AP Chem Unit 1
1/59
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
60 Terms
Crystalline
atoms or molecules are in patterns with long-range, repeating order
Amorphous
atoms or molecules do not have any long-range order
Liquids
atoms or molecules pack about as closely as they do in solid matter, but are free to move relative to each other
Fixed volume, but not fixed shape
Assume shape of container
Gas
atoms or molecules that have a lot of space between them and are able to move relative to one another
compressible
Solids
molecules are packed closely together in fixed locations
fixed volume and rigid shape
Pure substance
made up of only one component
can be individual atoms, or groups of atoms joined tg
Element
substance that cannot be chemically broken down into simpler substances (type of pure substance)
Compound
substance composed of two or more elements (type of pure substance)
mixture
made up of two or more components in proportions that can vary from one sample to another
Heterogeneous mixture:
comp varies from region to another
Homogenous
same composition throughout
Decanting
ex. water and sand - carefully pouring off the water into another container
allowing denser to settle at the bottom and pouring the rest off
Distillation
mixture is heated to boil off the more volatile liquid
Volatile
easily vaporizable
When composed of an insoluble solid and a liquid, separated by
filtration
Physical changes
changes that alter only state or appearance, not composition
can change state, as long as it is the same atoms
Chemical changes
atoms rearrange, transforming og substances into diff substances
Physical property
a property that a substance displays w/o changing its composition
Chemical property
a property that a substance displays only by changing its composition via a chemical change
ex. corrosiveness, flammability, acidity, toxicity, etc.
only chemical examination can confirm whether a particular change is physical or chemical
Kelvin
measure of temperature
also called absolute scale bc avoids negative temp
0k is coldest temp possible (-273.15 C or -459 F)
K = C + 273.15
temperature
measure of the average kinetic energy of the atoms or molecules that compose the matter
Intensive property
one that is independent of the amount of the substance
Extensive property
one that depends on the amount of the substance
ex. if you know only the mass of a sample of gold, that info alone will not allow you to identify it as gold
Accuracy
how close the measured value is to the actual value
Precision
how close a series of measurements are to one another or how reproducible they are
Wave particle duality
certain properties of light are best described by thinking of it as a wave, while others are best described thinking of it as a particle
Electromagnetic radiation
type of energy embodied i n oscillating electric and magnetic fields-what light is idk
Electric field
a region of space where an electrically charged particle experiences a force
Waves
characterized by amplitude and wavelength
Amplitude
vertical height of a crest or depth of a trough
determines light’s intensity or brightness - the greater the amplitude the greater the intensity
Wavelength (λ):
distance between adjacent crests
measured in meters
Frequency (v):
number of cycles that pass through a stationary point in a given period of time
inversely prop to wavelength - the farther apart the crests, the lower the number of crests that pass a fixed location per unit time
relationship between frequency and wavelength
equation v = c/λ
as wavelength increases, the frequency decreases
Visible light
light that can be seen by the human eye
wavelength determines color
Electromagnetic spectrum
all wavelengths of electromagnetic radiation
Electromagnetic spectrum from shortest to longest wavelength
Gamma ray
x ray
ultra violet radiation
visible light
infrared
microwaves
radio waves
Visible light
ranging from violet (shorter wavelength higher energy) to red (longer wavelength lower energy)
Infrared Radiation
heat when you place your hand near a hot object
interference
when waves interact with each other in a characteristic way like building each other up or aligning
constructive interference
if two waves of equal amplitude are in phase when they interact → wave with twice the amplitude
destructive interference
if two waves are out of phase, the waves cancel
out of phase
they align so that the crest from one source overlaps with the trough from the other source
Diffraction
when a wave encounters an obstacle or a slit that is comparable in in size to its wavelength, it bends or diffracts around it
interference pattern
the result of the diffraction of light through two slits separated by a distance comparable to the wave length of light coupled with interference results
Photoelectric effect
observation that many metals emit electrons when light shines upon them
sceintists found that a high frequency, low intensity light produces electrons without the predicted lag time
Threshold frequency
threshold below which no electrons are emitted from the metal, no matter the duration
Albert Einstein
determined that light energy must come in packets
suggested that light was lumpy rather than continuously variable
Energy is
quantized
Quantized Model
electrons orbit the nucleus in fixed paths (called energy levels or shells)
each energy level (n) has specific radii (energy)
these energy levels are quantized
valence shell
the outermost principal energy level with electrons
valence electrons
electrons located in the valence shell
Niels Bohr's Postulates for an atom
electrons can only exist in specific energy levels (n), not inbetween
shells closest to the nucleus are more stable (less energy)
electrons can absorb a photon with exactly the right amount of energy that would put it in a higher energy level
electrons in excited states will emit photons when they spontaneously drop to lower energy levels
ground state
the lowest possible energy state for an atom
excited state
when the electrons are put in a higher energy level
electrons in excited states will emit photons when they spontaneously drop to lower energy levels
Law of Conservation of Mass
mass/matter cannot be created nor destroyed; the mass before and after a chemical reaction is the same
Law of Definite Proportions/Constant Composition
a pure chemical compound always contains the same elements in the same fixed proportion by mass
Law of Multiple Proportions
if two elements form more than one compound the mass of one element will combine with the mass of the other in a ratio of small whole numbers
isotopic notation
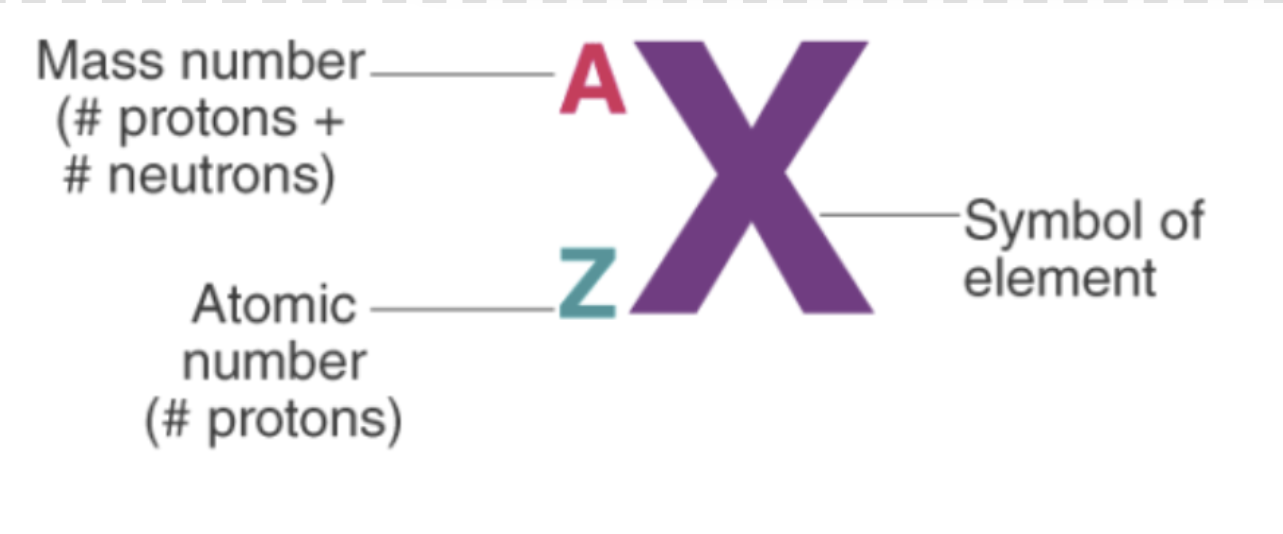
isotopes
atoms of the same element with different masses
isotopes have different numbers of neutrons
isotopes have different numbers of neutrons
weighted average
atomic mass=(%1)(mass 1)+(%2)(mass 2)+(%3)(mass 3).....