chemistry VSEPR
0.0(0)
Studied by 4 peopleCard Sorting
1/35
Earn XP
Description and Tags
Last updated 3:40 AM on 11/17/22
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
36 Terms
1
New cards
octet rule
atoms will gain or lose electrons to have a full octet (8 valence)
2
New cards
nonpolar covalent
0.4 or less
3
New cards
polar covalent
0.4-2.0
4
New cards
ionic
2.0 or more
5
New cards
ionic bonds
cation and anion, compound results when a metal reacts with a nonmetal
6
New cards
covalent bonds
electrons are shared by nuclei
7
New cards
VSEPR theory
valence shell electron pair repulsion, electrons are arranged as far apart from eachother as possible
8
New cards
Linear
180, 2 bonds connected
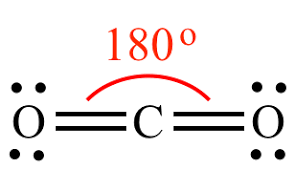
9
New cards
bent
105, 1 double bond and one sinle bond and then a lone pair, 2 single bonds and 2 lone pairs
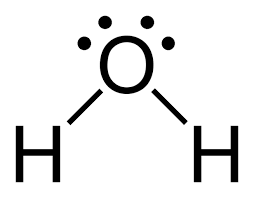
10
New cards
trigonal planar
120 degrees
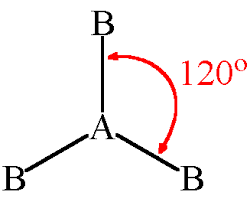
11
New cards
tetrahedral
109.5, 2 single bonds, one bond headed towards you and one headed away
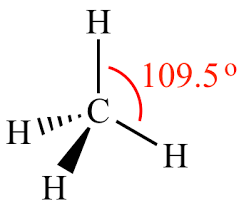
12
New cards
pyramidal
107, 1 single bond one headed towards you and one headed away
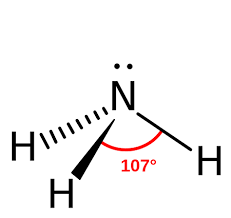
13
New cards
octahedral
90, 6 sides
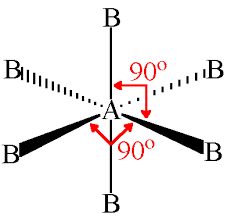
14
New cards
trigonal bipyramidal
120 and 90, 5 sides
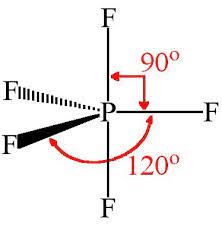
15
New cards
purple
boron
16
New cards
black
carbon
17
New cards
blue
nitrogen
18
New cards
green
halogen
19
New cards
white
hydrogen
20
New cards
red
oxygen
21
New cards
polar definition
electrons are shared unequally
22
New cards
nonpolar definition
electrons are shared equally; symmetrical
23
New cards
ionic electrons gained/lost
lost/gained
24
New cards
ionic types of atoms in bond
metal + nonmetal
25
New cards
ionic melting point
high
26
New cards
ionic dissolves in h2o
yes
27
New cards
other characteristics of ionic
brittle conductors
28
New cards
covalent electrons gaines/lost
sharing
29
New cards
covalent types of atoms in bond
nonmetals
30
New cards
covalent melting point
low
31
New cards
ovalent dissolves in h2o
no
32
New cards
other characteristics of covalent
nonconductors
33
New cards
why do metals and nonmetals usually form ionic compounds whereas two bonded nonmetals are never ionic
metals lose electrons and nonmetals gain electrons to achieve an octet
34
New cards
how to predict ionic, covalent, polar, nonpolar
determine element types, polar: 0.4-2.0, nonpolar: 0.4 or less
35
New cards
polyatomic ions
many atoms with a charge
36
New cards
common ions
the ion that forms by acheiving the octet