GCSE Chemistry: Chemical Changes, Reactions, and Electrolysis
1/50
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
51 Terms
What is the reaction of metals with oxygen called?
Oxidation reactions
What do metals form when they react with oxygen?
Metal oxides
What is the definition of reduction in terms of oxygen?
Loss of oxygen
What is the definition of oxidation in terms of oxygen?
Gain of oxygen
How are metals arranged in the reactivity series?
In order of their reactivity based on their tendency to form positive ions
Which metals are more reactive: potassium or copper?
Potassium
What happens when a more reactive metal displaces a less reactive metal?
The more reactive metal can displace the less reactive metal from a compound.
How can metals less reactive than carbon be extracted?
By reduction with carbon
What does the acronym OIL RIG stand for?
Oxidation Is Loss, Reduction Is Gain (of electrons)
What is produced when acids react with metals?
A salt and hydrogen
What type of reaction occurs when acids react with metals?
Redox reactions
What is the general equation for the reaction of an acid with a metal?
Acid + Metal -> Salt + Hydrogen
What is produced when an acid is neutralized by an alkali?
A salt and water
What is the equation for the neutralization of an acid by an alkali?
Acid + Alkali -> Salt + Water
What do acids produce when they react with metal carbonates?
Salts, water, and carbon dioxide
What type of salts does hydrochloric acid produce?
Chlorides
What type of salts does nitric acid produce?
Nitrates
What type of salts does sulfuric acid produce?
Sulfates
What is the first step in making soluble salts from acids?
Add the chosen solid insoluble substance to the acid.
How do you know when the acid has been neutralized during salt production?
Excess solid sinks to the bottom.
What is the process called when you evaporate water to obtain salt crystals?
Crystallisation
What do acids produce in aqueous solutions?
H+ ions
What do alkalis produce in aqueous solutions?
OH- ions
What is the pH range of the pH scale?
0 to 14
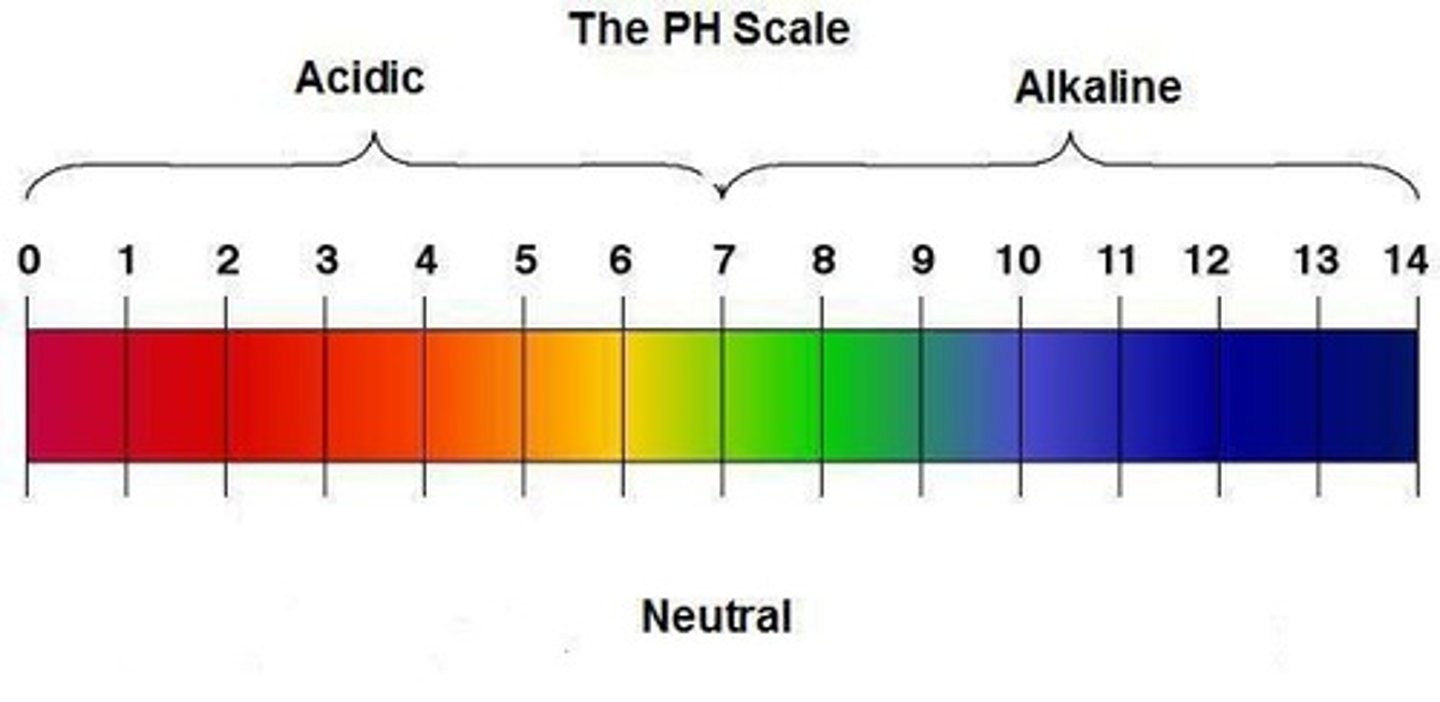
What pH value indicates a neutral solution?
pH 7
What pH value indicates an acidic solution?
pH < 7
What pH value indicates an alkaline solution?
pH > 7
What is the ionic equation for neutralisation?
H+(aq) + OH-(aq) -> H2O(l)
What is the purpose of titration in chemistry?
To measure the volumes of acid and alkali solutions that react with each other.
What is the first step in carrying out a titration?
Wash the burette using dilute hydrochloric acid and then water.
How much acid should be filled in the burette for titration?
100 cm³
How much alkali is typically added to the conical flask in a titration?
25 cm³
What indicator is commonly used in titrations?
Phenolphthalein
What color does phenolphthalein turn in an alkaline solution?
Pink
What is the titre in a titration?
The volume of acid needed to exactly neutralise the alkali.
What is the conversion factor between dm³ and cm³?
1 dm³ = 1000 cm³
What is the relationship between moles and grams for a substance?
One mole of a substance in grams is the same as its relative atomic mass in grams.
What characterizes a strong acid?
It is completely ionised in aqueous solution.
Give an example of a strong acid.
Hydrochloric acid, nitric acid, or sulfuric acid.
What characterizes a weak acid?
It is partially ionised in aqueous solution.
Give an example of a weak acid.
Ethanoic acid, citric acid, or carbonic acid.
How does the strength of an acid relate to its pH?
Stronger acids have lower pH values.
What happens to the H+ concentration when pH decreases by one unit?
The H+ concentration increases by a factor of 10.
What is the process of electrolysis?
Breaking down a substance into elements by passing a current through it when it is molten or in solution.
What happens to positively charged ions during electrolysis?
They move to the negative electrode (cathode).
What happens to negatively charged ions during electrolysis?
They move to the positive electrode (anode).
What is produced at the cathode during the electrolysis of molten ionic compounds?
The metal.
What is produced at the anode during the electrolysis of molten ionic compounds?
The non-metal.
What is required to extract metals more reactive than carbon?
Electrolysis of molten compounds.
What is the role of cryolite in the electrolysis of aluminium oxide?
It lowers the melting point of aluminium oxide, making the process cheaper.
What are the order of reactivity series
Potassium
Sodium
Lithium
Calcium
Magnesium
Aluminium
Carbon
Zinc
Iron
Hydrogen
Copper