immuno quiz 2 (starts chapter 4 slide 42)
1/119
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
120 Terms
how is polyclonal antisera collected
by immunizing with antigen of choice then collecting the fluid component of the clotted blood
antisera
a heterogeneous collection of polyclonal Abs that bind to different epitopes present on the antigen (the Ag stimulates multiple different B cells)
why is antiserum of limited use in many clinical and laboratory assaays and diagnostic tests
limited supply; always need to make more
batch to batch variability (not consistent, makes bad for testing)
range of affinities
multiple epitopes recognized
cross reactivity for similar antigens
monoclonal antibodies (mAbs)
antibody produced by a single clone of B lymphocytes so that the antibody molecules are all identical in structure (homogenous)
and have the same antigen specificity (bind to the same epitope on the antigen)
myeloma cells
cancerous plasma cells that secrete Abs and are immortal (can grow/divide forever)
The ones they fuse with B cells are engineered to not produce Abs
Hybridoma
a cell created by fusing a normal activated Ab-producing B cell with a myeloma cell
Has immortal growth properties (which comes from the myeloma cell) and secretes mAbs (derived from the activated B cell)
out of the B cell population a majority of B cells respond, True or False
False, a minority respond
steps to producing mAbs
Immunize mouse with antigen of choice (stimulating a polyclonal B cell response)
collect all B cells from spleen (mast majority will not have the potential to secrete the desired Abs)
fuse the the B cells and myeloma cells using polyethylene glycol
This results in a mixture of hybridomas and unfused B cells and myeloma (Myeloma cells must be removed or killed or they will overgrow the culture
hybridoma cells are grown on a selective durg media that allows only for hybridoma growth, unfused myelomas will be killed by drugs and the unfused B cells have limited life spans and will die
A vast majority of these hybridomas are not making desired Abs, so each hybridoma has to be individually cultured and tested for highest affinity
once desired hybridoma is found it is cultured and the mAbs are purified and can be stored indefinitely
research applications that use mAbs
western blots
ELISAs
flow cytometry
immunohistochemisty
diagnostic applications that use mAbs
testing for...
HIV
bacterial infections
pregnancy
what was the first mAbs used for therapeutically and why did it fail? what is the solution?
a mouse mAb used to prevent organ rejection. however its use caused severe complications because humans made Abs specific for the C regions of the mouse Ab
the solution to this is to make mAbs less foreign to the human body via genetic engineering
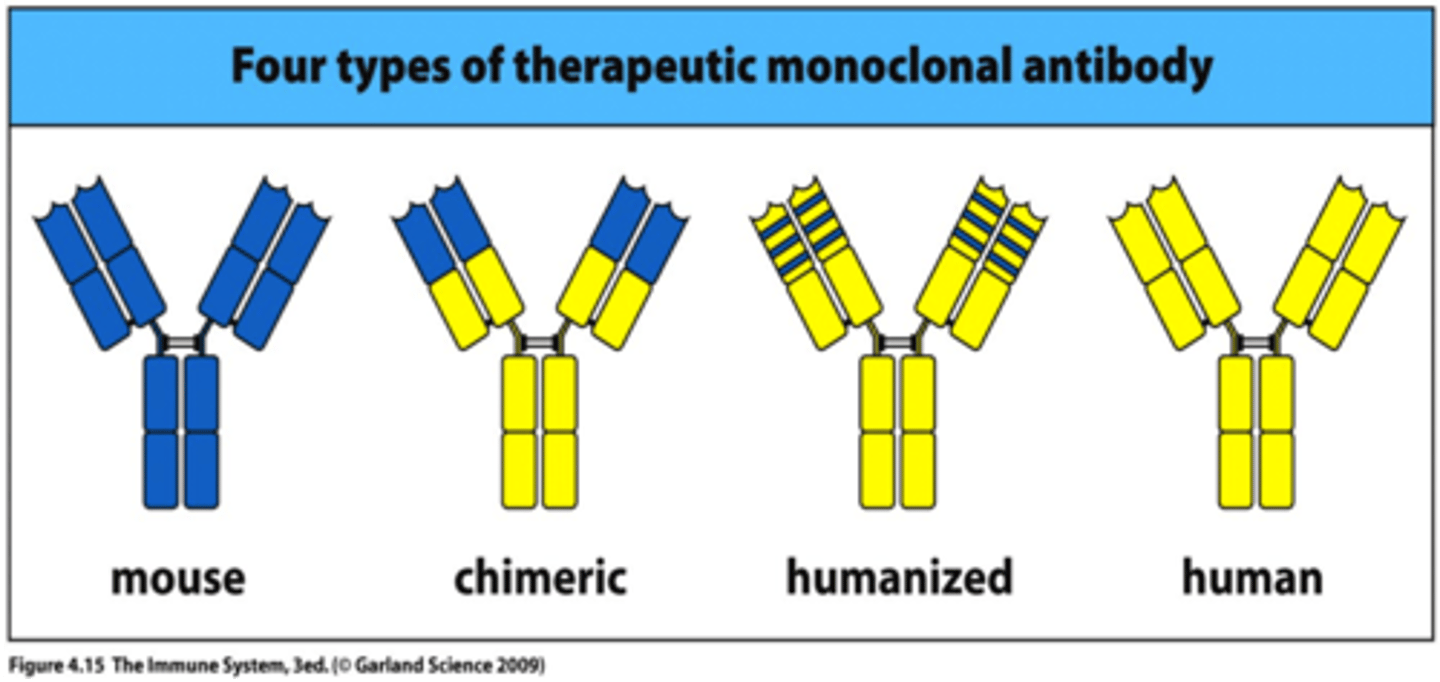
what are the 4 levels of mAbs as therapeutic reagents
mouse mAbs (poor)
Chimeric mAbs (fair)- only contain mouse variable regions
Humanized mAbs (good) - only contain the mouse HR (CDR) regions which bind to the target
Fully human mAbs (best)- made by replacing the Ig genes of a mouse with human Ig genes
what does the T cell receptor resemble
FAb fragment of immunoglobulin
what is a T cell receptor consist of
an alpha and beta chains
4 Ig domains
one antigen binding site
Va and Vb have 3 HV regions each (same as Abs)
what is the TCR specific for
the antigenic peptides and the surrounding surface of the major histocompatibility complex (MHC)
residue
amino acid
Where is TCR diversity generated?
in the thymus by VDJ gene segments
alpha and beta chains are like to what chain of Abs
alpha is like to light (VJC)
beta is like to heavy (VDJC)
what process of diversity DOES NOT happen in TCR
there is no somatic hypermutation, so no affinity maturation but other processes like gene rearrangement, junctional diversity, recombinase complex, 12/23 rule all happen
are there Isotypes of the TCR, and what does this mean for the amount of C genes
there are fewer C genes because there is no isotypes of TCR
Severe combined immunodeficiency syndrome (SCID)
results in the absence of both functional B and T cells
die in infancy from common infections unless they receive a bone marrow transplant
many genetic defects can lead to this
genetic defects that lead to T cell deficiencies are worst. why? because influences humoral and cell mediated. No T cells, no Ab produced either
one cause is a genetic deficiency of RAG1 or 2 proteins. If B and T don't successfully undergp VDJ recombination during development, these developing cells dine in the bone marrow or thymus
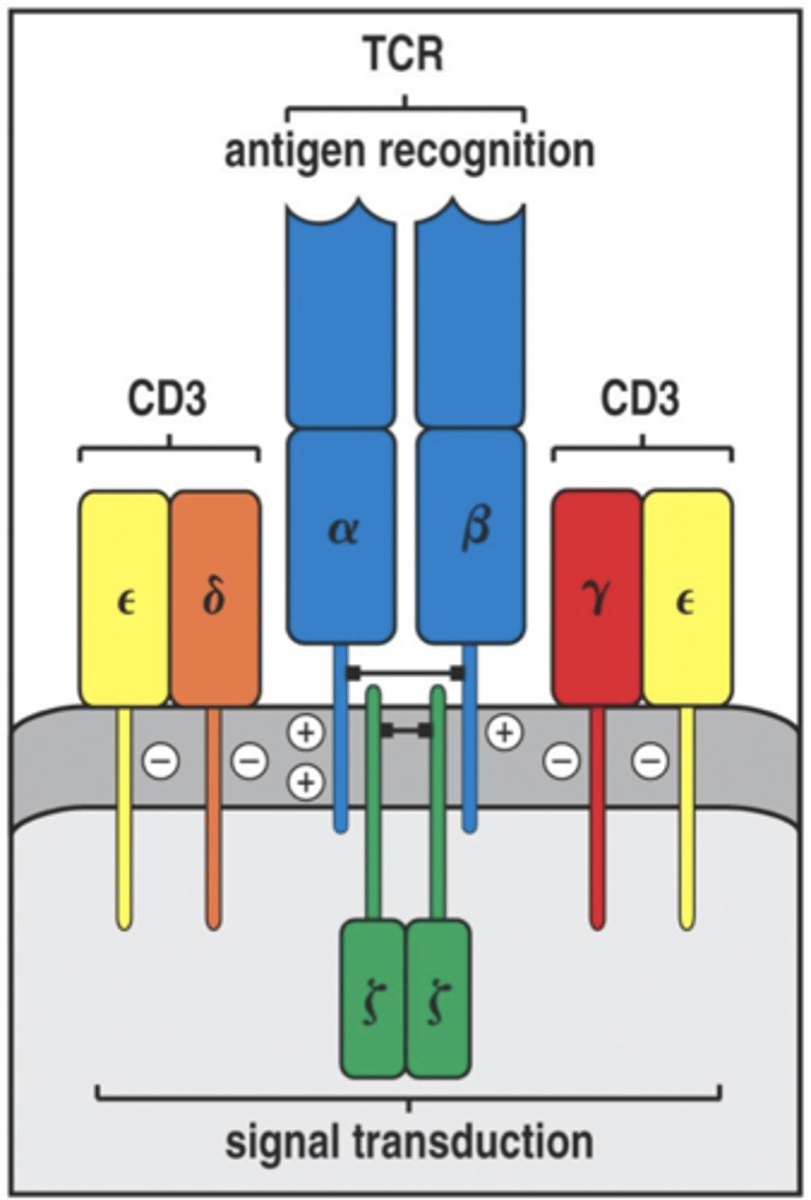
what are the components of TCR complex
composed of 8 polypeptides
2 CD3 one with gamma and one with a delta both having an epsilon
there are two copies of zeta chain (homodimer)
these invarint polypeptides are necessary for the transduction of signals to the cells interior after the TCR has bound antigen
if any of these proteins arent expressed the a and the b chains do not leave the ER
what is the second class of T cells
γδ T cells
less diverse than those of aB T cells. Lack CD4 and CD8. TCRs do not bind MHC and antigenic peptides
their binding sites recognize a wide range of chemical structures that distinguish stressed, infected, or malignant cells from healthy cells
function more in innate immunity, similar to NK cells (mechanism of killing)
Low numbers are present in blood and peripheral tissues
also express PRRs
antigen processing
process where pathogen's protein antigens are broken down into antigenic peptides inside of the cell
antigen presentation
process where peptide antigens are bound by major histocompatibility complex (MHC) molecules and sent to the cell surface. AB TCRS only recognze ags presented by MHC molecules, these cells are antigen-presenting cells
MHC class I molecules present
peptide Ags derived from intracellular organisms whose proteins were degraded in the cytosol
present to CD8 cytotoxic cells
MHC class II molecules present
presents peptide antigens derived from extracellular organisms that were taken by the cells and whose proteins were degraded into peptides within endocytic vesicles or lysosomes
present CD4 helper T cells
Co-receptor CD8
expressed by cytotoxic T cells
must bind to the MHC class I, but binds to a different part than the TCR
CD4 co-receptor
expressed by helper T cells can recognize on macrophages (trigger to secrete cytokines to proliferate macrophages) or they can recognize on B cells (secretes cytokines that drive the differentiation of the b cell into a plasma cell making bacterial specific antibodies)
MHC I components
one a chain complexed with a smaller protein called the B2-microglobulin. same B2 used with all of the different MHC class I subtypes
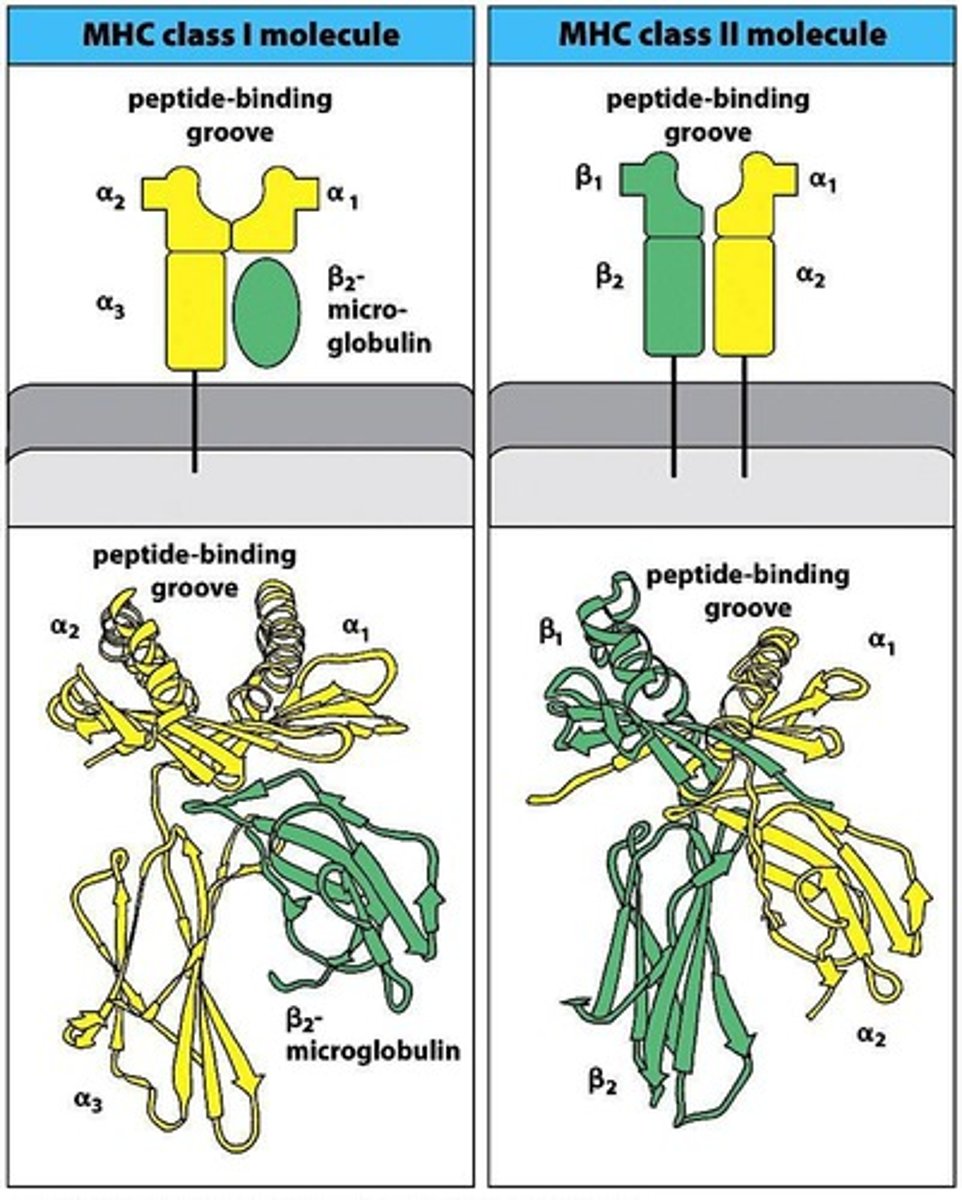
MHC II components
one a chain and one B chain
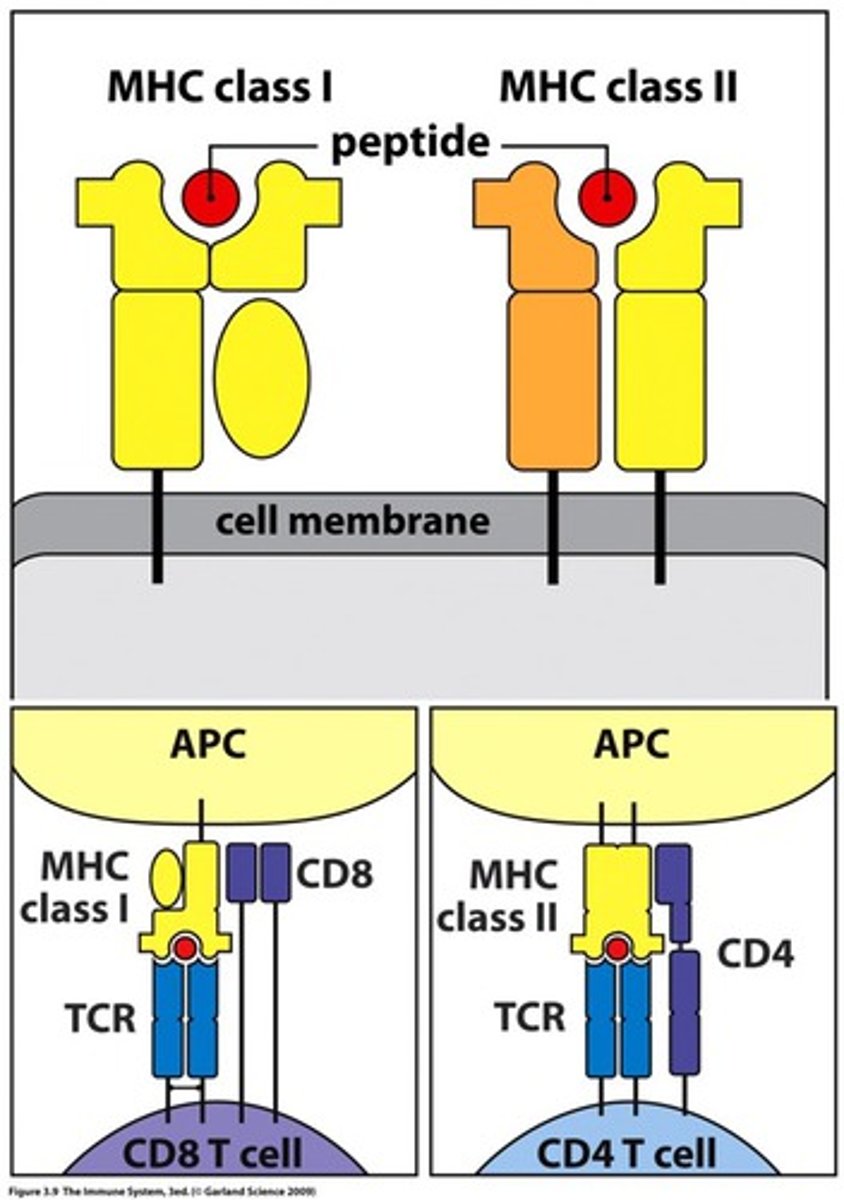
what domain does CD8 bind to of MHC I
it binds to the a3
what domain does the CD4 bind to of the MHC II
it binds to the B2
why is it important that co-receptors bind to the correct
it ensures that the correct T cell binds to the antigen
The T cell will not be activated if CD4 or 8 is not bound to MHC
peptide binding grove of the MHC I
binds peptides 8-10 amino acids in length (smaller)
most peptides that bind have a hydrophobic or basic amino acid at C terminus
peptide binding grove of the MHC II
Binds peptides ~10-25 amino acids in length
promiscuous binding specificity
ALL MHCs can bind to a variety of different antigenic peptides (not an endless number and not at the same time)
binding is therefore degenerate
pathogenic proteins
endogenous Ags as well as host proteins that are damaged. misfolded, or no longer needed, get degraded into peptides in the cytosol and then are transported into the ER.
how is MHC I made
by ribosomes on the ER and is trasported into the ER lumen. Within the ER both self-peptides and peptides from pathogens bind to MHC class I molecules and the complex is transported to the cell surface.
in healthy cells what will the MHC I present as opposed to infection
self peptides but during infection some antigenic peptides are presented
MHC class I cytosolic pathway step 1
degradation of proteins into peptides by proteasomes, which are barrel-shapped protein complexes that normally break down host proteins that are damaged, mis-folded, or no longer needed
what do NK cells do to proteasomes during infection
Nk cells release INF-gamma which can modify the proteasome in cells. This creates an immunoproteasome which releases peptides faster and specializes in making peptides with hydrophobic or basic amino acids at C terminus
this makes them better able to bing to the MHC I molecules
MHC class I cytosolic pathway step 2
peptide transport from cytosol into the ER
Transporter Associated with Ag Processing (TAP) use ATP to transport peptides into the ER. TAP is a heterodimer of TAP 1 and 2
TAP has an affinity towards peptides greater than or equal to 8 amino acids and hydrophobic or basic (C terminus)
TAP genes are polymorphic what is the significance of this
many different alleles in the population, some aren't as good at transporting polypeptides so less peptides get into the ER
this leads to increased viral respiratory infections
bare lymphocyte syndrome type I
a genetic immunodeficiency disease where people have a dysfunctional TAP
low CD8 responses because surfaces lack MHC I because it must have an antigen bound to go to the surface of the cell
MHC class I cytosolic pathway step 3
assembly of peptides with MHC class I and transport to the cell surface
chaperones promote the folding and peptide loading of the MHC class I molecules in the ER
only peptides with a high affinity for the MHC class I can bind
The peptide:MHC class I complex then leaves the ER via a vesicle, travels through the golgi and is then expressed on the cell surface
can a MHC class one leave the ER without a bound peptide
no
how do infections lead to a better MHC class I pathway
Infection leads to more INF-gamma which leads to proteasome being transformed to immunoproteasome which is better able to generate peptides that bind MHC class I
What pathogens or disease inhibit the MHC class I pathway and how
herpes simplex viruses- makes and inhibitor protein that partially blocks TAP from transporting peptides
Adenovirus (colds)-synthesizes a proteins that partially inhibits MHC class I from leaving ER
Bare Lymphocyte syndrome type I- no functional TAP
what cells express MHC class I molecules
all cells constantly express them excepts RBCs
allows CD8 cytotoxic T cells to kill all virally infected cells
which cells express MHC class II molecules
only professional antigen presenting cell:
B cells
Macrophages
Dendritic cells (most effective)
MHC class II expression can be unregulated during infection (for example by INF-gamma)
MHC class II endocytic pathway step 1
professional APC take up extracellular antigens
Macs and dendritic cells take up pathogens via phagocytosis (bacteria, toxins, viral particles)
B cells take up small soluble Ags (bacterial toxins) by receptor mediated endocytosis using BCRs (they are not phagocytes)
ALSO some bacteria are intracellular pathogens that invade cells and normally replicate in intracellular vesicles. their antigens are also processed by the MHC class II pathway
MHC class II endocytic pathway step 2
extracellular antigens are processed into peptides in endocytic vesicles that become progressively acidic. These vesicles contain proteolytic enzymes that become more active as they move inward and the pH decreases
processing generates 10-25 aa peptides that binds MHC class II
MHC class II endocytic pathway step 3
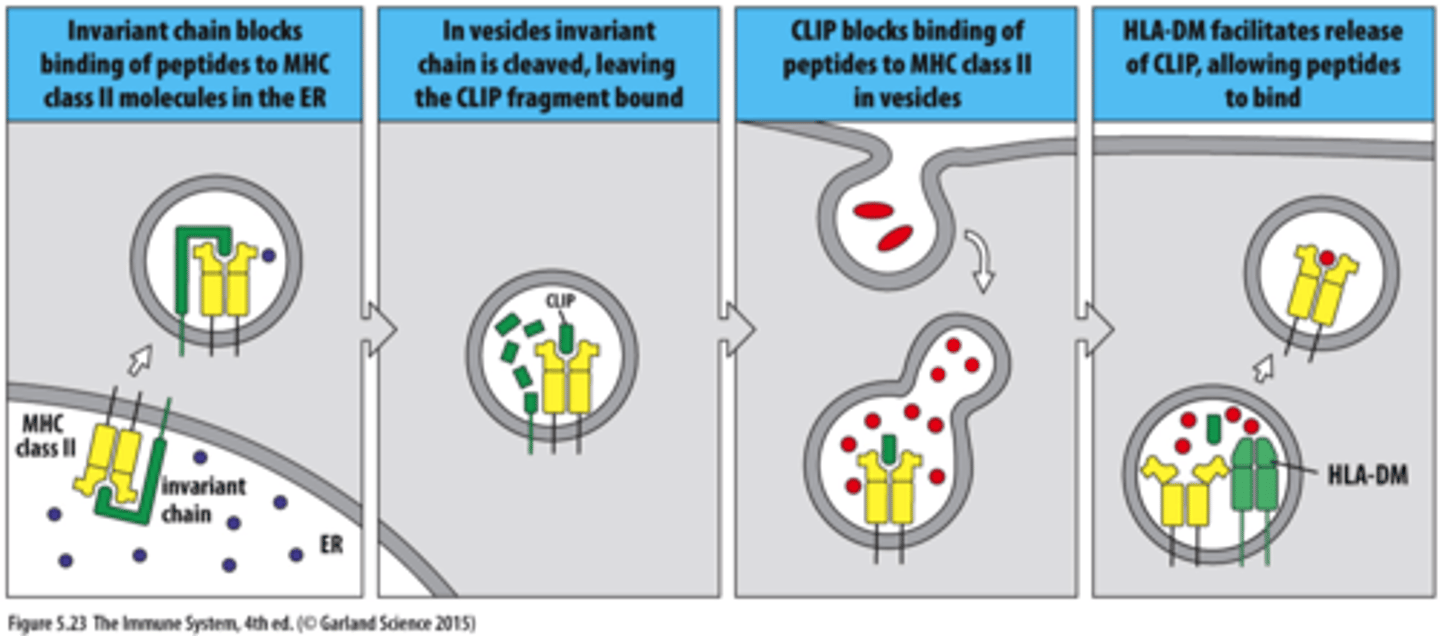
The invariant chain prevents peptides from binding to MHC class II until the complexes reaches the site of extracellular protein degradation
MHC II is made on ER. Invariant chain prevents binding of self-peptides to MHC II in ER (if no invarient MHC is degraded in ER) then leaves the ER as a vesicle
Invariant chain is cleaved leaving a bound fragment CLIP
CLIP blocks binding of peptides to MHC II in vesicles
HLA-DM removal of CLIP and loading of a peptide that binds with sufficient affinity. vesicle goes to cell surface
leprosy and tuberculosis is caused by what and how
intracellular mycobacteria avoid destruction and thrive (grow/replicate) within the vesicular system of macrophages and dendritic cell
these bacterias want to be eaten
they survive in modified phagosome that do not fuse with lysosome (which they prevent)
these modified phagosomes also limit the production of antigenic peptides that could be processed and presented by MHC class II to CD4 T cell
sometimes it is necessary that an extracellular antigen be processed by the MHC class I pathway, why?
dendritic cells are the major APC that presents viral peptides to initiate CD8 cytotoxic T cell clonal expansion within secondary lymphoid tissues
however most viruses only infects cell types not located in the secondary lymphoid and they do not usually infect dendritic cells
then, how can naive specific CD8 T cells located in the secondary lymphoid tissues be activated to undergo clonal expansion and generate effector CD8 T cell IF the infected cells expressing MHC class I/ viral peptides are located in non lymphoid tissue
cross presenting
a third pathway of Ag processing that enabled extracellular antigens to be presented by MHC I
ability of dendritic cells to take up an extracellular virus particles (or dying virally infected cell) by phagocytosis (or by receptor-mediated endocytosis or macropinocytosis) and release the viral proteins from the endocytic vesicles into the cytoplasm from where the viral proteins can be processed by the proteosome and the MHC I pathway
Thus dendritic cells can present viral peptides to CD8 T cells in secondary lymphoid
macropinocytosis
endocytic mechanism in which there is nonselective incorportation of fluid, macromolecules, antigens, and other small substaces, into cells by membrane invagination and the pinching off of relatively small vesicles
what is the MHC called in humans and on what chromosome is it found
the human leukocyte antigen (HLA) complex and it is a cluster of tightly linked genes on chromosome 6
the region was first recognized as the site of genes that caused T cell rejection of transplanted tissues
what region of the genome is most strongly correlated with human disease
the MHC region
what two components contribute to MHC diversity
1. existence of gene families- there are multiple similar genes in one individual that encode different MHC class I and II molecules
a cell can express more than one type of MHC I and II proteins simultaneously (codominant expression, MHC genes do not rearrange like Ab or TCR)
2. Genetic polymorphism- the existance of two or more alleles (different forms) of a MHC gene within a population
in humans how many MHC class I isotypes are there and how many (and which ones) are recognized by T cells
6 are made 3 are recognized by T cells
HLA-A
HLA-B
HLA-C
in humans how many MHC class II isotypes are there and how many (and which ones) are recognized by T cells
5 are made, 3 are recognized by T cells
HLA-DP
HLA-DQ
HLA-DR
allotypes
protein products of the alleles
genetic polymorphism of MHC
there are many MHC alleles in the population, these genes are among the most highly polymorphic genes that are known
the variability between alleles in at the site that binds TCRs and peptides
MHC class I and II are both very polymorphic, but which is more diverse
MHC class I
where is the majority of the diversity in the MHC class II
in the B chain
whats the difference between the Beta part of class I and II region on the gene
For class I the B microglobulin gene is on a different chromosome than the alpha chians
For class II the B chain is close to the genes that code for the alpha
What is special about the MHC class II HLA-DR
Every human on the plant has one DRA for the a chain and one gene DRB1 for the B chain
however SOME individuals have an extra gene (DRB3-5) that can encode an extra B chain of class II.
ANY DRB can pair with DRa. so some individuals can express more than one HLA class II encoded at HLA-DR
on which human chromosome are the human MHC genes held
chromosome 6
haplotype
particular combination of HLA alleles on one chromosome
HLA type
particular combination of HLA alleles from BOTH chromosomes
In one heterzygous individual up to how many different class II isotypes can be expressed by a single cell
16
In one heterzygous individual up to how many different class I isotypes can be expressed by a single cell
6
where is the variability in HLA or MHC class I compared to II
In class I its found in the a chain
In class II its found in the B chain
how many amino acids can MHC differ
1-50
What secondary structure can make contact with both peptides and TCRs
alpha helices
what makes a relatively small number of MHC molecules able to present an endless number of antigenic peptides to T cells
degenerate or promiscuous binding specificity
anchor residues
those amino acids in a peptide sequence that are actually responsible for binding to a particular MHC isotype. They dont need to be identical but they are related
for example if the antigen is of the proper length and anchor residues in the correct positions ( 9 aa, and in certain positions there are large hydrophobic AAs)
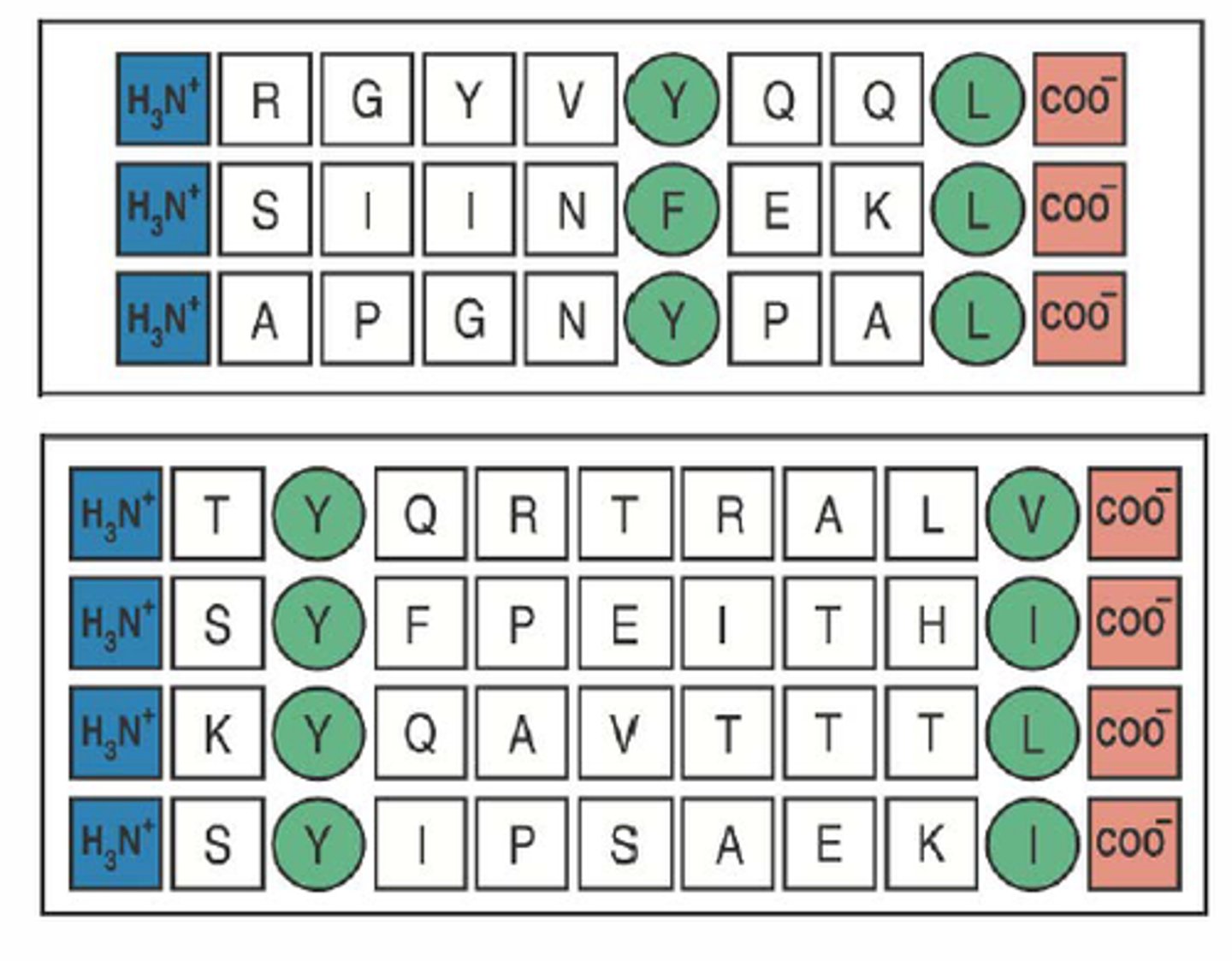
peptide binding motif
combination of anchor residues that bind to a particular MHC isotype
MHC restriction
property that a given TCR recognizes its peptide antigen only when the peptide is bound to a particular form of MHC molecule
T/F, more than one T cell can recognize and bind to the same MHC:peptide complex
true, but if a single AA in the TCR binding site, MHC molecule, or the peptide could result in the TCR no longer having affinity
is MHC diversity a good thing? why?
It is a good this because it allows for a greater number of pathogen-derived peptides to be presented during any infection.
there is no difference in disease susceptibility or resistance within different MHC alleles T/F
false, some MHC isotypes may not bind to a particular peptide very well. In other cases, a particular MHC isotype may be able to present a variety of antigenic peptides
sweaty t shirt experiment conclusion
women smelt mens sweaty T shirts and the ones who had MHC most different to them smelt the best
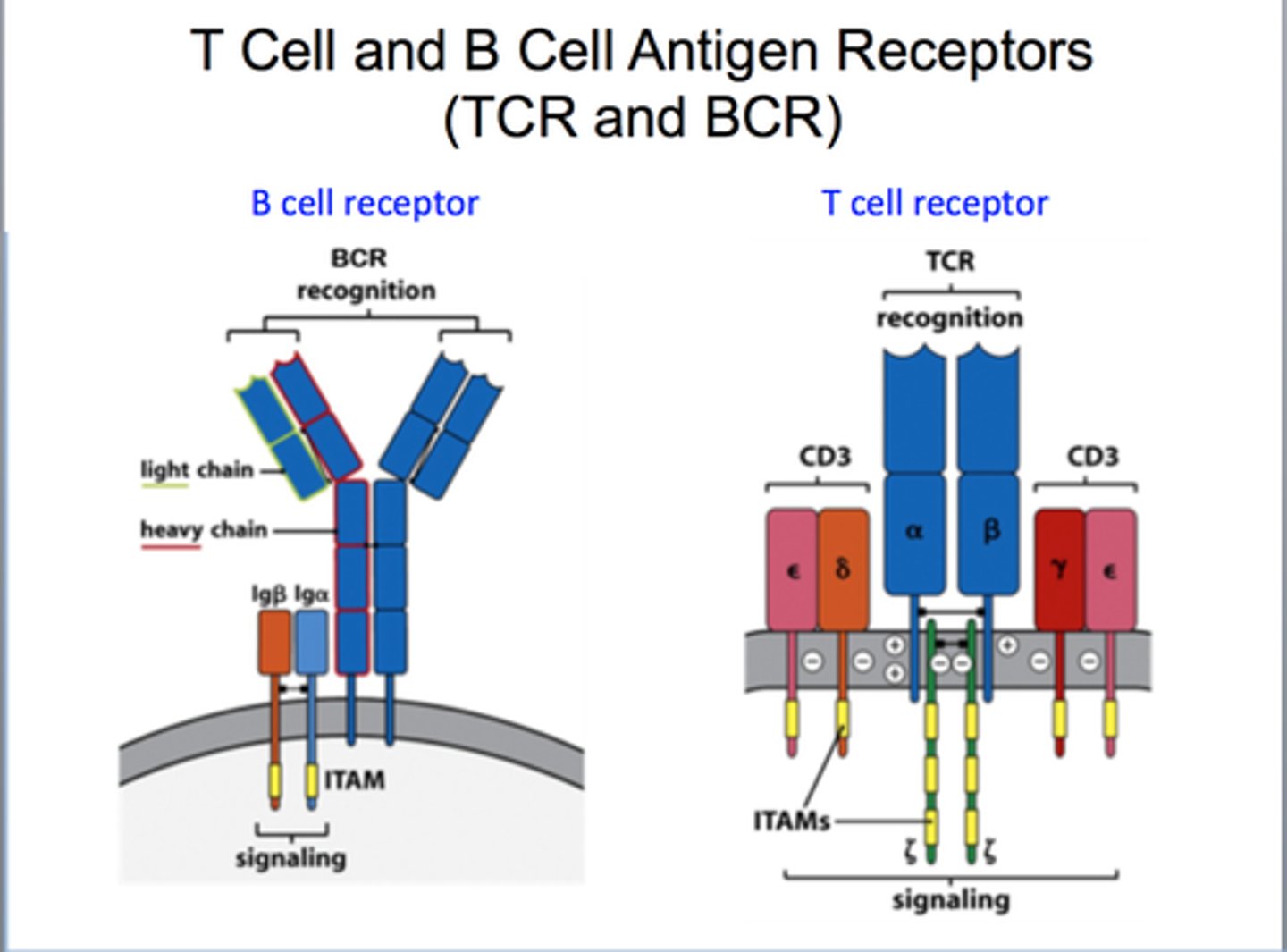
BCR complex consist of
Ig and 2 other proteins
Iga and B have long cytoplasmic tails. These proteins are required for all BCR signaling (and to get newly made Ab from the ER to the cell surface)
Ig heavy chain have no cytoplasmic signaling tail portion to interact with intracellular signaling proteins
Pro-B cell
Heavy chain VDJ rearrangement
Light chain is in germline config, but a surrogate L chain is produced
(in bone marrow)
Pre-B cell
H chain is rearranged, binds to surrogate Light chains and forms a pre-BCR
if it is able to form the pre-BCR on its surface the real light chain undergoes VJ rearrangement
(In bone marrow)
what are productive and nonproductive rearrangements
productive- when the H chain rearranges and generates a functional H chain polypeptide
nonproductive- H chain is not generated
what happens if a Pro-B cell fails to generate a productive rearrangement
they are signaled to die via apoptosis, and pre-B cells are not formed.
this is the 1st checkpoint in B cell development
why is rearrangement of the H chain genes an inefficient process
junctional diversity (random addition of nucleotides) at VDJ joints can change the reading frame. 1/3 chance of maintaining the reading frame
what does the surrogate light chains consist if
it resembles a real Light chain and consists of two proteins, but these proteins are encoded by conventional genes which do not rearrange
what happens when the Pre BCR correctly forms
it dimerizes (with another pre-BCR) and signals the cell (via Ig-a and B) to further differentiate and to rearrange its real L chains in order to become immature B cell
what is the 2nd checkpoint in B cell development and the two ways it can go
its between pre-b and immature b
if light chain rearrangement is productive then the cell becomes a immature B cell that expresses IgM but NOT IgD
if it does not rearrange productively then the cell does not form BCR and dies by apoptosis
after the second checkpoint when a preB cell forms a BCR and becomes an immature B cell what Abs does it express
IgM but NOT IgD
what amount of your developing B cells go through apoptosis in the bone marrow and why
over half
this is because they cant rearrange and produce functional heavy and light chain genes
encapsulated bacteria
have a polysaccharide capsule that surrounds the microbe and renders them resistant to phagocytosis. this is because they decrease the ability of complement and phagocytic receptor binding to bacteria
Most of these microbes are pyogenic (pus-forming)
these cause alot of ear, lung, and sinus infections
solution is to produce opsonizing abs that coat bacteria
X-linked agammaglobulinemia
immunodeficiency disease where there is an absence of B cells and thus an inability to make antibodies. caused by a mutation of the BTK gene. because the cell cant pass 2nd checkpoint
patients suffer from recurrent infections with extracellular pyogenic bacteria beginning early in life
X linked recessive trait
No tonsils/small lymph nodes (tonsils are 80-90% B cells)
brutons tyrosine kinase BTK
becomes activated by the Pre-BCR on Pre-B cells. when BTK phosphorylates tyrosine residues on other signaling proteins, it signals L chain rearrangement so the cell can become an immature B cell
what is the treatment of X-linked agammaglobulinemia
gamma globulin (ab) collected and pooled from about 1000 normal healthy human plasma donations