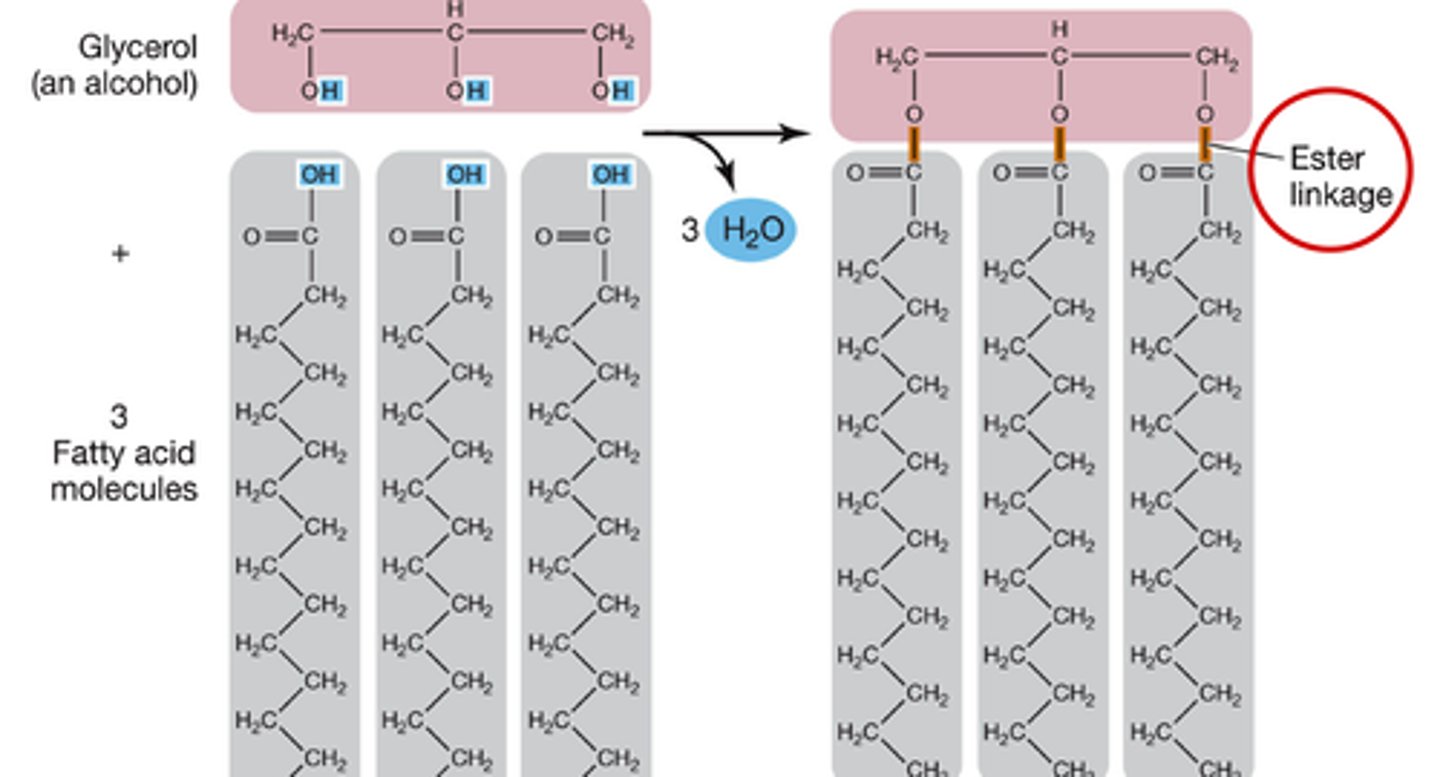
Glycerol; fatty acid chains
Triglyceride is composed of a ______ and three ____ _____ ______.
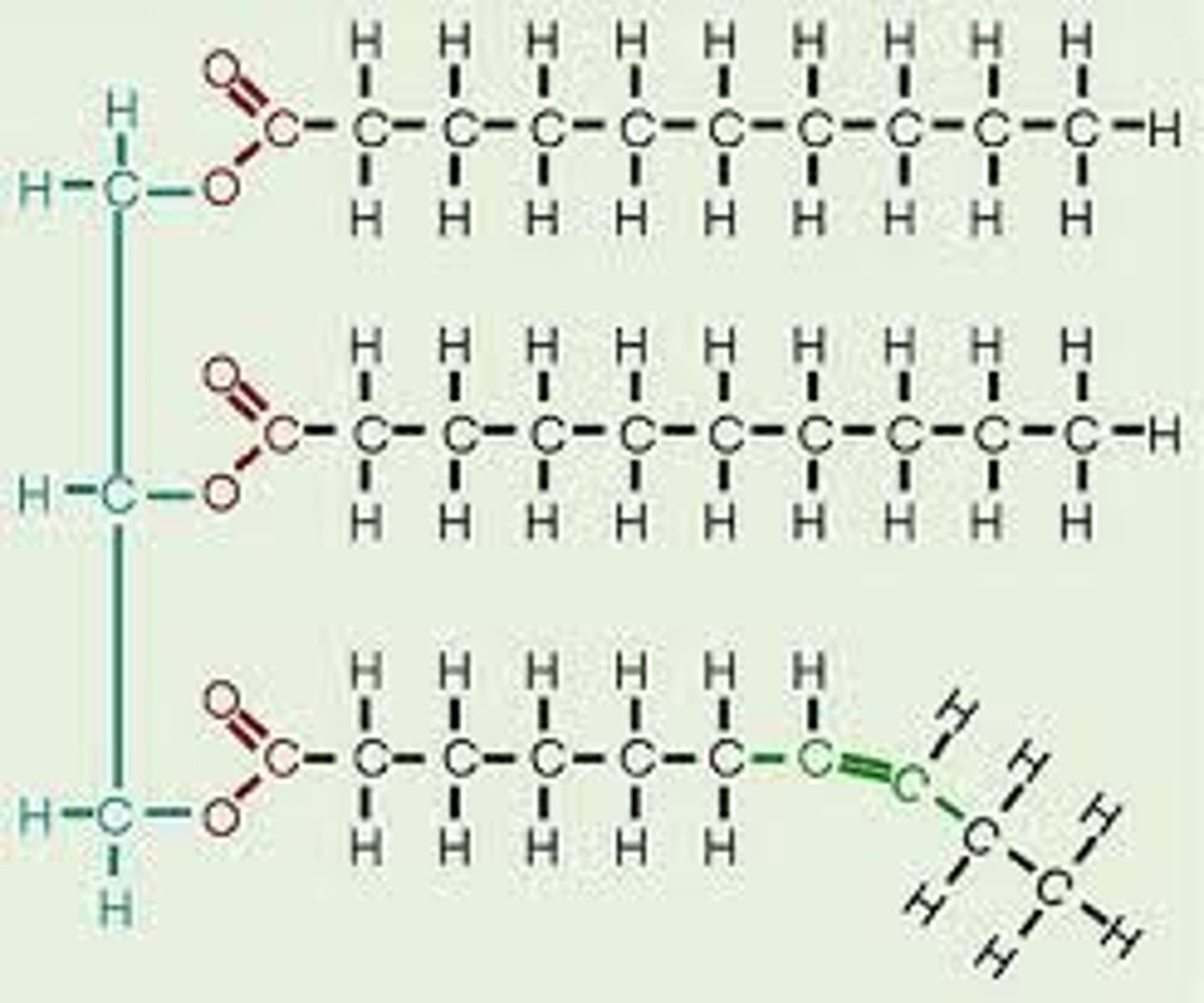
Unsaturated fats
Fats with one or more double covalent bonds, which is less likely to solidify and more flexible.
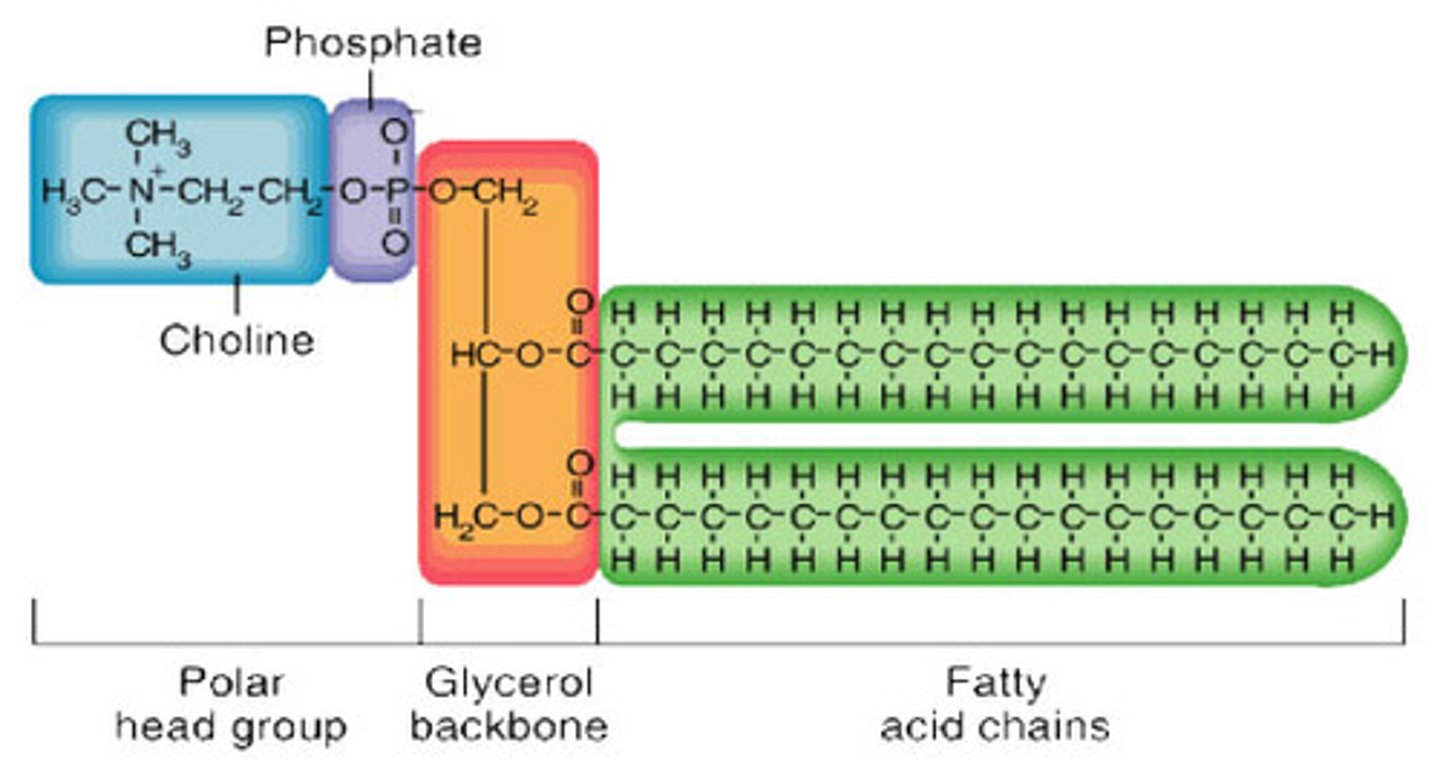
Phospholipid
Basic structural component of cell membranes, with a polar/hydrophilic head region and a nonpolar/hydrophobic tail region, giving membranes their selective permeability.
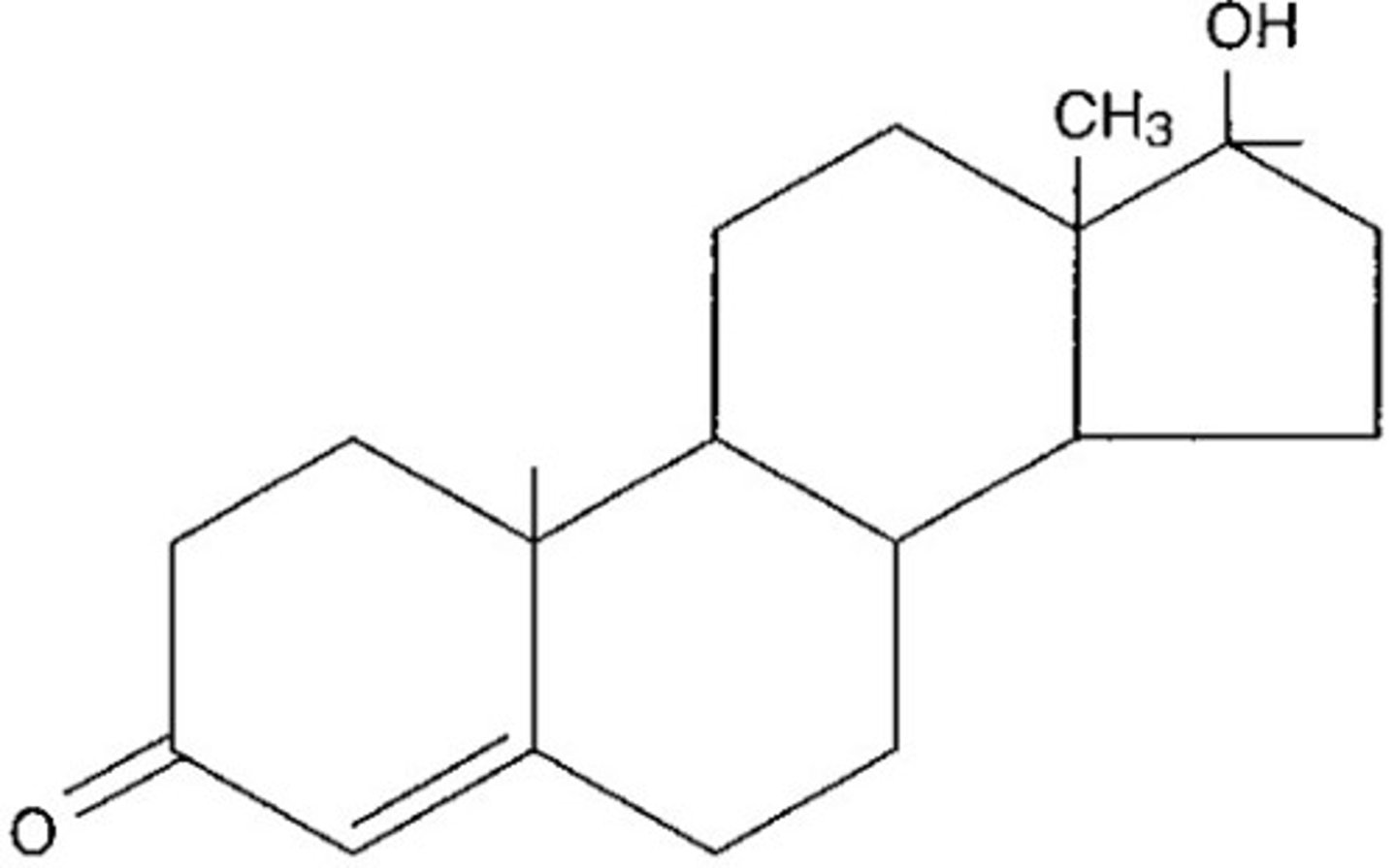
Steroid
Lipid characterized by a carbon skeleton consisting of hydrogen-fused rings.
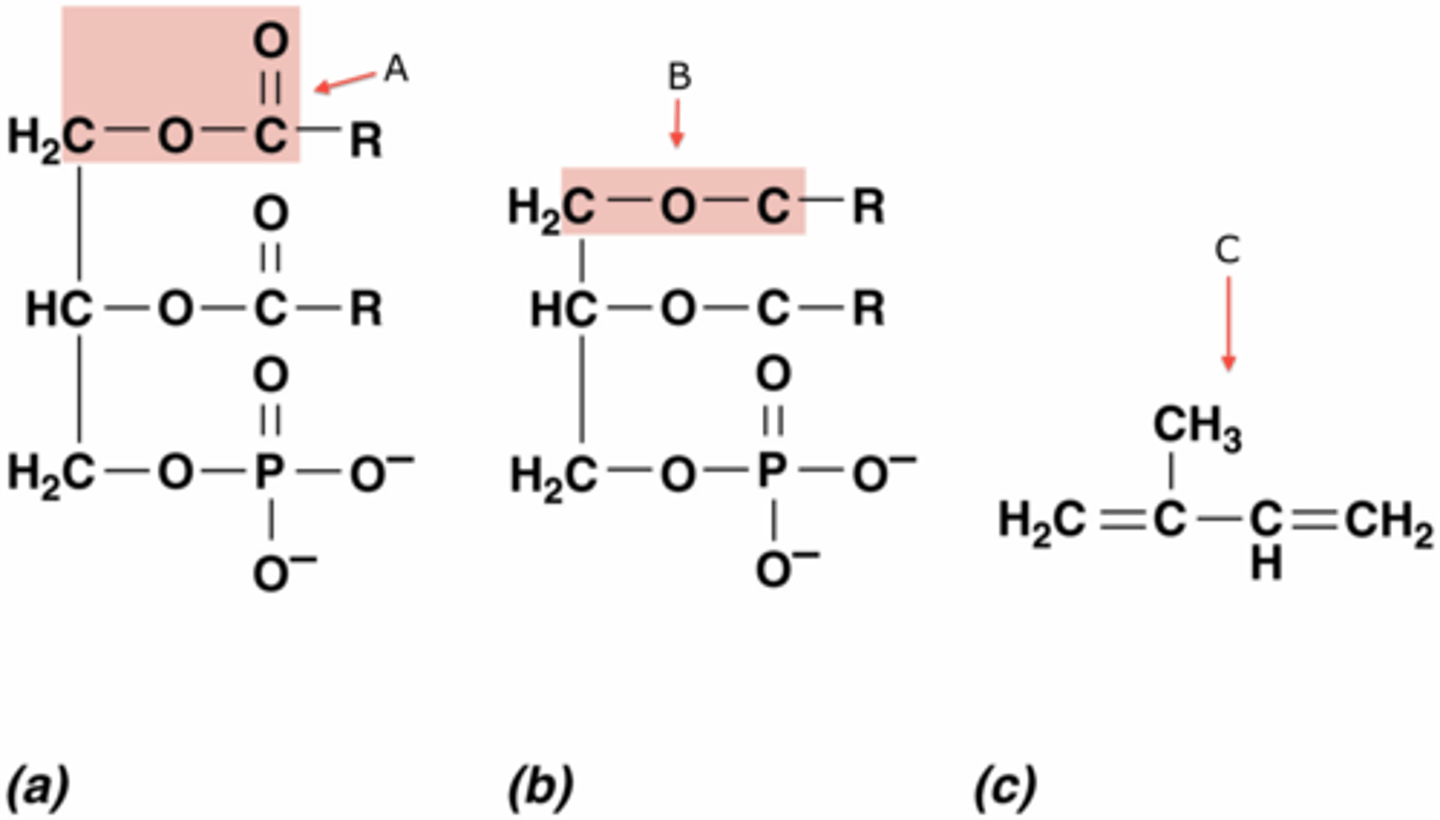
Ester linkage
In a triglyceride, the ---OH of glycerol loses a H and the ---COOH of the fatty acid chain loses a ---OH which joins to form water in a(n) _____ _______.
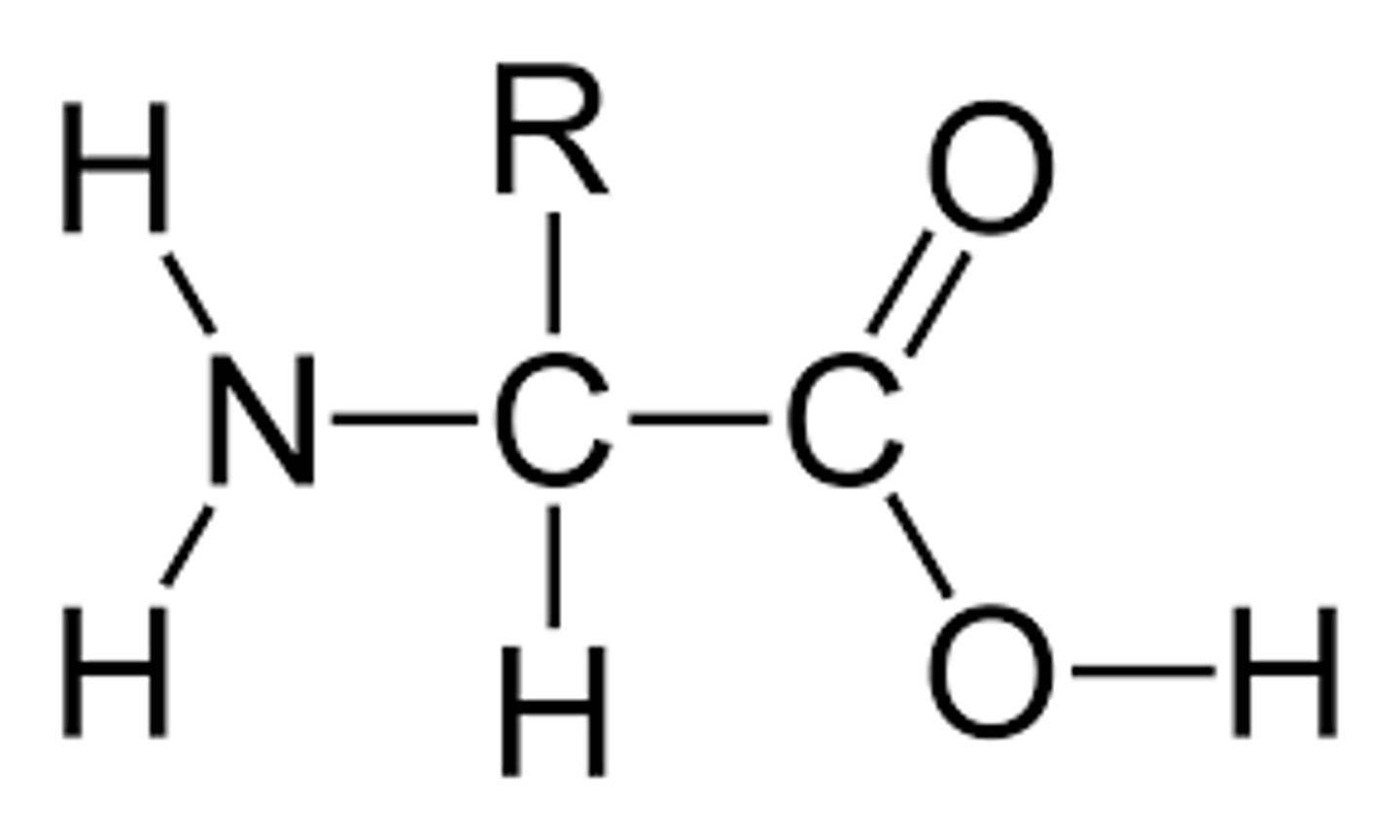
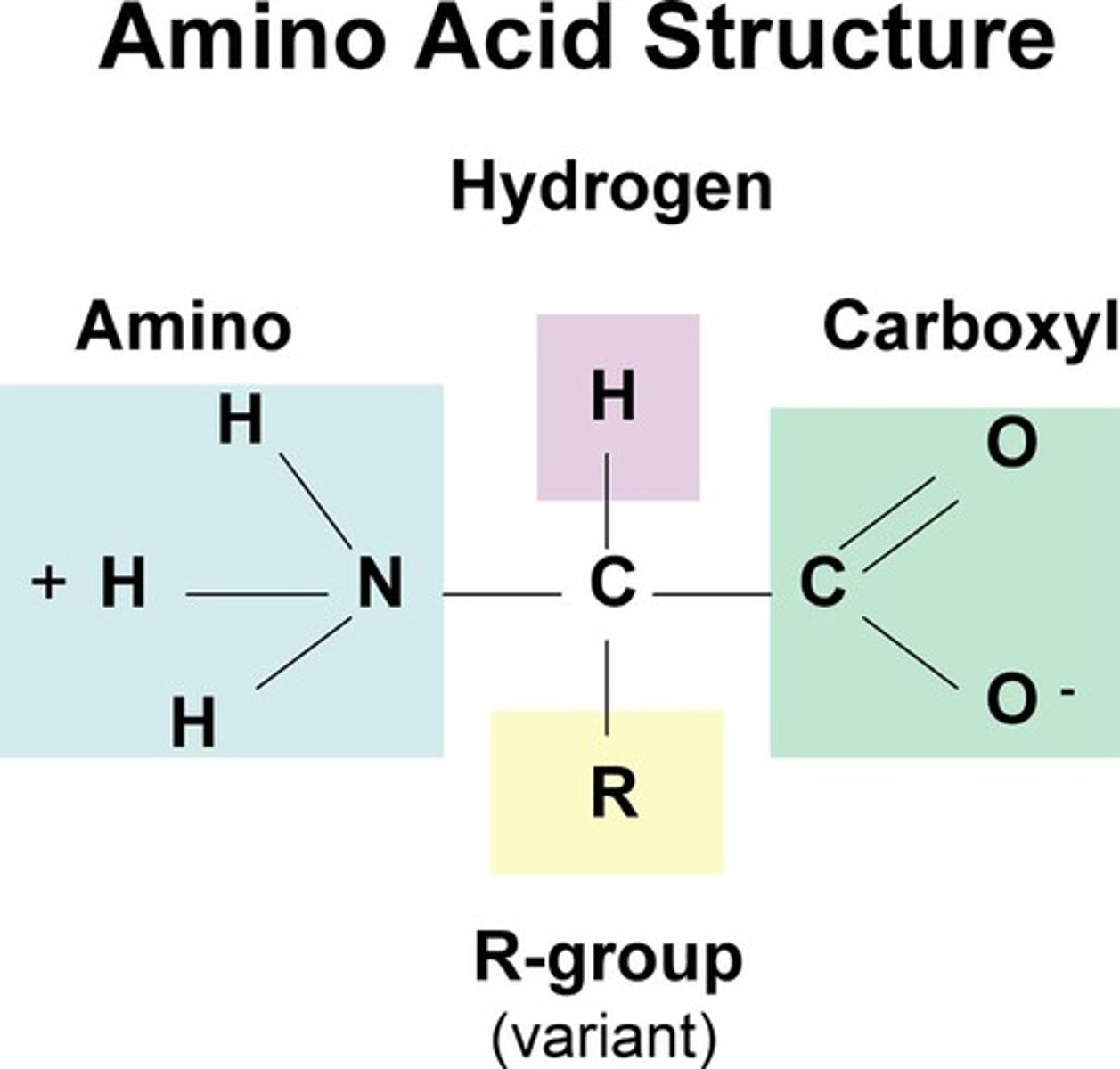
Amino acid
The monomer in a protein is a(n) _____ ____.
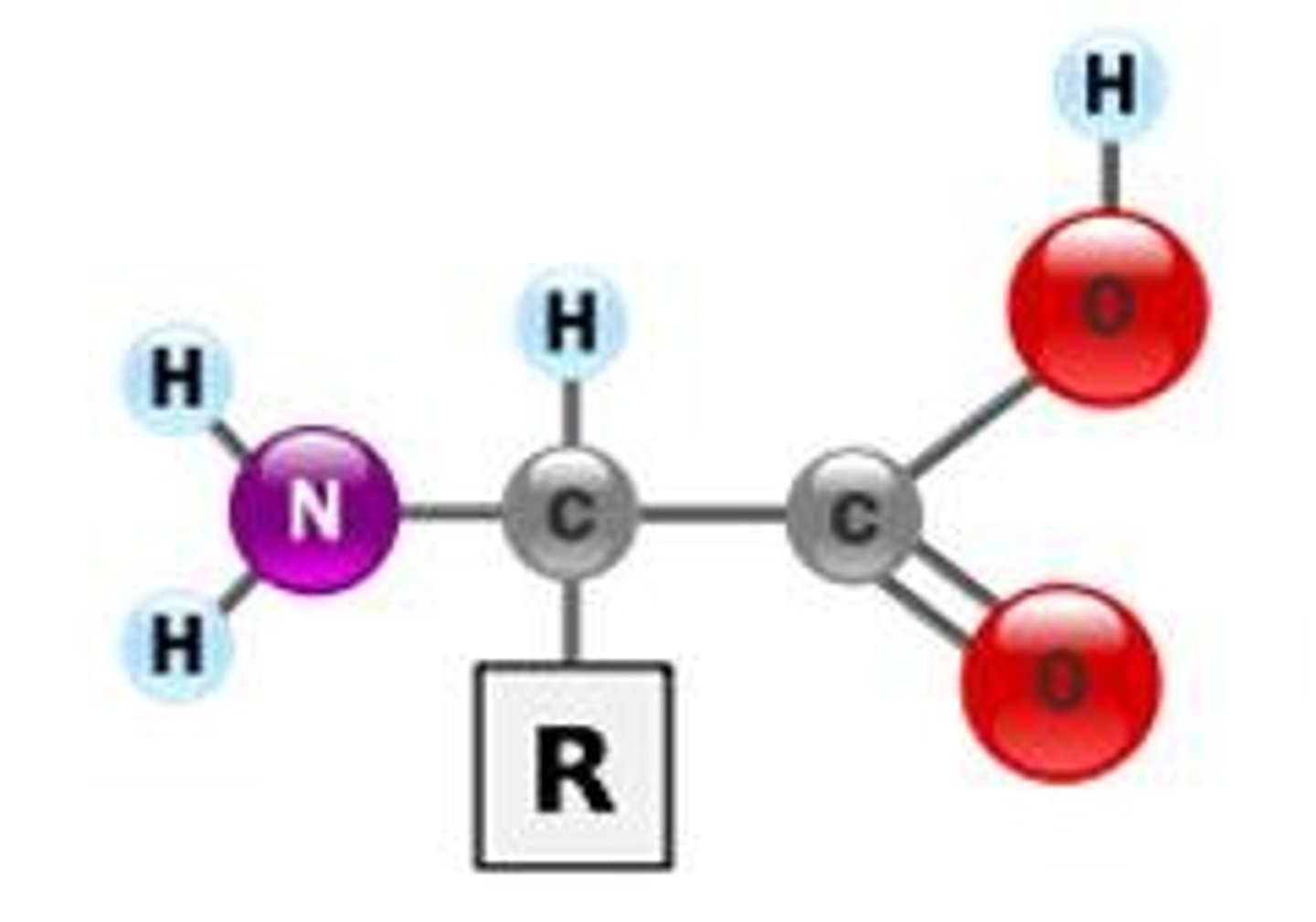
Amino; carboxyl; R
Amino acids are formed of a(n) _____ group with a(n) ________ group and a(n) _ group side chain.
20
# different amino acids which can assemble according to genetic code to make different proteins.
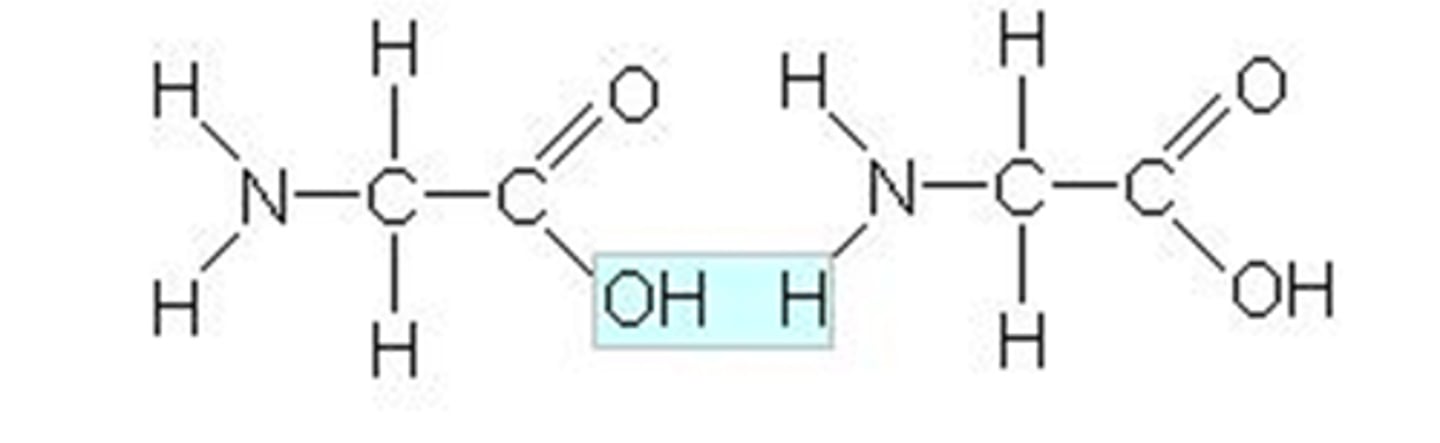
Peptide
In a _______ bond, the ---COOH group in one amino acid loses an ---OH and the ---NH2 group in another amino acid loses a H.
Polypeptide
The polymer in a protein is a(n) ___________.
3D structure
The amino acid sequence ultimately determine the __ _________ of proteins.
Structure
A protein's _________ determines its function.
Primary
At the _______ level of protein structure, a linear sequence of amino acids is joined by peptide bonds to form a polypeptide.

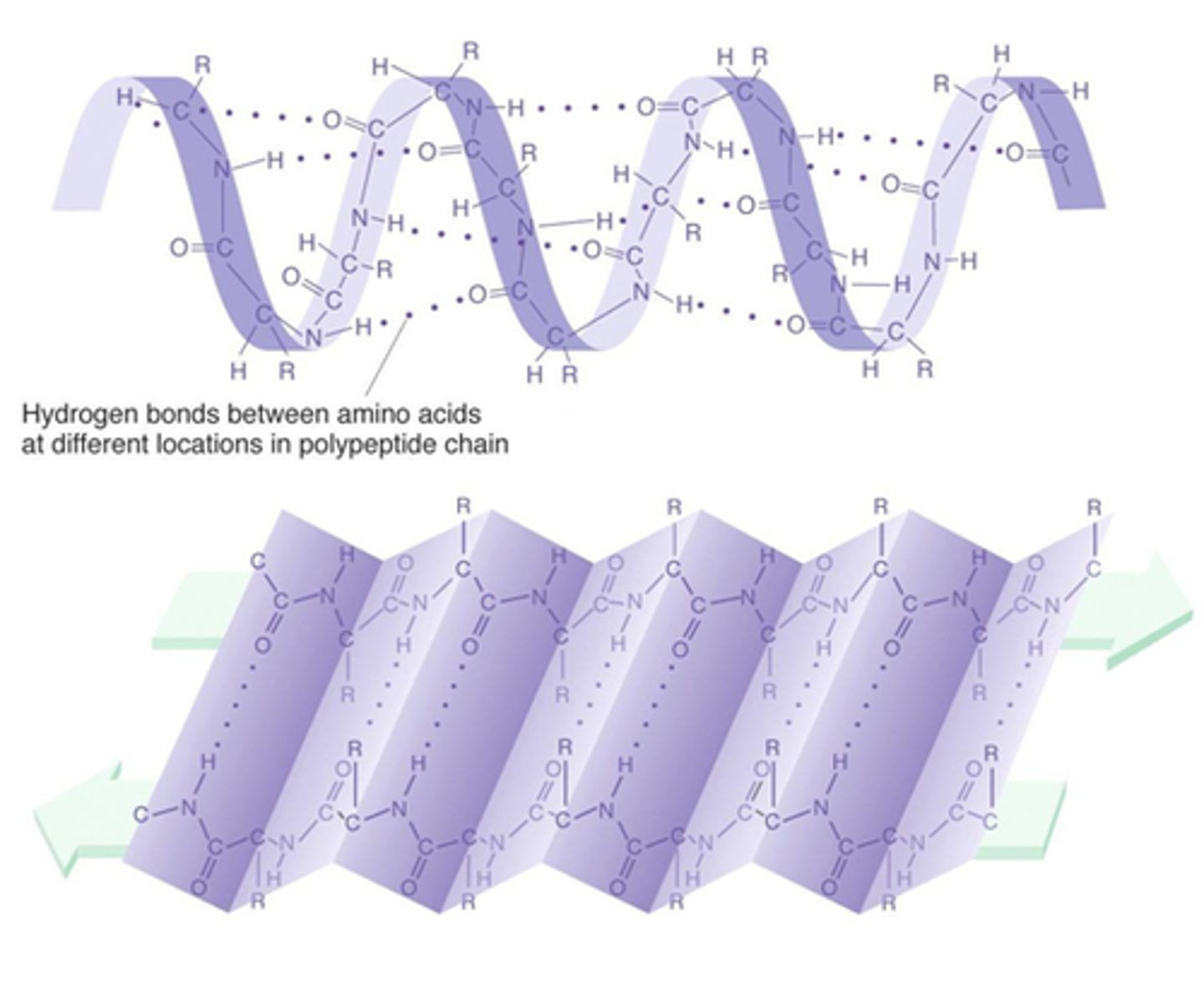
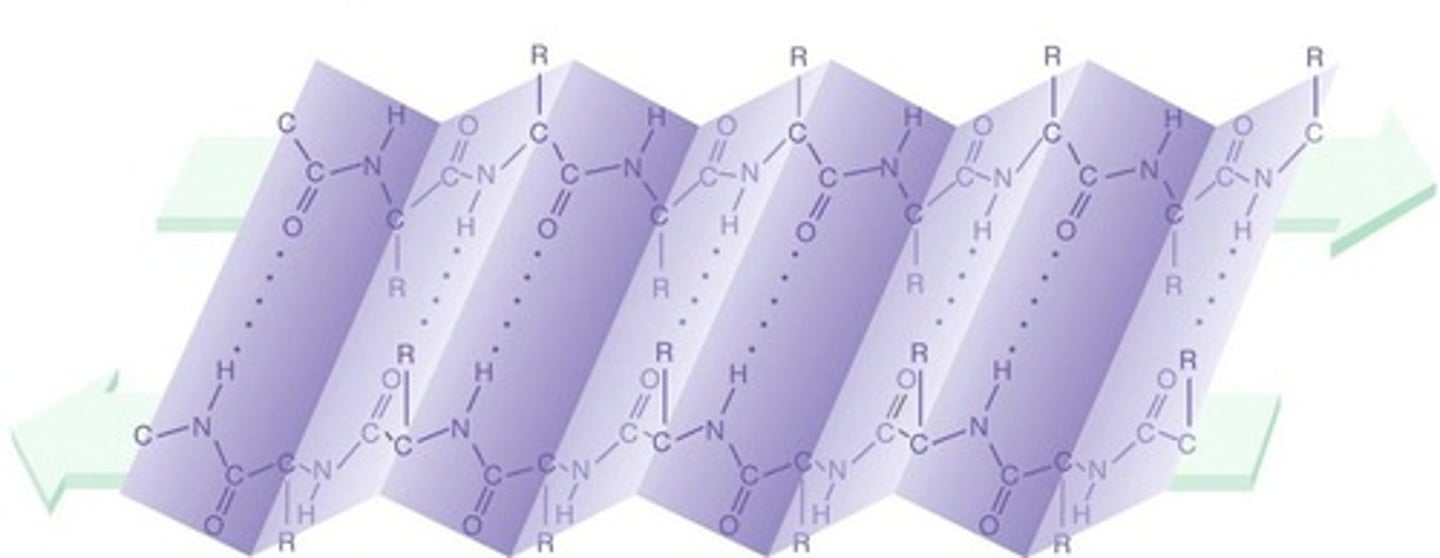
Secondary
At the _________ level of protein structure, hydrogen bonds form between the partial positive hydrogen atom of amine groups and the partial negative oxygen atom of carboxyl groups.
β Pleated Sheet
Secondary structure found in proteins
α Helix
Secondary structure found in proteins
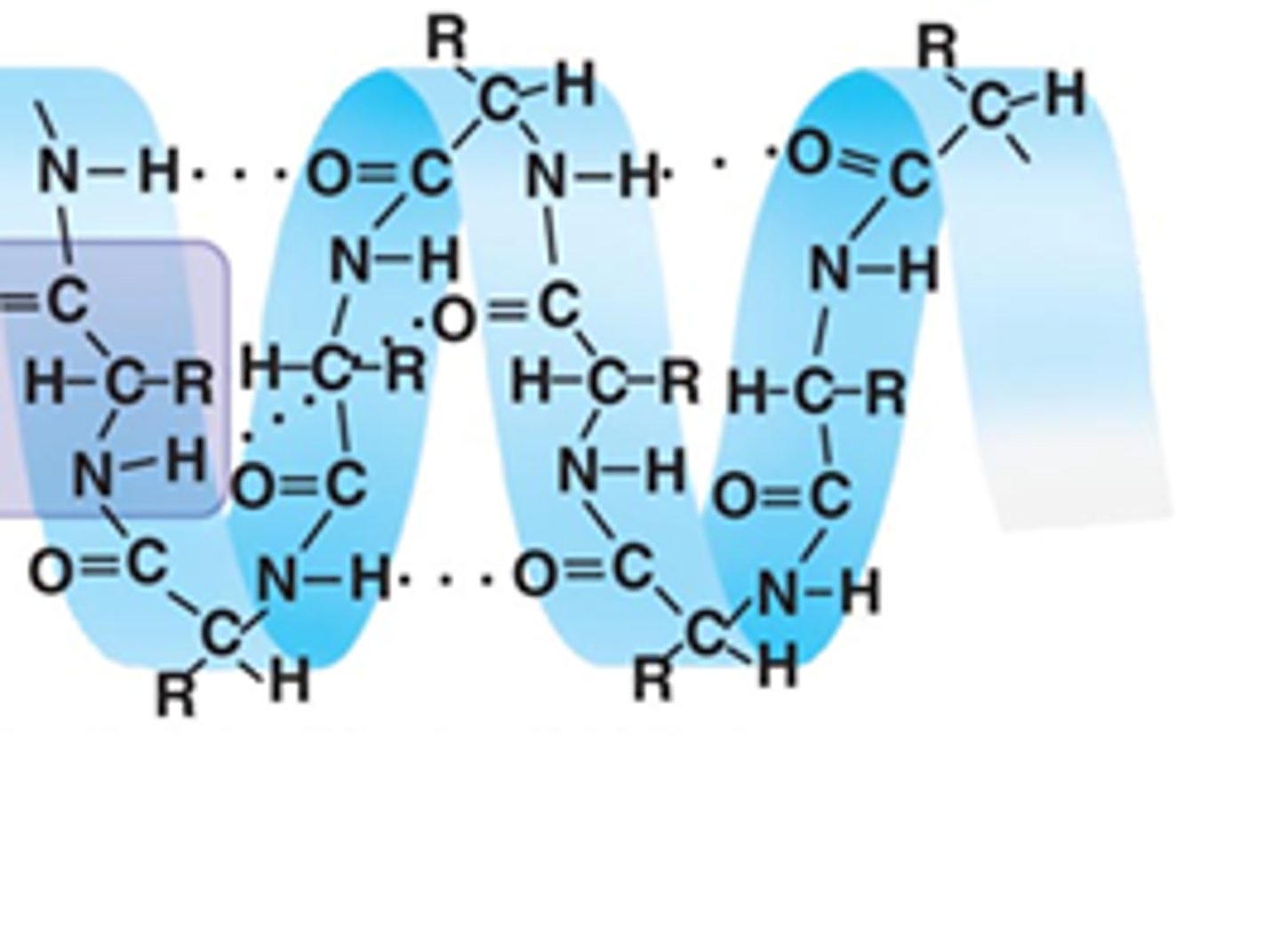
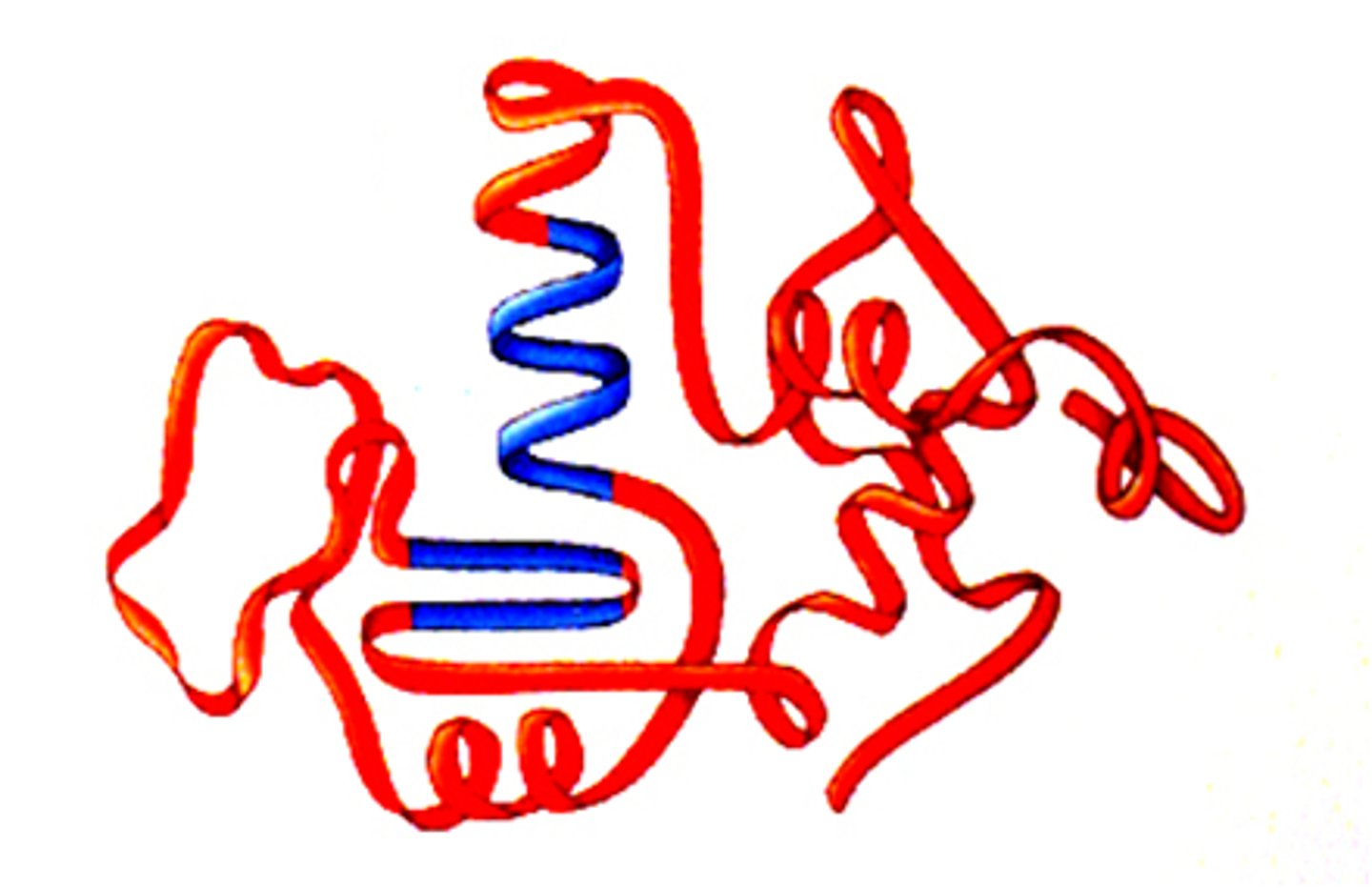
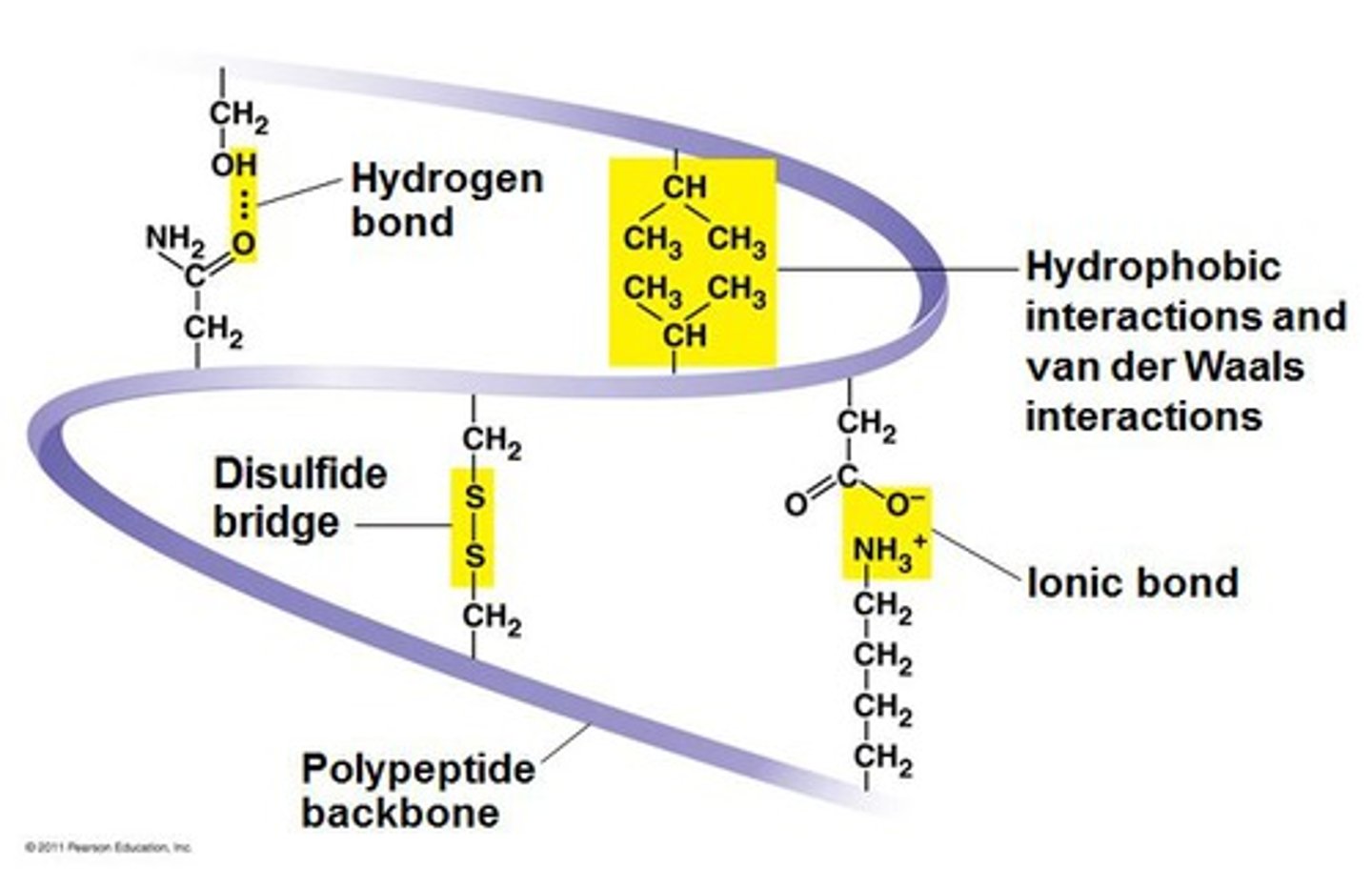
Tertiary
At the ________ level of protein structure, various chemical associations in precise regions of a polypeptide cause it to fold into a 3D shape that will determine its function.
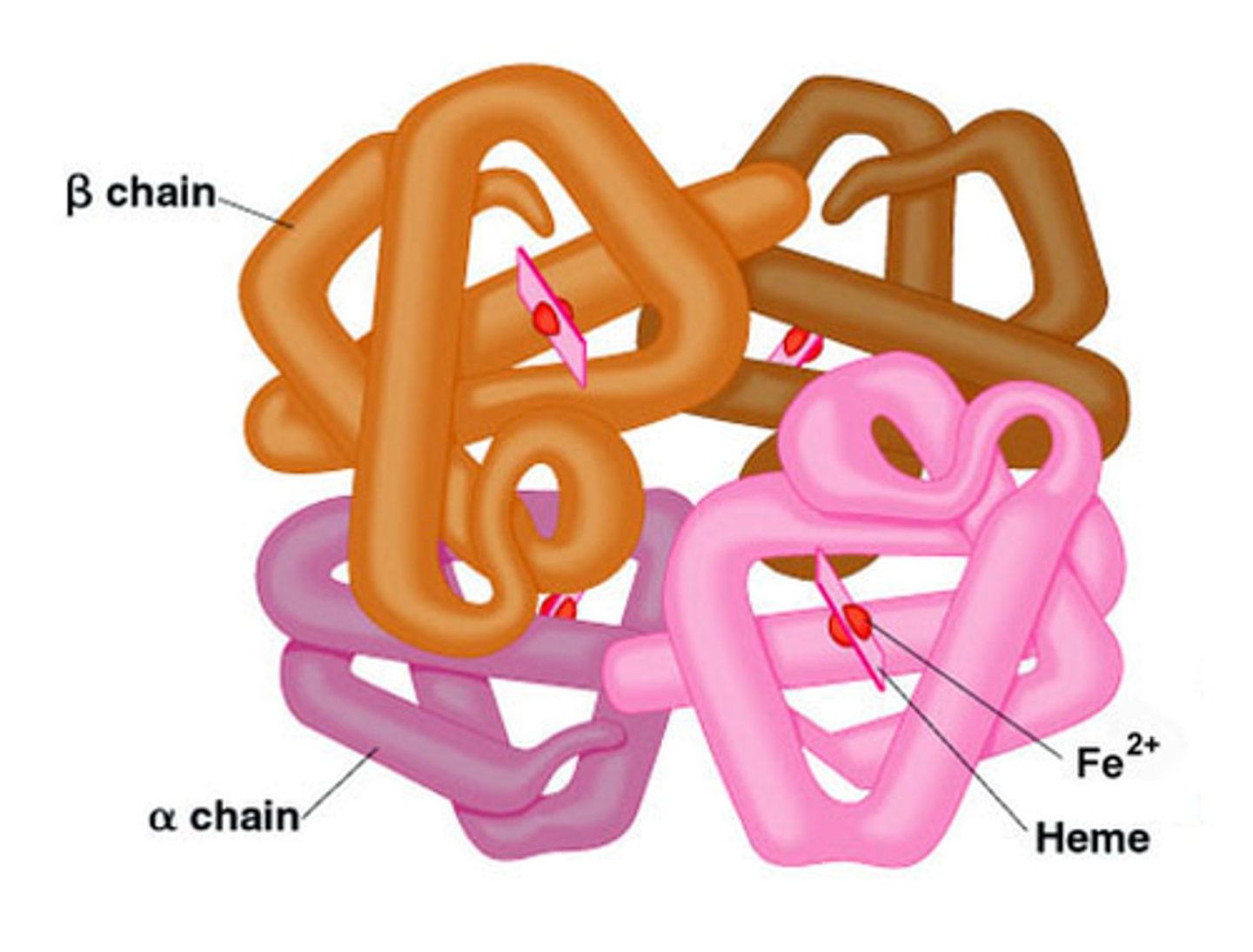
Quaternary
At the __________ level of protein structure, 2 to 4 completely formed polypeptides combine.
Receptor
________ proteins are embedded in all membranes and allow cells to interact with molecules and other cells.
Transport
_____________ proteins move molecules into and out of cells.

Defensive
_________ proteins, like antibodies, are produced in response to antigens and bind to them.
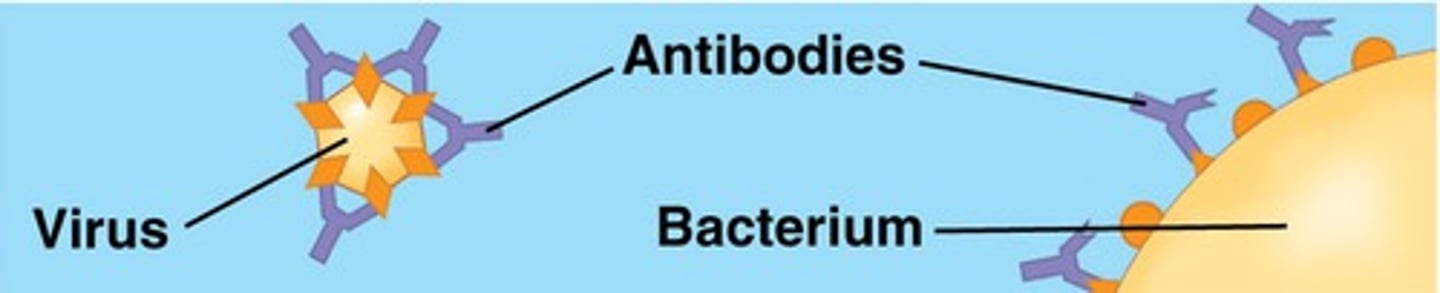
Denaturation
Excessive heat can cause ____________, in which a protein unravels and loses its native shape.

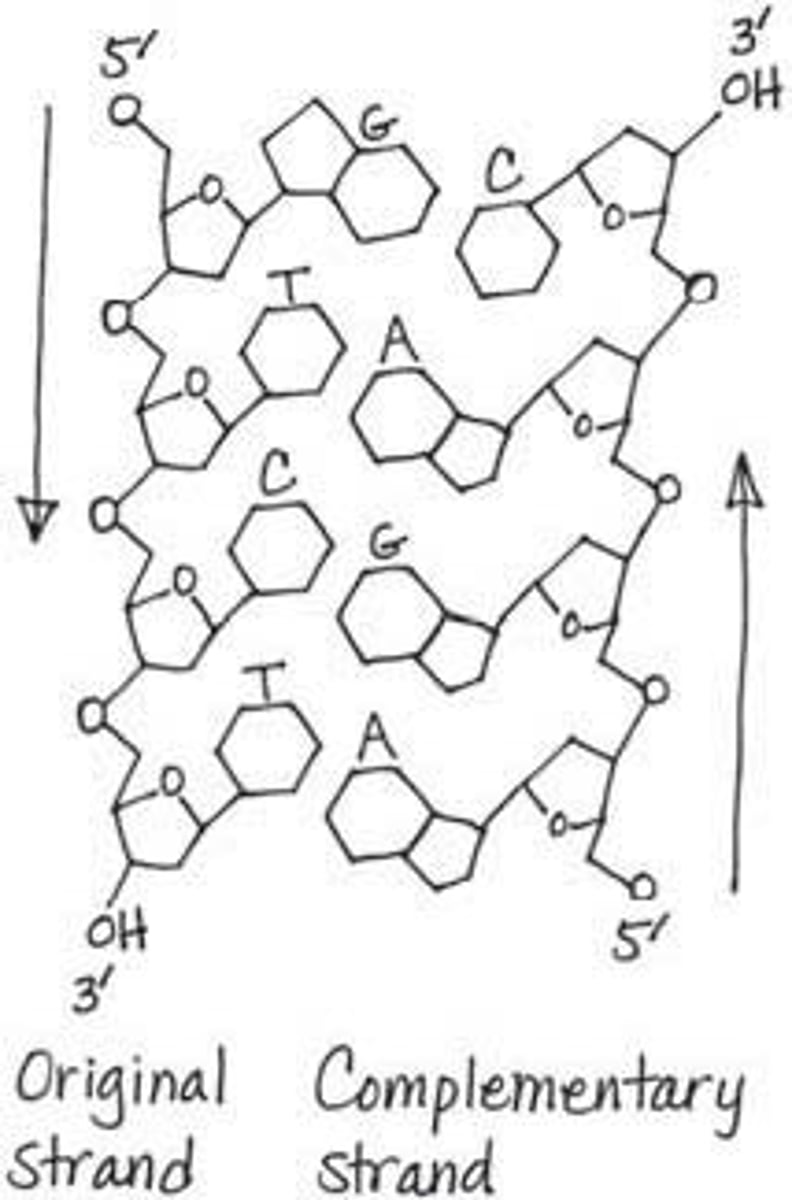
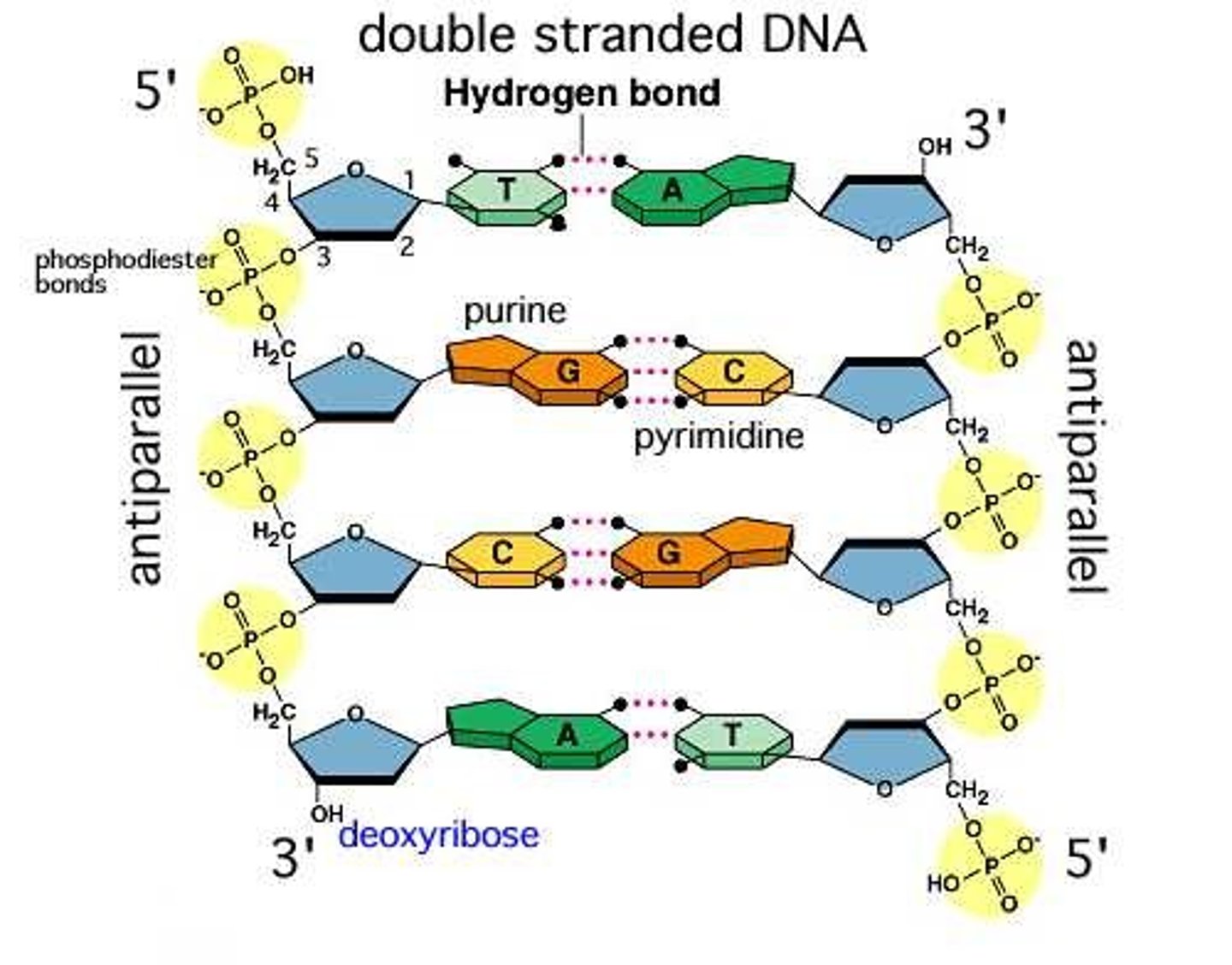
Polynucleotide
Nucleic acid Polymer
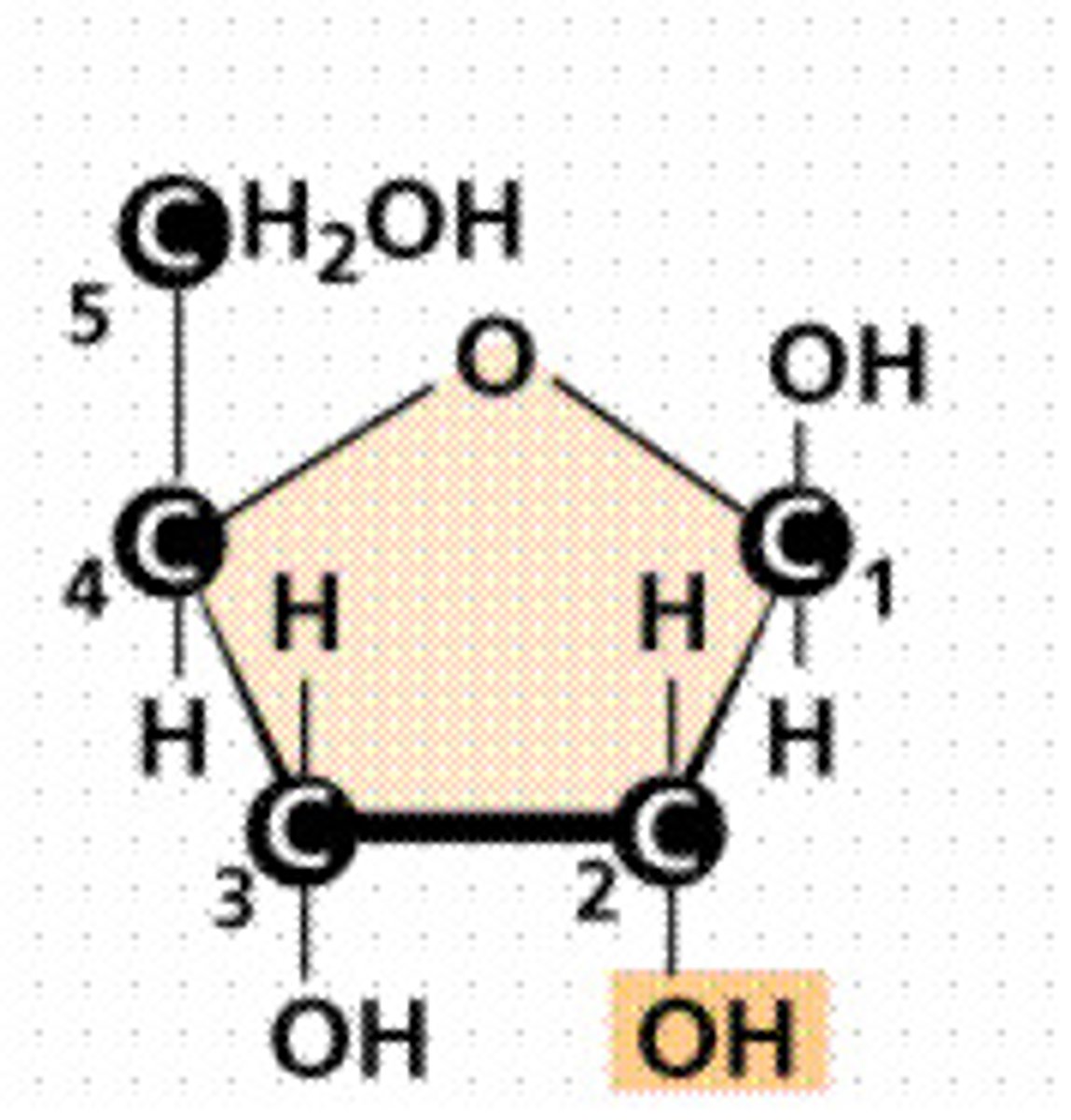
Ribose
Sugar in RNA
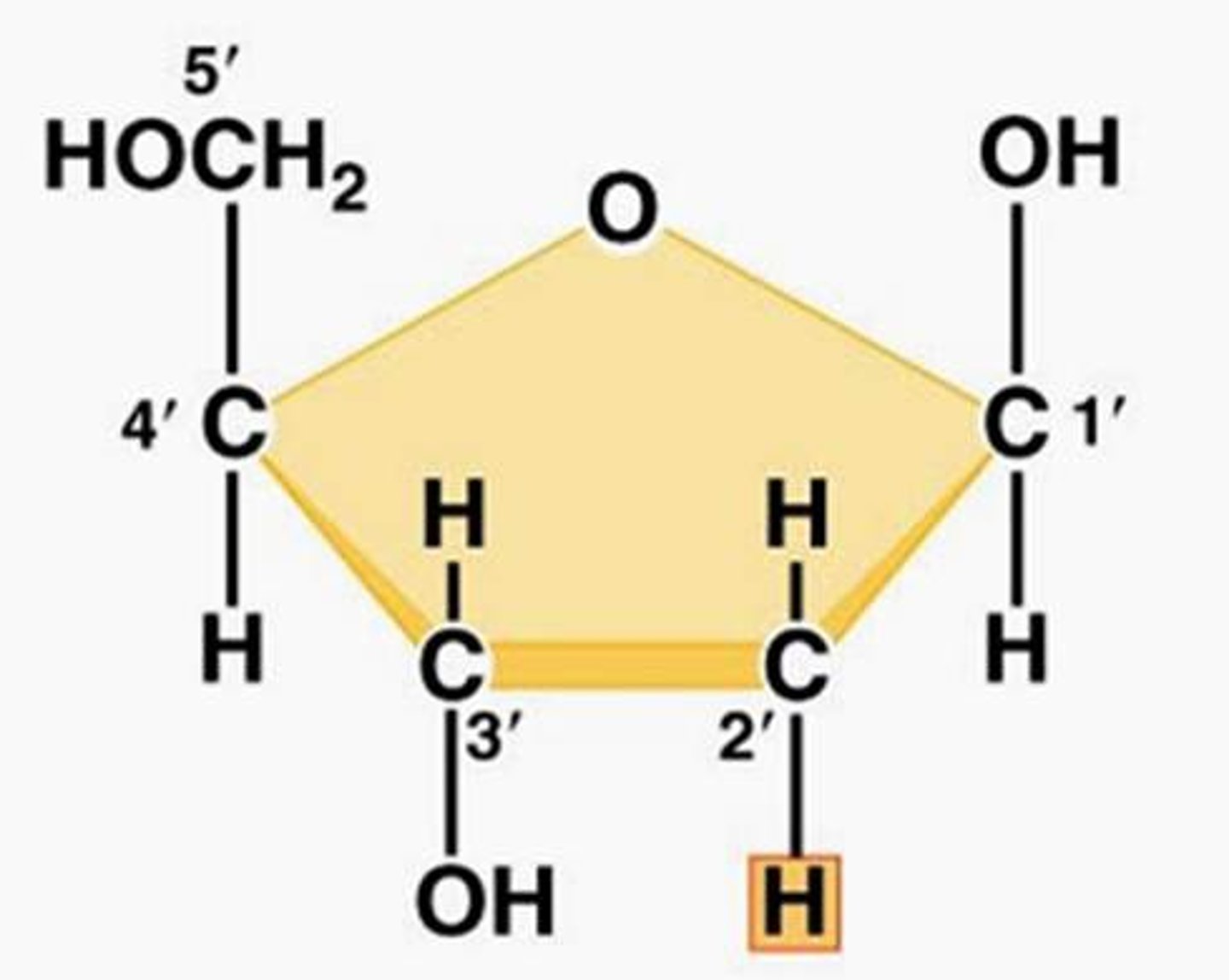
Deoxyribose
Sugar in DNA
A, T, C, G
Nitrogenous bases in DNA
A, U, C, G
Nitrogenous bases in RNA
sugars; phosphate groups
The backbone of DNA and RNA molecules is composed of alternating _____________ and ______________.
Starch
Storage form of glucose in plants
Glycogen
Storage form of glucose in animals
Nucleotide
Monomer of nucleic acids made up of a 5-carbon sugar, a phosphate group, and a nitrogenous base
Acid
A substance that increases the hydrogen ion concentration of a solution.
Adhesion
The clinging of one substance to another, such as water to plant cell walls by means of hydrogen bonds.
aqueous solution
A solution in which water is the solvent.
Base
A substance that reduces the hydrogen ion concentration of a solution.
Buffer
Minimizes changes in pH when acids or bases are added to the solution
cohesion
The linking together of like molecules, often by hydrogen bonds.
evaporative cooling
Process in which the surface of an object becomes cooler during evaporation, a result of the molecules with the greatest kinetic energy changing from the liquid to the gaseous state.
heat of vaporization
Quantity of heat a liquid must absorb for 1 g of it to be converted from the liquid to the gaseous state
Hydrophobic
Having no affinity for water; tending to coalesce and form droplets in water.
Hydrophilic
Having an affinity for water
hydroxide ion
A water molecule that has lost a proton; OH−.
kinetic energy
The energy associated with the relative motion of objects.
pH
A measure of hydrogen ion concentration equal to − log[H+] and ranging in value from 0 to 14.
polar covalent bond
A covalent bond between atoms that differ in electronegativity. The shared electrons are pulled closer to the more electronegative atom, making it slightly negative and the other atom slightly positive.
polar molecule
A molecule (such as water) with an uneven distribution of charges in different regions of the molecule.
surface tension
A measure of how difficult it is to stretch or break the surface of a liquid.
Solvent
The dissolving agent of a solution
Atom
The smallest unit of matter that retains the properties of an element.
chemical bond
An attraction between two atoms, resulting from a sharing of outer-shell electrons or the presence of opposite charges on the atoms.
Compound
A substance consisting of two or more different elements combined in a fixed ratio.
covalent bond
A type of strong chemical bond in which two atoms share one or more pairs of valence electrons.
electronegativity
The attraction of a given atom for the electrons of a covalent bond.
Hydrogen bond
A type of chemical bond that is formed when the slightly positive hydrogen atom of a polar covalent bond in one molecule is attracted to the slightly negative atom of a polar covalent bond in another molecule or in another region of the same molecule.
Ion
An atom or group of atoms that has gained or lost one or more electrons, thus acquiring a charge.
Ionic bond
A chemical bond resulting from the attraction between oppositely charged ions.
Matter
Anything that takes up space and has mass.
Molecule
Two or more atoms held together by covalent bonds.
Trace element
An element indispensable for life but required in extremely minute amounts.