GI polyps and nonpolyposis cancer syndromes
1/54
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
55 Terms
Types of colon polyps
1. Adenoma
2. Hyperplastic
3. Inflammatory
4. Hamartomatous
Adenoma
Pre-cancerous
- tubular adenoma (TA)
- tubulovillous adenoma (TVA) !!worst!!
- serrated adenoma
Hyperplastic
Benign
Inflammatory
This is not a real polyp
Hamartomatous
Benign
- juvenile polyps (JPS)
- Peutz-Jeghers (PJ) polyps
Polyp descriptors
1. Sessile
2. Pedunculated
3. Dysplasia
Sessile
Dome-shaped or flattened
**more difficult to remove, considered higher risk
Pedunculated
Hanging from the colon wall on a stalk
Dysplasia
How atypical do the cells look?
High-grade vs low-grade
Types of gastric cancers
1. Intestinal
2. Diffuse
Intestinal gastric cancer
1. Adenocarcinomas ~95%
2. Gastrointestinal stromal Tumor (GIST)
**almost never genetic
Diffuse gastric cancer
1. Signet ring cell
*Cancer of the lining of the stomach
*Highly metastatic
*removal of stomach
**may be genetic
Stomach cancer genes
High risk: CDH1
Moderate risk: APC, BMPR1A, EPCAM, MLH1, MSH2, MSH6, PMS2, SDHB, SDHD, SMAD4, STK11, TP53
Possible risk: CTNNA1, CHEK2, KIT, NF1, PDGFRA, SDHC
Hereditary diffuse gastric cancer syndrome (gene)
CDH1
CDH1
- chromosome 16
- AD inheritance
Features of HDGC
- diffuse gastric cancer (67-70% lifetime risk AMAB, 40-83% risk AFAB)
- average age of onset is 38y
- lobular breast cancer (41-60% lifetime risk)
- colorectal cancer
Clinical diagnostic criteria for HDGC
1. Diffuse gastric cancer and a family history of one or more first- or second-degree relatives with gastric cancer OR
2. A personal and/or family history of diffuse gastric cancer diagnosed before 40y OR
3. A personal and/or family history of diffuse gastric cancer and lobular breast cancer, one diagnosed before 50y
HDGC management
- consider gastrectomy, endoscopy every 6-12 months
- breast awareness (18), biannual clinical breast exam (25), annual mammography/MRI (30)
- consider increased colonoscopy frequency
Do mammograms prevent cancer?
NO
Do colonoscopies prevent cancer?
YES
Types of pancreatic cancer
1. Exocrine
2. Endocrine
Endocrine pancreatic cancer
- PNETs or Islet cell tumors (very rare <4%)
*tough to determine malignancy
**associated with a better prognosis
Exocrine pancreatic cancer
- Adenocarcinoma (~95%)
- Begins in glands surrounding the ducts of the pancreas
**Highly lethal
** Symptoms are nonspecific, inability to screen
**7% 5-year survival
Pancreatic cancer genes
High risk: APC, BRCA1, BRCA2, CDKN2A, CDK4, EPCAM, MEN1, MLH1, MSH2, MSH6, PALB2, PMS2, STK11, TP53, VHL
Moderate risk: ATM
Melanoma-pancreatic cancer syndrome (gene)
CDKN2A
Familial atypical multiple mole melanoma (FAMMM)
CDKN2A
CDKN2A
- tumor supressor gene
- encodes two proteins: p16INK4a + p14ARF
- chromosome 9
- AD inheritance
Features of FAMMM
1. melanoma (28-76% lifetime risk)
2. pancreatic adenocarcinoma (17% lifetime risk)
Clinical diagnostic criteria for FAMMM
1. malignant melanoma in one or more first- or second-degree relatives
2. High total body nevi (>50) including some of which are clinically atypical
3. Nevi with certain histologic features on microscopy
CDKN2A (p16INK4a)
1. Cutaneous melanoma
2. Pancreatic cancer
CDKN2A (p14ARF)
1. Cutaneous melanoma
2. Pancreatic cancer
3. Neural system tumors (astrocytoma) + nerve sheath tumors
FAMMM management
- monthly skin self-exams (10), sun protection
- pancreatic cancer screening (40 or -10y)
Colon cancer genes
High risk: APC, BMPR1A, EPCAM, MLH1, MSH1, MSH6, MUTYH, PMS2, PTEN, SMAD4, STK11, TP53
Moderate risk: CDH1, GALNT12
Possible risk: AXIN2, ATM, BLM, GREM1/SCG5, MSH3, NTHL1, POLD1, POLE, RPS20
Hereditary nonpolyposis colorectal cancer (HNPCC)
- aka Lynch Syndrome
- prevalence 1 in 279
- AD inheritance
HNPCC genes
MLH1
MSH2
MSH6
PMS2
EPCAM
HNPCC-associated cancers (AMAB)
1. Stomach
2. Colorectal
3. Pancreatic
4. Urinary tract
5. Prostate
HNPCC-associated cancers (AFAB)
1. Stomach
2. Colorectal
3. Pancreatic
4. Urinary tract
5. Ovarian + uterine
Features of HNPCC
- predominantly right-sided colorectal cancer
- multiple tumors
- more likely to see mucinous, signet-ring, infiltrating lymphocytes, Chrohn's-like lymphocytic reaction, medullary growth pattern
Average lifetime risk for colorectal cancer
5%
Untreated lifetime risk for colorectal cancer with HNPCC
up to 50%
Genetic testing criteria for HNPCC
1. Known HNPCC PV in the family
2. Personal history of an HNPCC-related cancer <50y
3. Family history of HNPCC-related cancer <50y or multiple family members diagnosed at any age
HNPCC Management
- colonoscopy every 1-2 years (20), aspirin
- annual pelvic exam (30), consider hysterectomy
- consider BSO (40), consider ultrasound (30), OCP
- upper endoscopy every 2-5 years (30)
- consider annual urinalysis (30)
- annual pancreatic cancer screening (50 or -10y)
- annual physical/neurological exam (25)
- consider regular DRE/PSA (45)
Time for colon polyp to turn into cancer
10 years
**may be as low as 1-2 years with HNPCC
HNPCC clinical diagnostic criteria (Amsterdam criteria)
3+ family members with HNPCC-related cancers
2+ successive affected generations
1+ HNPCC-related cancers diagnosed before age 50y
Naming scheme: HNPCC
Family meets Amsterdam criteria
*clinical diagnosis
Naming scheme: Muir-Torre
HNPCC + sebaceous gland tumors/keratoacanthomas
Naming scheme: Turcot variants
HNPCC + brain tumor (gliomas)
**Not to be confused with Turcot syndrome (variant of FAP)
Naming scheme: Lynch syndrome
Genetic mutation identified in a mismatch repair gene
*genetic/molecular diagnosis
Mismatch repair (MMR) pathway
MSH2-MSH6
MLH1-PMS2
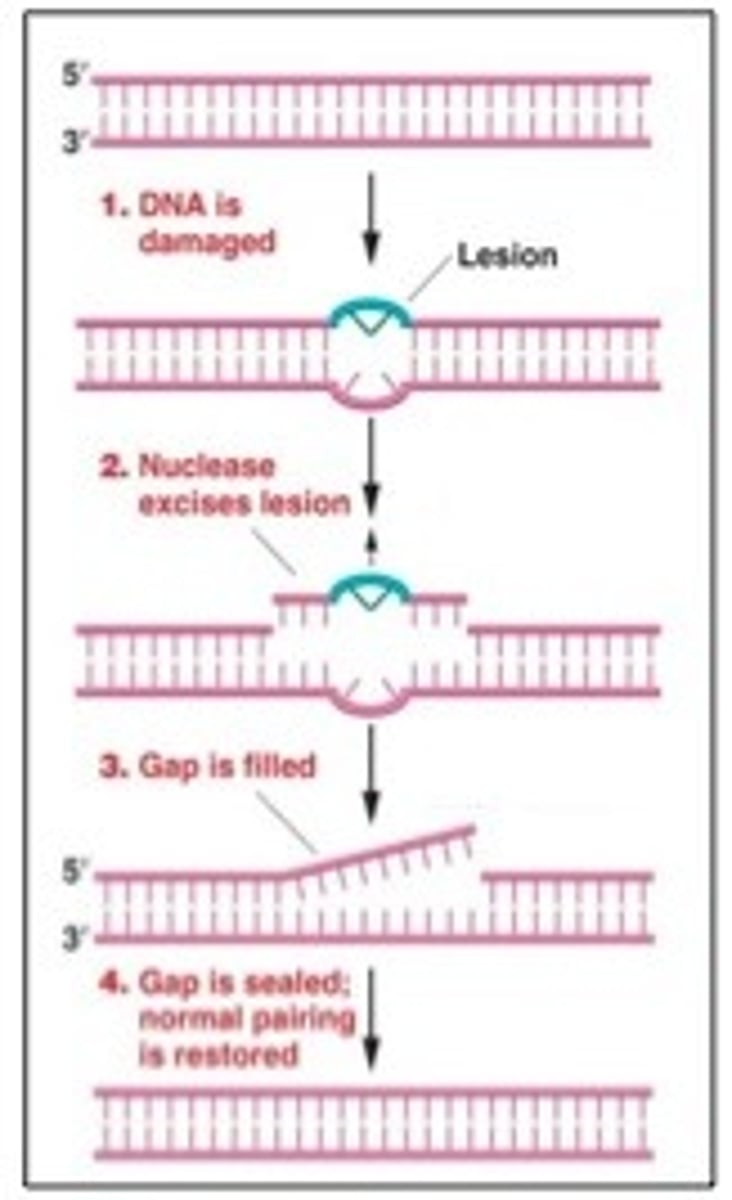
Microsatellite instability testing
- presence of MSI in a tumor indicates a poorly functioning DNA MMR process
- 10% of SPORADIC colorectal cancers display MSI
- 95% of LYNCH SYNDROME colorectal cancers display MSI
Immunohistochemistry (IHC) staining
- loss of staining for specific proteins that are associated with hereditary cancer genes is suggestive of a mutation
MLH1 considerations
- BRAF (V600E) mutation
- Somatic promoter hypermethylation
BRAF (V600E) mutation
Somatic promoter hypermethylation
Constitutional mismatch repair deficiency (CMMRD) syndrome
- biallelic germline mutation in the MMR pathway
- severe autosomal recessive childhood onset cancer predisposition syndrome
- associated with increased risk for childhood diagnosis (~7y) of brain tumors, GI cancers, hematologic cancers
- associated with café-au-lait macules, polimatricomas (benign skin lesion), colon polyposis