Strong Acids and Bases Memorization List
0.0(0)
Studied by 0 peopleCard Sorting
1/17
Earn XP
Last updated 2:13 AM on 4/9/26
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
18 Terms
1
New cards
Hydrochloric acid
HCl
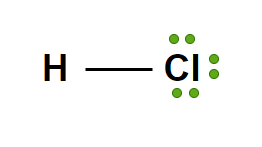
2
New cards
Hydrobromic acid
HBr
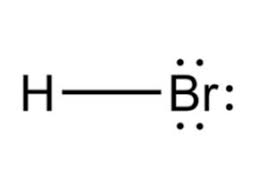
3
New cards
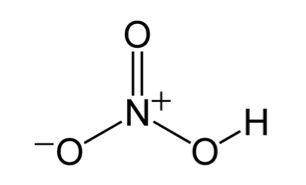
Nitric acid
HNO3
4
New cards
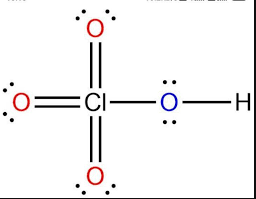
Perchloric acid
HClO4
5
New cards
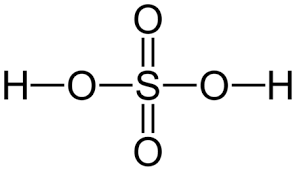
Sulfuric acid
H2SO4 (special case, two acidic protons, two ionization steps, first ionization is strong)
6
New cards
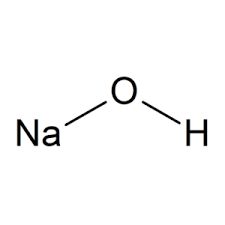
Sodium hydroxide
NaOH
7
New cards
Potassium hydroxide
KOH
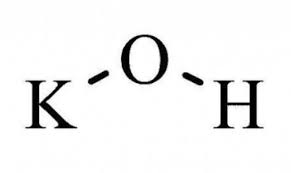
8
New cards
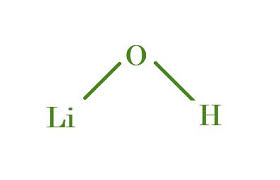
Lithium hydroxide
LiOH
9
New cards
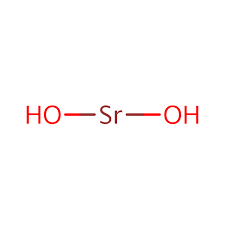
Strontium hydroxide
Sr(OH)2
10
New cards
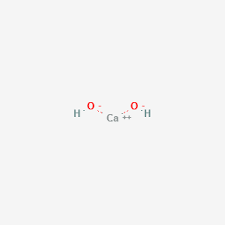
Calcium hydroxide
Ca(OH)2
11
New cards
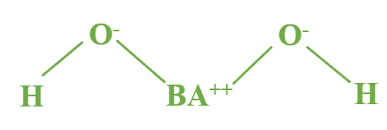
Barium Hydroxide
Ba(OH)2
12
New cards
Hydroiodic acid
HI

13
New cards
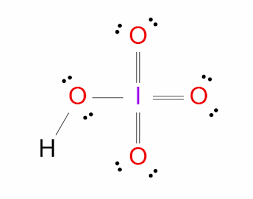
Periodic acid
HIO4
14
New cards
Perbromic acid
HBrO4
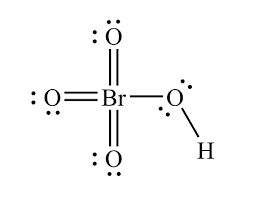
15
New cards
Magnesium Hydroxide
Mg(OH)2
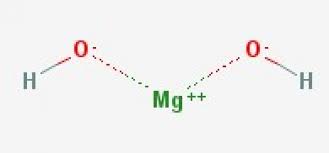
16
New cards
Hydrogen anion
H-
17
New cards
Oxygen ion
O2-
18
New cards
Sulphur ion
S2-